Statil

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 21.0 nM
Statil, also known as ICI 128436, is an aldose reductase inhibitor, inhibits the conversion of glucose to sorbitol. Statil has the ability to activate the activity of lipoprotein lipase (LPL) both in vivo and in vitro. Also, statil alleviates the cachectic symptoms induced by B16 melanoma in mice. LPL, as a key regulatory enzyme, is responsible for the hydrolysis of triglyceride-rich lipoproteins.
In vitro: Statil strongly, dose-dependently, inhibited the enzyme activity of tumor marker Aldo-keto reductase 1B10. In addition, statil suppressed the cell growth and proliferation dose-dependently in both lung cancer cells NCI-H460 and breast cancer cells BT-20. Also, it was shown that statil induced apoptotic cell death [1].
In vivo: Statil was given to diabetic male rats at 25mg/kg orally by gavage once daily. After five days, statil inhibited rat, bovine, and human aldose reductase and reduced sorbitol levels in sciatic nerve, retina, lens, and renal cortex. Moreover, statil played an important role in rodent models of the lenticular and neural complications of diabetes. And the development of cataracts was completely prevented in diabetic rats at doses as low as 25 mg/kg daily [2].
References:
[1]. Cao, Z., Zhou, B., Chen, X., Huang, D., Zhang, X., & Wang, Z. et al. Statil suppresses cancer cell growth and proliferation by the inhibition of tumor marker AKR1B10. Anti-Cancer Drugs, 2014; 25(8): 930-937.
[2]. Stribling, D., Mirrlees, D., Harrison, H., & Earl, D. Properties of ICI 128,436, a novel aldose reductase inhibitor, and its effects on diabetic complications in the rat. Metabolism, 1985; 34(4): 336-344.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 391.2 |
| Cas No. | 72702-95-5 |
| Formula | C17H12BrFN2O3 |
| Synonyms | ICI 128436,MK-538,Ponalrestat |
| Solubility | ≤2mg/ml in ethanol;2mg/ml in DMSO;2mg/ml in dimethyl formamide |
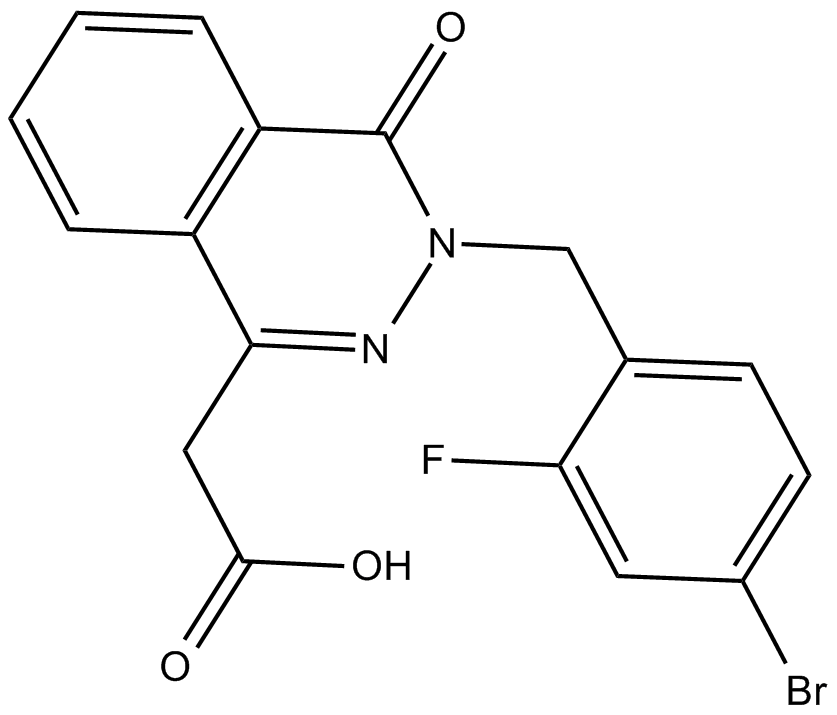
| Chemical Name | 3-[(4-bromo-2-fluorophenyl)methyl]-3,4-dihydro-4-oxo-1-phthalazineacetic acid |
| SDF | Download SDF |
| Canonical SMILES | BrC1=CC=C(CN2C(C(C=CC=C3)=C3C(CC(O)=O)=N2)=O)C(F)=C1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构