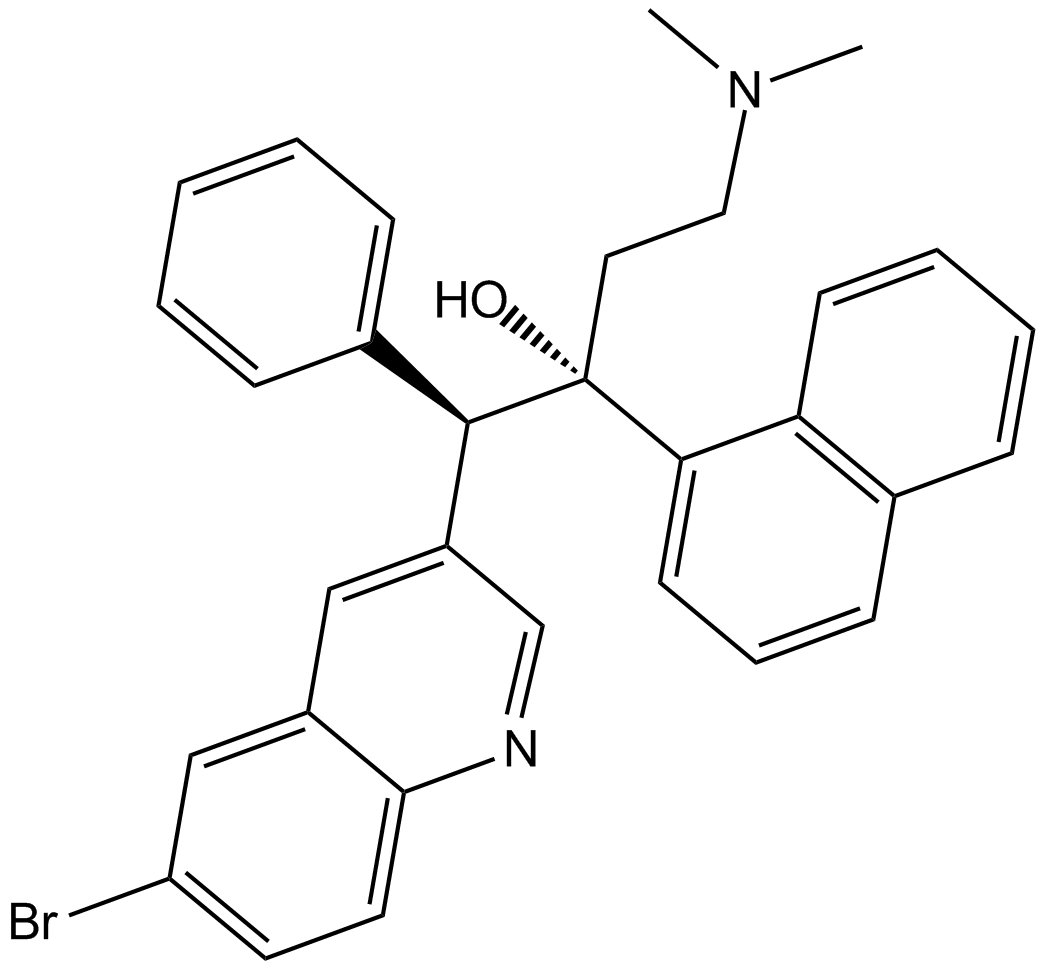
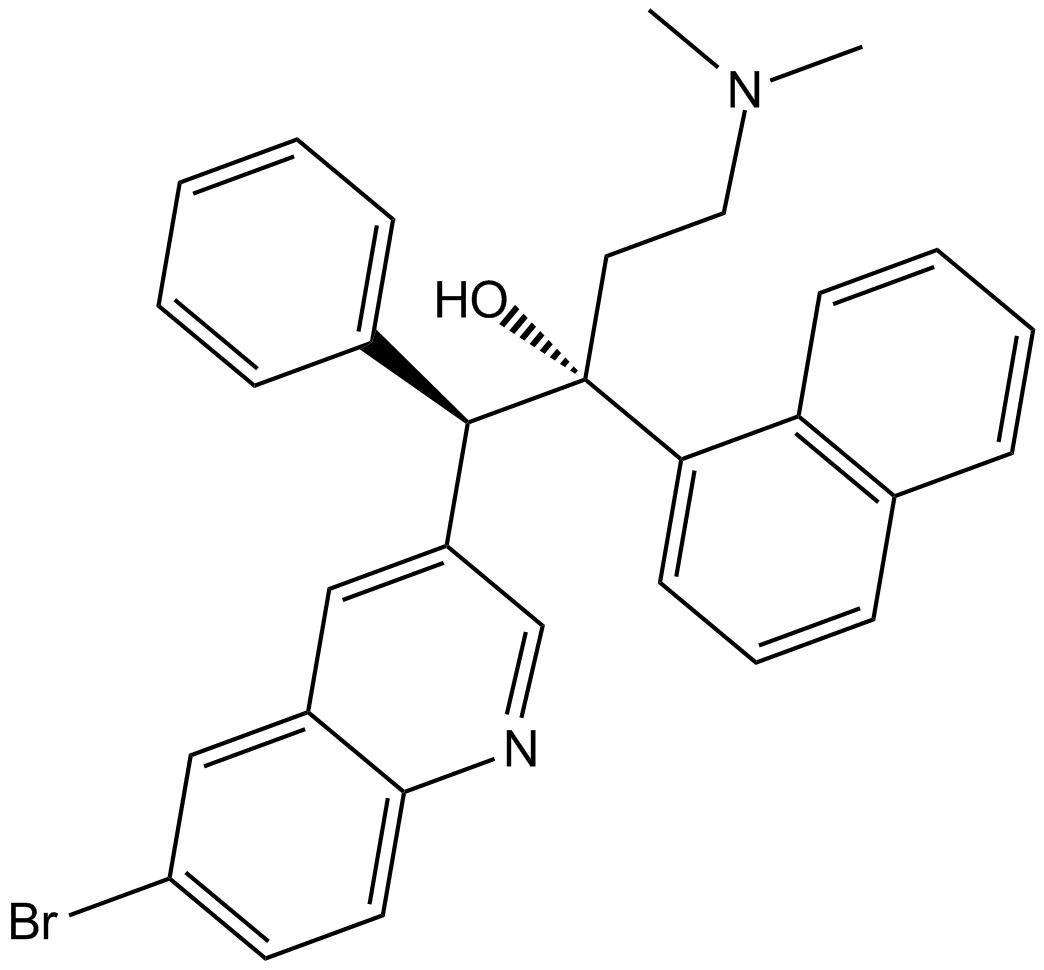
Bedaquiline

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Bedaquiline is a diarylquinoline drug and inhibits Mycobacterium tuberculosis F1FO-ATP synthase by simultaneously targeting the subunit c and subunit ε. Bedaquiline has uncoupling activity and is used for the multi-drug resistant tuberculosis[1].
Bedaquiline has anticancer activity against cancer stem cell-like cells. Bedaquiline treatment of MCF7 breast cancer cells can inhibit mitochondrial oxygen consumption and glycolysis, but can induce oxidative stress. Bedaquiline can reduce mitochondrial membrane potential and significantly increase ROS levels[2].
Bedaquiline follows the principle of three-stage elimination, and its terminal half-life is very long, approximately 173 hours in humans[3].
References:
[1]. Jang J C, Jung Y G, Choi J, et al. Bedaquiline susceptibility test for totally drug-resistant tuberculosisMycobacterium tuberculosis. Journal of Microbiology, 2017, 55(6): 483-487.
[2]. Fiorillo M, Lamb R, Tanowitz H, et al. Bedaquiline, an FDA-approved antibiotic, inhibits mitochondrial function and potently blocks the proliferative expansion of stem-like cancer cells (CSCs). Aging, 2016, 8(8): 1593-1607.
[3]. Lakshmanan M, Xavier A S. Bedaquiline–The first ATP synthase inhibitor against multi drug resistant tuberculosis. Journal of Young Pharmacists, 2013, 5(4): 112-115.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 525.5 |
| Cas No. | 843663-66-1 |
| Formula | C31H29BrN2O |
| Solubility | ≥22.05 mg/mL in DMSO with gentle warming; insoluble in EtOH; insoluble in H2O |
| Chemical Name | (1R,2S)-1-(6-bromoquinolin-3-yl)-4-(dimethylamino)-2-(naphthalen-1-yl)-1-phenylbutan-2-ol |
| SDF | Download SDF |
| Canonical SMILES | CN(C)CC[C@@]([C@H](C1=CC=CC=C1)C2=CC3=CC(Br)=CC=C3N=C2)(O)C4=CC=CC5=C4C=CC=C5 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Cell experiment:[1] | |
|
Cell lines |
MCF-7 human breast cancer cells |
|
Reaction Conditions |
48 h incubation |
|
Applications |
Bedaquiline treatment (10 μM) of MCF-7 breast cancer cells inhibited mitochondrial oxygen-consumption, as well as glycolysis, but induced oxidative stress. Importantly, bedaquiline significantly blocked the propagation and expansion of MCF-7-derived cancer stem cells, with an IC50 value of ~ 1 μM. |
| Animal experiment:[2] | |
|
Animal models |
Mice infected with Mycobacterium tuberculosis (M. tuberculosis) |
|
Dosage form |
25 mg/kg Administered orally, 5 days per week, for 14 weeks |
|
Applications |
The bedaquiline-containing regimen (rifampicin, isoniazid, pyrazinamide and bedaquiline) achieved total organ cfu count clearance at 8 weeks after treatment initiation, faster than the standard regimen consisting of rifampicin, isoniazid, pyrazinamide and ethambutol (14 weeks). Furthermore, the bedaquiline-containing regimen removed culture filtrate-dependent persistent bacilli at 8 weeks, leading to no disease relapse. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Fiorillo M, Lamb R, Tanowitz HB, et al. Bedaquiline, an FDA-approved antibiotic, inhibits mitochondrial function and potently blocks the proliferative expansion of stem-like cancer cells (CSCs). Aging (Albany NY), 2016, 8(8): 1593-1607. 2. Hu Y, Pertinez H, Liu Y, et al. Bedaquiline kills persistent Mycobacterium tuberculosis with no disease relapse: an in vivo model of a potential cure. Journal of Antimicrobial Chemotherapy, 2019, 74(6): 1627-1633. |
|
质量控制和MSDS
- 批次:
化学结构