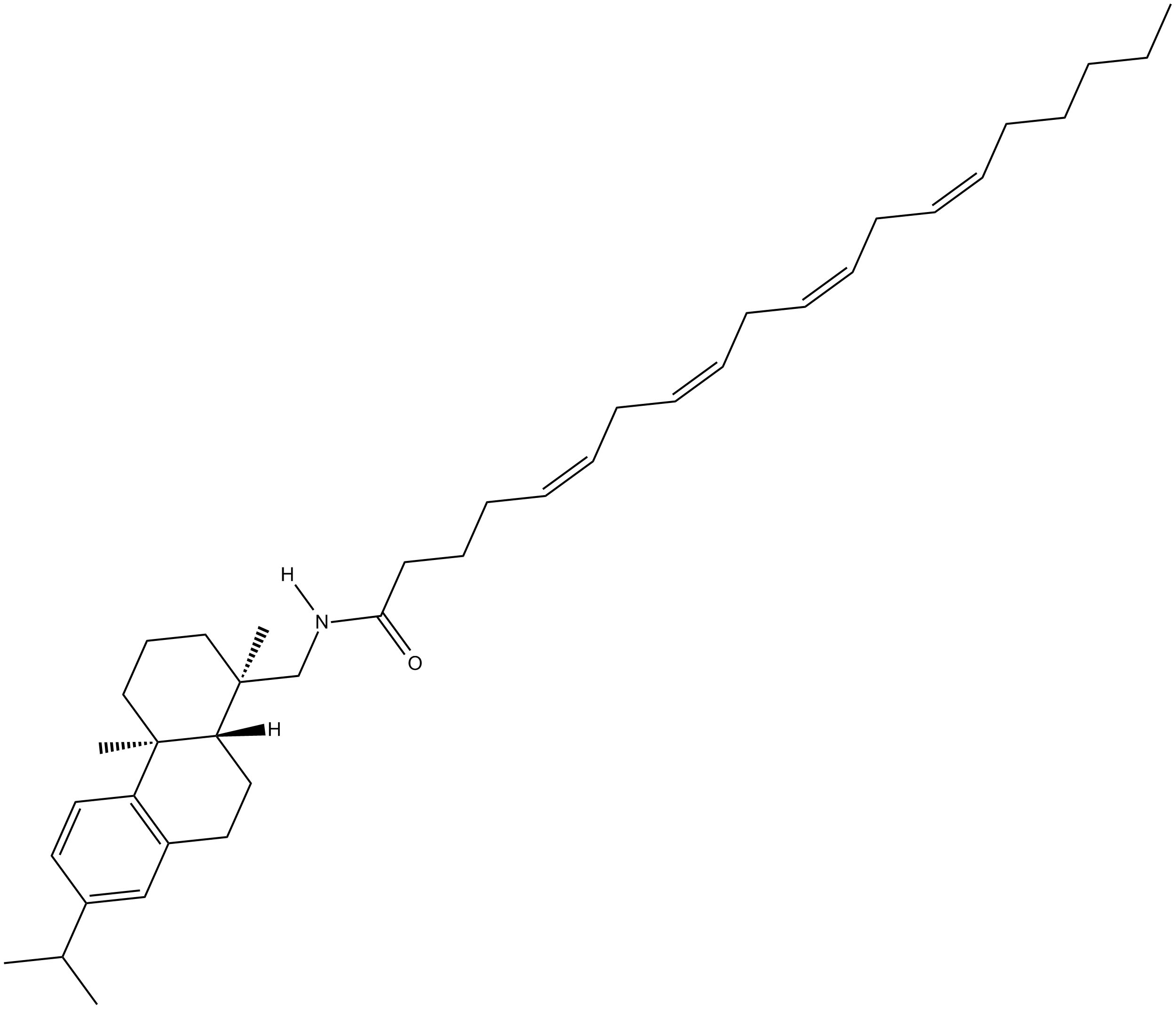
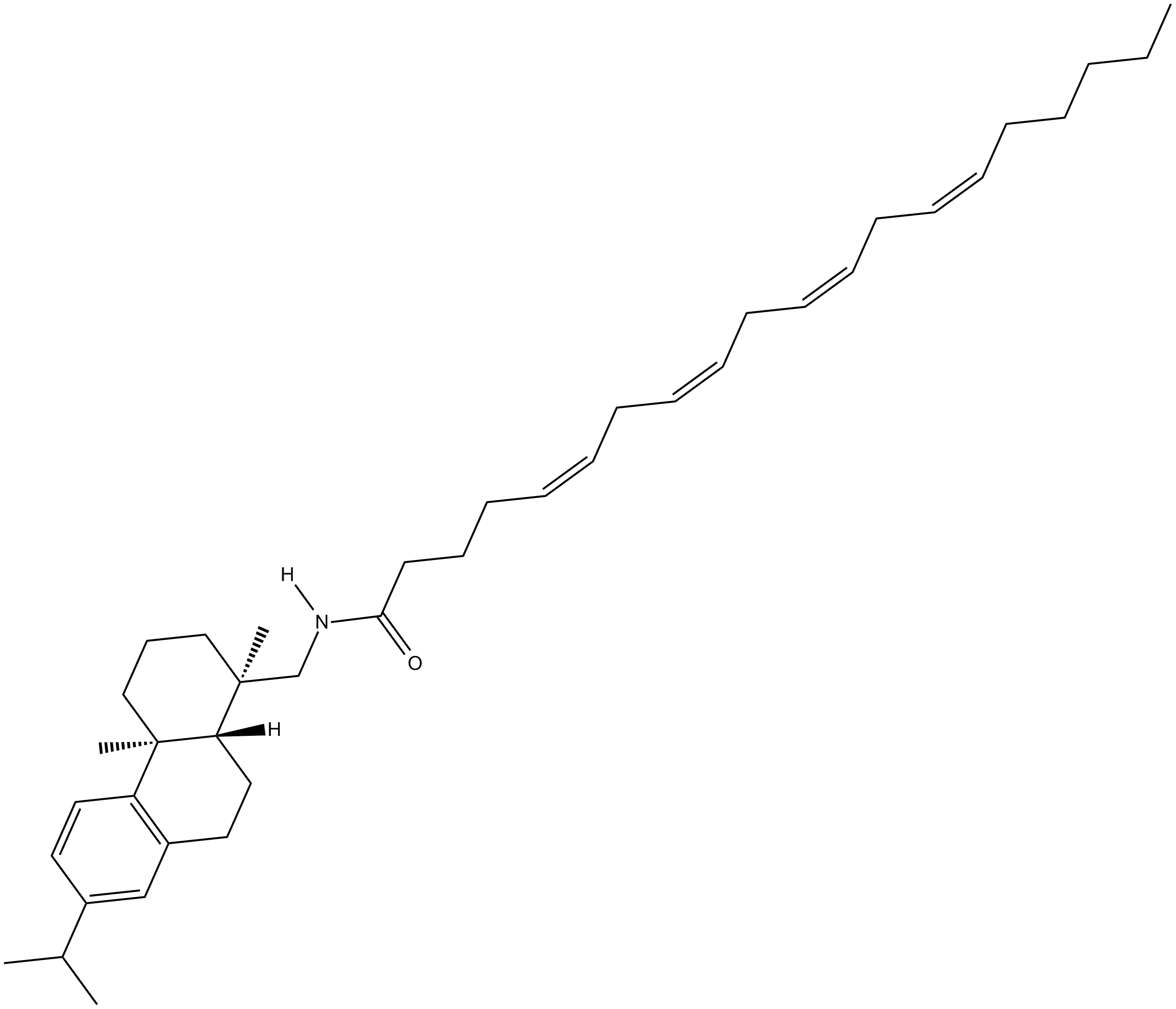
Arachidonic Acid Leelamide

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Arachidonic acid leelamide is a phospholipase A2 inhibitor.
Phospholipase A is a hydrolase responsible for the release of arachidonic acid from the sn2 position of phospholipids. The released arachidonic acid is then converted to mediators of inflammation by the enzymes prostaglandin synthetase and 5lipoxygenase, respectively. The inhibition of phospholipase A leads to a decrease in the release of arachidonic acid and, consequently, the inflammatory mediators.
In vitro: Arachidonic acid leelamide is the arachidonic amide analog of leelamine with no published pharmacological properties. For leelamine, it was found that electron micrographs of leelamine-treated cancer cells had accumulation of autophagosomes, membrane whorls, and lipofuscin-like structures. In addition, leelamine-mediated killing was a caspase-independent event triggered by cholesterol accumulation in the early process [1].
In vivo: In a previous study, authors identified the inductive effect of leelamine on CYP2B at doses of 5, 10, or 20 mg/kg in male ICR mice for 1 or 3 days. It was found that in liver, the activity of CYP2B significantly increased 3.6-fold after leelamine treatment. Activities of benzyloxyresorufin O-dealkylase and pentoxyresorufin O-dealkylase significantly increased 6.3- and 5.3-fold, respectively, with a single treatment of 20 mg/kg leelamine. Moreover, immunoblot analyses showed that significantly and dose-dependently increased CYP2B10 protein levels in liver. However, PCR results demonstrated that there were no significant changes in the CAR and CYP2B mRNA levels after leelamine treatment [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Kuzu OF, Gowda R, Sharma A, Robertson GP. Leelamine mediates cancer cell death through inhibition of intracellular cholesterol transport. Mol Cancer Ther. 2014 Jul;13(7):1690-703.
[2] Sim J, Nam W, Lee D, Lee S, O H, Joo J, Liu KH, Han JY, Ki SH, Jeong TC, Lee T, Lee S. Selective induction of hepatic cytochrome P450 2B activity by leelamine in vivo, as a potent novel inducer. Arch Pharm Res. 2015;38(5):725-33.
| Physical Appearance | A solution in ethanol. To change the solvent, simply evaporate the ethanol under a gentle stream of nitrogen and immediately add the solvent of choice. |
| Storage | Store at -20°C |
| M.Wt | 571.9 |
| Formula | C40H61NO |
| Solubility | ≤20mg/ml in DMSO;20mg/ml in dimethyl formamide |
| Chemical Name | 1R,2,3,4,4aS,9,10,10aR-octahydro-1,4a-dimethyl-7-(1-methylethyl)-1-phenanthrene-5Z,8Z,11Z,14Z-eicosatetraenamide |
| SDF | Download SDF |
| Canonical SMILES | CC(C)C(C=C1)=CC2=C1[C@]3(C)[C@](CC2)([H])[C@@](CN([H])C(CCC/C=C\C/C=C\C/C=C\C/C=C\CCCCC)=O)(C)CCC3 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构