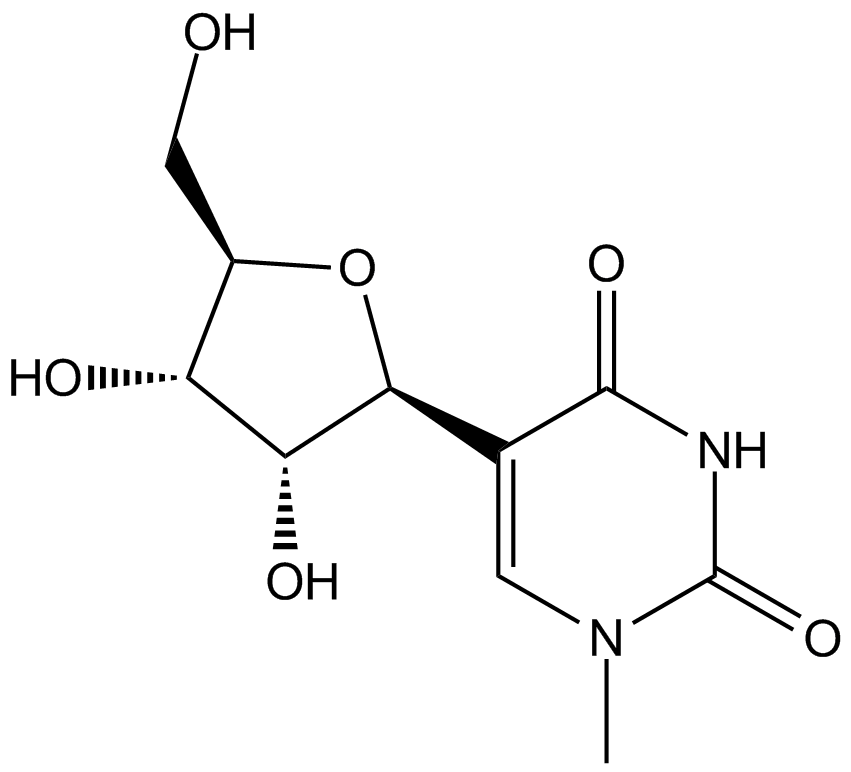
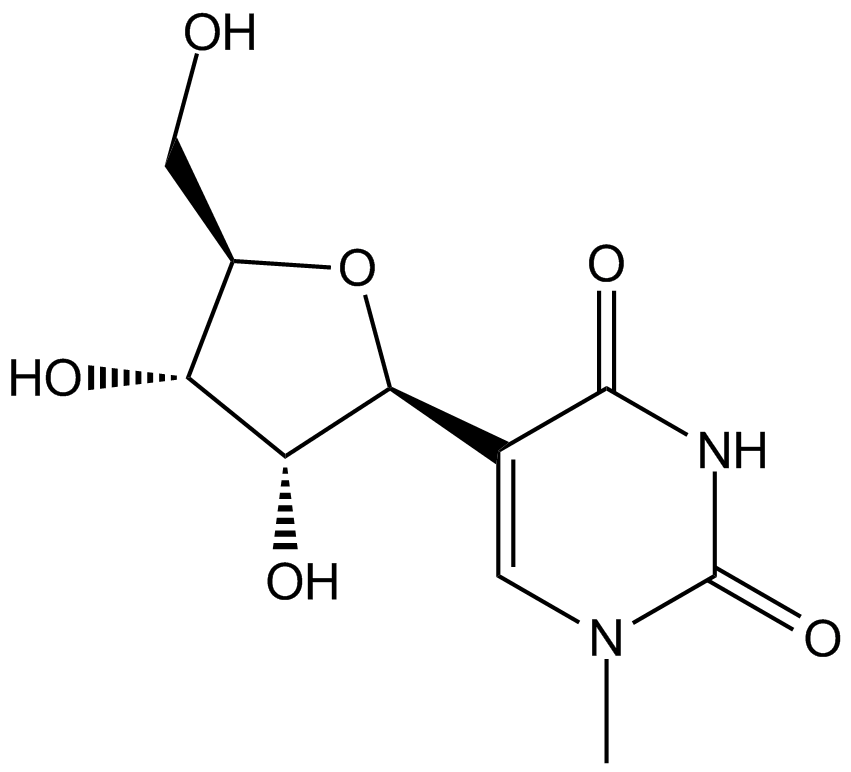
N1-Methylpseudouridine

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
N1-Methylpseudouridine is a modified nucleoside used for enhancing mRNA translation. Incorporation of N1-Methylpseudouridine into mRNA turns off the immune/eIF2α phosphorylation-dependent inhibition of translation, and increases ribosome pausing and density on the mRNA, thus dramatically facilitating the translation process. N1-Methylpseudouridine outperforms several other modified nucleosides (e.g. 5-Methylcytidine) and corresponding combinations in terms of translation capacity.
References:
1. Svitkin YV, Cheng YM, Chakraborty T, et al. N1-methyl-pseudouridine in mRNA enhances translation through eIF2α-dependent and independent mechanisms by increasing ribosome density. Nucleic Acids Research, 2017, 45(10): 6023-6036.
2. Andries O, Mc Cafferty S, De Smedt SC, et al N(1)-methylpseudouridine-incorporated mRNA outperforms pseudouridine-incorporated mRNA by providing enhanced protein expression and reduced immunogenicity in mammalian cell lines and mice. Journal of Controlled Release, 2015, 217: 337-344.
| Physical Appearance | Solution |
| Storage | Store at -20°C or below |
| M.Wt | 258.23 |
| Cas No. | 13860-38-3 |
| Formula | C10H14N2O6 |
| Solubility | ≥50 mg/mL in H2O with ultrasonic; ≥20 mg/mL in EtOH; ≥20.65 mg/mL in DMSO |
| Chemical Name | 5-((2S,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-1-methylpyrimidine-2,4(1H,3H)-dione |
| SDF | Download SDF |
| Canonical SMILES | O=C(N1)N(C)C=C([C@@H]2O[C@H](CO)[C@@H](O)[C@H]2O)C1=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Cell experiment:[2] | |
|
Cell lines |
Various mammalian cells including A549, BJ, C2C12, HeLa and primary keratinocytes |
|
Reaction Conditions |
24 h incubation |
|
Applications |
In various mammalian cells, mRNAs in the N1-Methylpseudouridine + 5-Methylcytidine group showed reduced cytotoxicity compared to the Pseudouridine + 5-Methylcytidine group. The mRNAs simultaneously modified by N1-Methylpseudouridine and 5-Methylcytidine also exhibited reduced activation of the intracellular innate immune response. |
| Animal experiment:[2] | |
|
Animal models |
7-week-old Balb/c mice |
|
Dosage form |
20 μg Once daily through intradermal or intramuscular routes by lipofection, for 21 days |
|
Applications |
N1-Methylpseudouridine alone and its combination with 5-Methylcytidine outperformed the current state-of-the-art Pseudouridine and its combination with 5-Methylcytidine, in terms of translation capacity. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Svitkin YV, Cheng YM, Chakraborty T, et al. N1-methyl-pseudouridine in mRNA enhances translation through eIF2α-dependent and independent mechanisms by increasing ribosome density. Nucleic Acids Research, 2017, 45(10): 6023-6036. 2. Andries O, Mc Cafferty S, De Smedt SC, et al N(1)-methylpseudouridine-incorporated mRNA outperforms pseudouridine-incorporated mRNA by providing enhanced protein expression and reduced immunogenicity in mammalian cell lines and mice. Journal of Controlled Release, 2015, 217: 337-344. |
|
质量控制和MSDS
- 批次:
化学结构