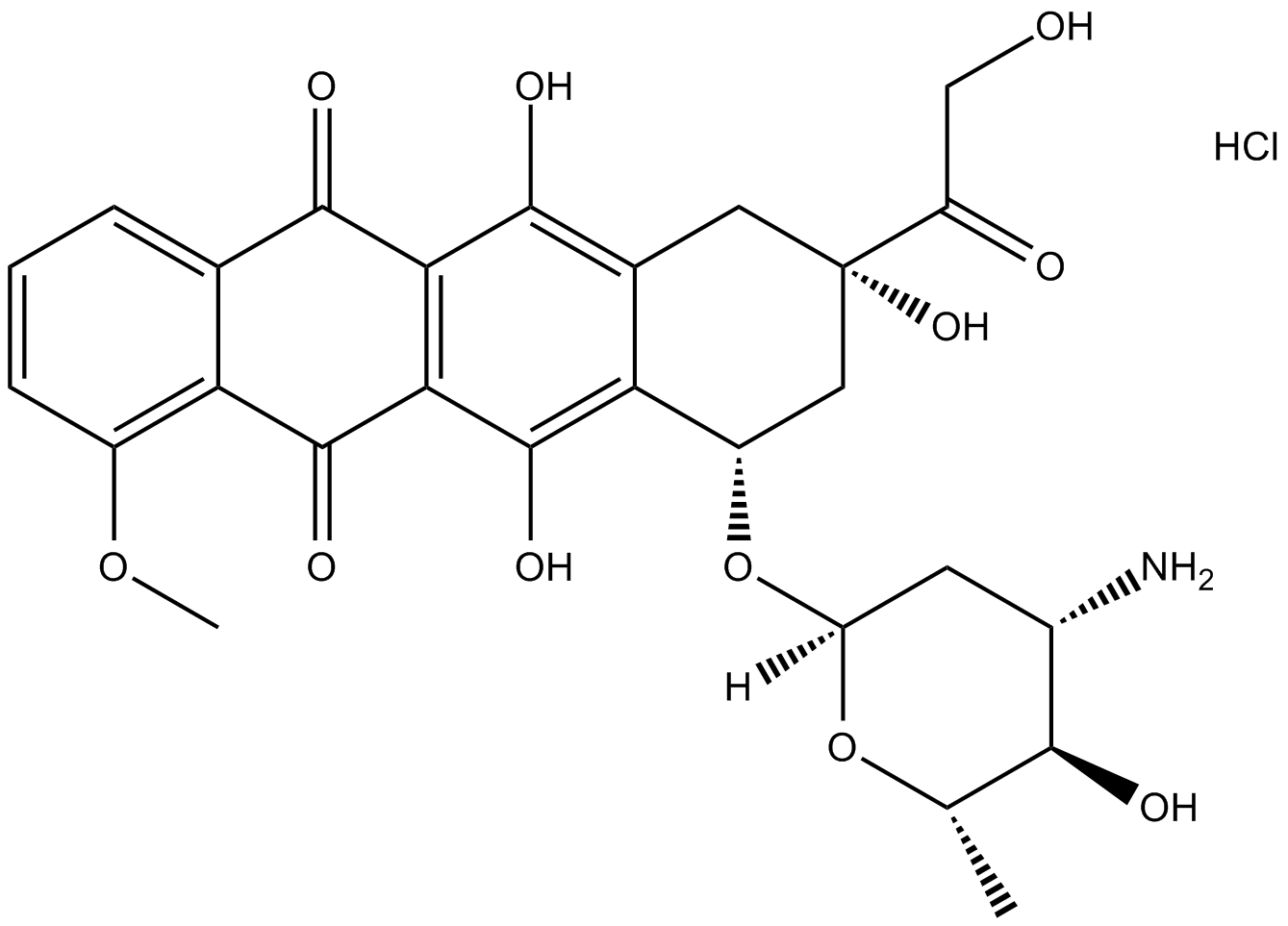
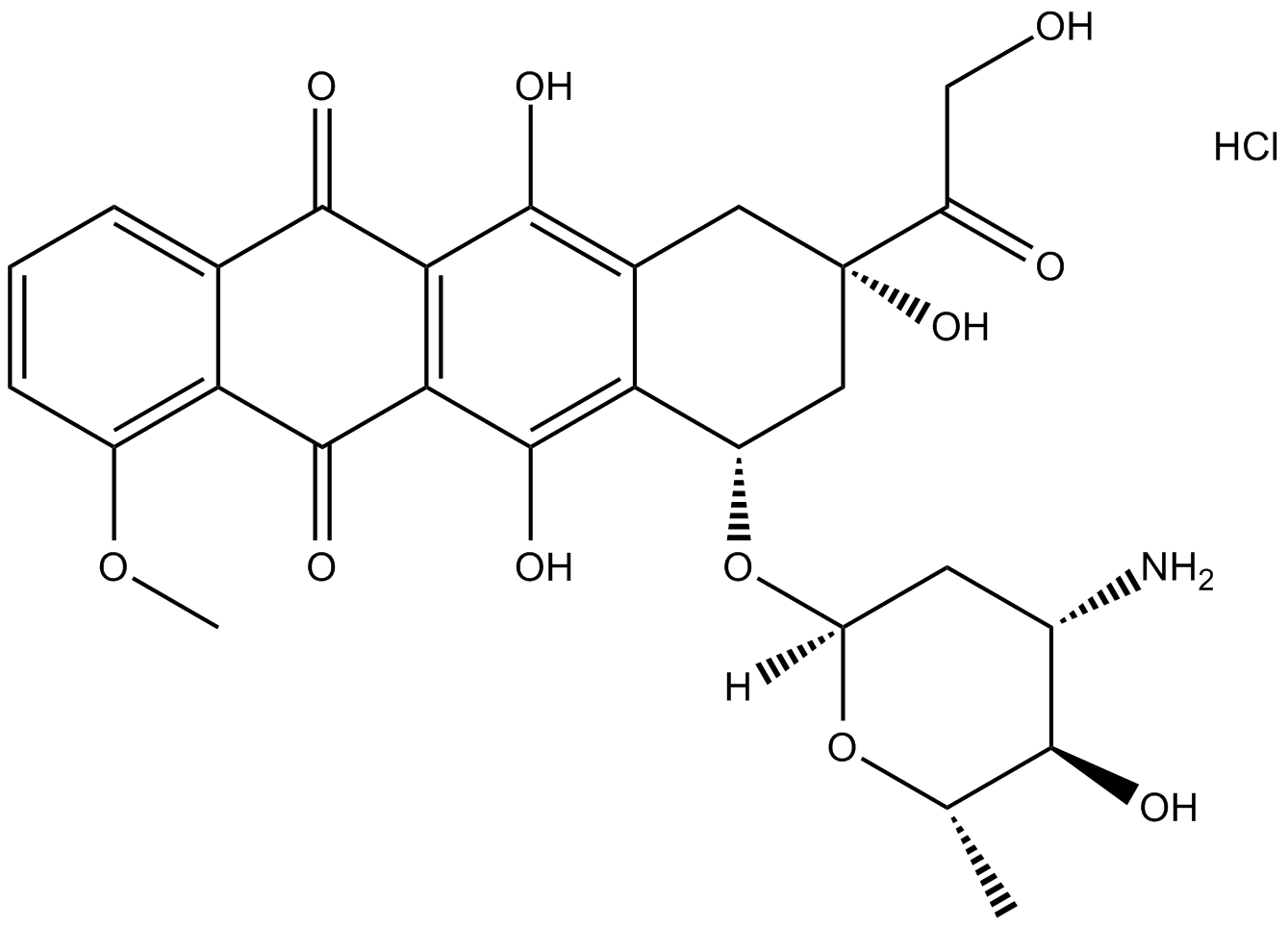
Epirubicin HCl

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Epirubicin是一种DNA拓朴异构酶(TOPII)抑制剂[1]。
Epirubicin属于Anthracylines化学家族,是一种DNA拓朴异构酶抑制剂。Epirubicin可抑制DNA拓扑异构的再连接途径,导致5’磷酸-DNA复合体(剪切复合体)稳定化。这些损伤具有细胞毒性,可引起DNA损伤应答激活,并可能导致细胞凋亡。因此,Epirubicin通常用于癌症治疗。不幸的是,Epirubicin也存在遗传毒性副作用,包括可引发白血病的染色体易位形成[1]。
Epirubicin是一种用于治疗骨肉瘤的化疗剂。Epirubicin可通过诱发细胞凋亡,抑制肿瘤生长。相反地,Epirubicin可在OS细胞中通过活化NF-κB减少细胞凋亡。据报道,epirubicin与浅蓝菌素结合后可提高体内和体外的抗肿瘤活性[2]。
参考文献:
[1] Ian G. Cowell, Caroline A. Austin. Mechanism of Generation of Therapy Related Leukemia in Response to Anti-Topoisomerase II Agents. International Journal of Environmental Research and Public Health. 2012 (9): 2075-2091.
[2] Z. L. LIU, G. WANG, Y. SHU, P.A. ZOU, Y. ZHOU and Q.S. YIN. Enhanced antitumor activity of epirubicin combined with cerulenin in osteosarcoma. Molecular Medicine Reports. 2012 (5): 326-330.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 579.98 |
| Cas No. | 56390-09-1 |
| Formula | C27H29NO11·HCl |
| Solubility | ≥29.00 mg/mL in DMSO; ≥10.12 mg/mL in EtOH with gentle warming and ultrasonic; ≥40.33 mg/mL in H2O with gentle warming |
| Chemical Name | (7S,9S)-7-[(2R,4S,5R,6S)-4-amino-5-hydroxy-6-methyloxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione;hydrochloride |
| SDF | Download SDF |
| Canonical SMILES | CC1C(C(CC(O1)OC2CC(CC3=C(C4=C(C(=C23)O)C(=O)C5=C(C4=O)C=CC=C5OC)O)(C(=O)CO)O)N)O.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Cell experiment:[1] | |
|
Cell lines |
Human-liver derived hepatoma G2 cells (Hep G2 cells) |
|
Reaction Conditions |
0 ~ 12 μg/ml epirubicin HCl for 24, 48 or 72 h incubation |
|
Applications |
Epirubicin HCl produced a concentration- and time-dependent cytotoxicity to Hep G2 cells. The mechanism of cytotoxicity of epirubicin HCl (IC50 value of 1.6 μg/ml within 24 h) appeared to involve a production of free radical species since activities of free radical scavenging enzymes (SOD, catalase, Se-dependent GPx) were increased. |
| Animal experiment:[2] | |
|
Animal models |
Nude mice xenografted with human breast carcinoma cell line R-27 |
|
Dosage form |
3.5 or 7 mg/kg Administered intravenously every 4 d for 3 times |
|
Applications |
Epirubicin treatment alone suppressed tumor growth in a dose-dependent manner. At a dose of 3.5 mg/kg, epirubicin alone suppressed tumor mass of human breast tumor xenograft R-27 by 74.4 %. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Ozkan A, Fişkin K. Epirubicin HCl toxicity in human-liver derived hepatoma G2 cells. Polish Journal of Pharmacology, 2004, 56(4): 435-444. 2. Asanuma F, Yamada Y, Kawamura E, et al. Antitumor activity of paclitaxel and epirubicin in human breast carcinoma, R-27. Folia Microbiologica (Praha), 1998, 43(5): 473-474. |
|
质量控制和MSDS
- 批次:
化学结构