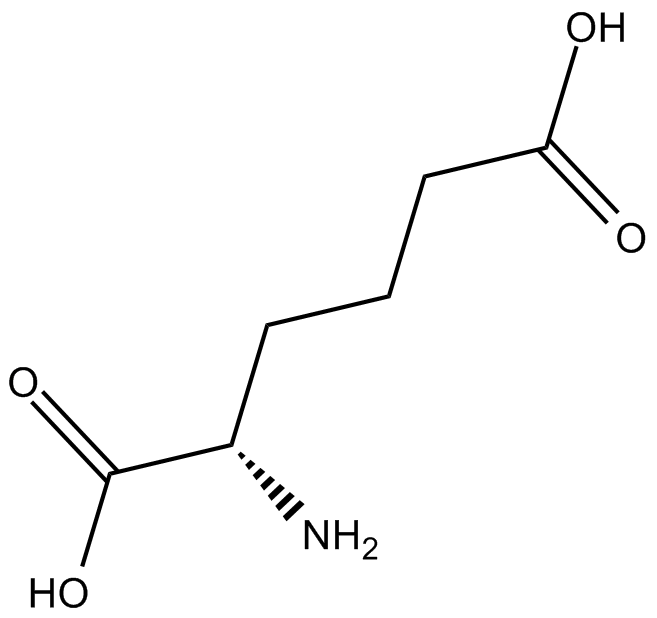
L-α-Aminoadipic Acid

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Ki value of 209 μM
L-α-Aminoadipic Acid is a glutamine synthetase inhibitor.
Glutamine synthetase is an enzyme that plays an essential role in the metabolism of nitrogen by catalyzing the condensation of glutamate and ammonia to form glutamine.
In vitro: Previous study found that DL- and L-alpha-aminoadipic acid (alpha-AA) were specific gliotoxins in vitro. HPLC analysis of cultures incubated with D- or L-alpha-AA and DL-[14C]-alpha-AA autoradiograms conducted in the presence of D- or L-alpha-AA suggested a stereospecificity of astroglial L-alpha-AA uptake. Both the uptake of alpha-AA by astrocytes and alpha-AA-induced gliotoxicity were sodium dependent [1]. Another study found that the L-isomer of alpha aminoadipate was able to competitively inhibit the transport protein, whereas the D-isomer of alpha aminoadipate was ineffective. Moreover, it was found that L-alpha aminoadipate was a competitive inhibitor of both glutamine synthetase, and gamma-glutamylcysteine synthetase. In constrast, the D-isomer of alpha aminoadipate was a far weaker inhibitor of either enzyme [2].
In vivo: Animal study showed that L-a-aminoadipic acid could lower the levels of endogenous extracellular kynurenic acid in the hippocampus in a dose-dependent fashion), though the effect of L-a-aminoadipic acid seemed to be less pronounced than its reduction of de novo produced kynurenic acid [3].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Huck, S. ,Grass, F., and Hrtnagl, H. The glutamate analogue α-aminoadipic acid is taken up by astrocytes before exerting its gliotoxic effect in vitro. Journal of Neuroscience 4(10), 2650-2657 (1984).
[2] McBean GJ. Inhibition of the glutamate transporter and glial enzymes in rat striatum by the gliotoxin, alpha aminoadipate. Br J Pharmacol. 1994 Oct;113(2):536-40.
[3] Wu HQ, Ungerstedt U, Schwarcz R. L-alpha-aminoadipic acid as a regulator of kynurenic acid production in the hippocampus: a microdialysis study in freely moving rats. Eur J Pharmacol. 1995 Jul 25;281(1):55-61.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 161.2 |
| Cas No. | 1118-90-7 |
| Formula | C6H11NO4 |
| Synonyms | (S)-2-Aminohexanedioic Acid |
| Solubility | insoluble in H2O; insoluble in EtOH; insoluble in DMSO |
| Chemical Name | 2S-amino-hexanedioic acid |
| SDF | Download SDF |
| Canonical SMILES | OC(CCC[C@H](N)C(O)=O)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构