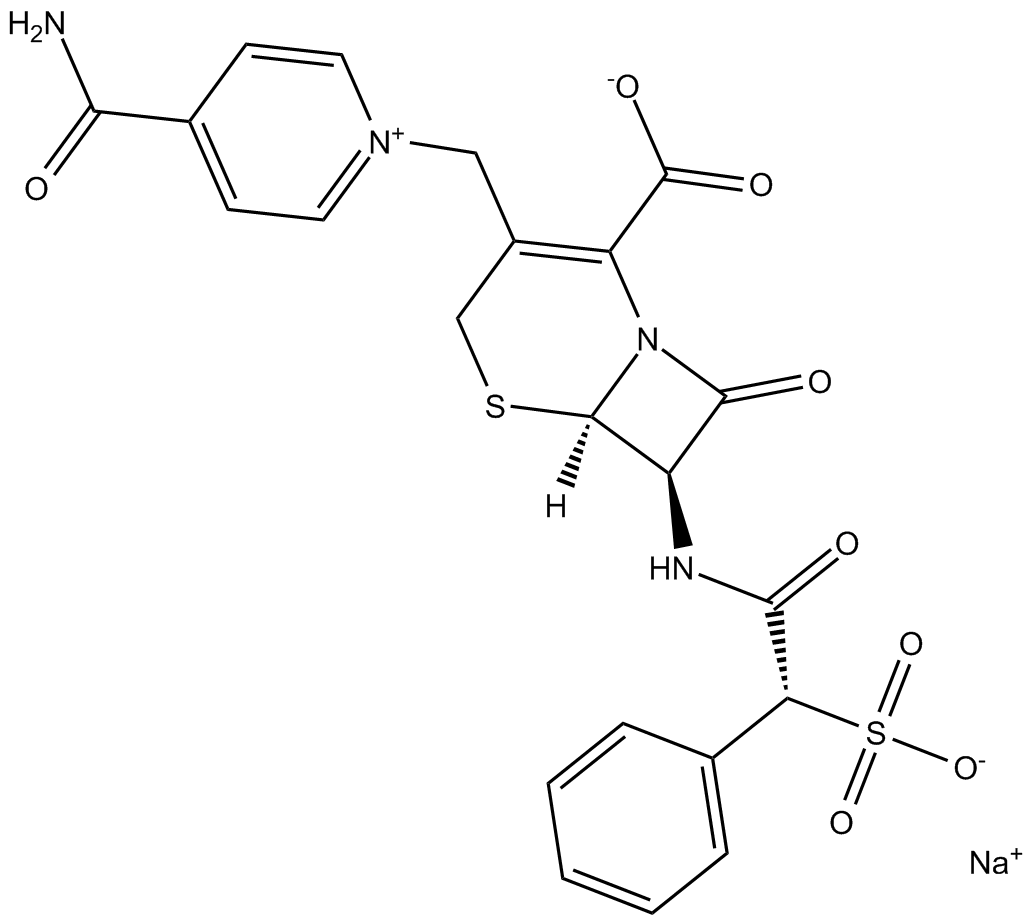
Cefsulodin (sodium salt)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Cefsulodin, formerly named as SCE-129, is a cephalosporin with a spectrum of antibacterial activity against Staphylococcus aureus, Pseudomonas aeruginosa, and most other gram-positive cocci [1]. Cefsulodin shows little activity against other species of Acinetobacter spp., Pseudomonas, or members of the Enterobacteriaceae [1]. Cefsulodin is a β-lactam antibiotic that involved in lysing actively-growing E. coli by specifically binding to the intermembrane proteins, penicillin-binding proteins 1a and b, whose transglycosylase and transpeptidase activities are involved in cell elongation and septation [2].
Cefsulodin was active against P. aeruginos. Cefsulodin was active against penicillinase-producing strains of S. aureus. The MICs of cefsulodin for Pseudomonas aeruginosa and its mutants Pseudomonas aeruginosa PAO4089 were 0·78 and 12· mg/l [3]. Cefsulodin was active in minimum inhibitory concentrations (MICs) of 0.5 to 64 μg/ml. Cefsulodin was active against P. diminuta, P. maltophilia, P. paucimobilis, and P. pseudoalcaligenes with MICs of 1-32 μg/ml. Cefsulodin was not hydrolyzed by the β-lactamase induced in P. aeruginosa by growth in the presence of benzylpenicillin and was a poor substrate for β-lactamases from Enterobacter cloacae and Proteus morganii [4].
References:
[1] Barry A L, Jones R N, Thornsberry C. Cefsulodin: antibacterial activity and tentative interpretive zone standards for the disk susceptibility test[J]. Antimicrobial agents and chemotherapy, 1981, 20(4): 525-529.
[2] Jacoby G H, Young K D. Cell cycle-independent lysis of Escherichia coli by cefsulodin, an inhibitor of penicillin-binding proteins 1a and 1b[J]. Journal of bacteriology, 1991, 173(1): 1-5.
[3] Bryan L E, Kwan S, Godfrey A J. Resistance of Pseudomonas aeruginosa mutants with altered control of chromosomal beta-lactamase to piperacillin, ceftazidime, and cefsulodin[J]. Antimicrobial agents and chemotherapy, 1984, 25(3): 382-384.
[4] King A, Shannon K, Phillips I. In vitro antibacterial activity and susceptibility of cefsulodin, an antipseudomonal cephalosporin, to beta-lactamases[J]. Antimicrobial agents and chemotherapy, 1980, 17(2): 165-169.
| Storage | Store at -20°C |
| M.Wt | 554.5 |
| Cas No. | 52152-93-9 |
| Formula | C22H19N4O8S2·Na |
| Solubility | ≥22.75 mg/mL in DMSO with gentle warming; insoluble in EtOH; ≥49.3 mg/mL in H2O |
| Chemical Name | 4-(aminocarbonyl)-1-[[(6R,7R)-2-carboxy-8-oxo-7-[[(2R)-2-phenyl-2-sulfoacetyl]amino]-5-thia-1-azabicyclo[4.2.0]oct-2-en-3-yl]methyl]-pyridinium inner salt, monosodium salt |
| SDF | Download SDF |
| Canonical SMILES | O=C(N[C@@H]1C(N2[C@]1([H])SCC(C[N+]3=CC=C(C(N)=O)C=C3)=C2C([O-])=O)=O)[C@H](S([O-])(=O)=O)C4=CC=CC=C4.[Na+] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |