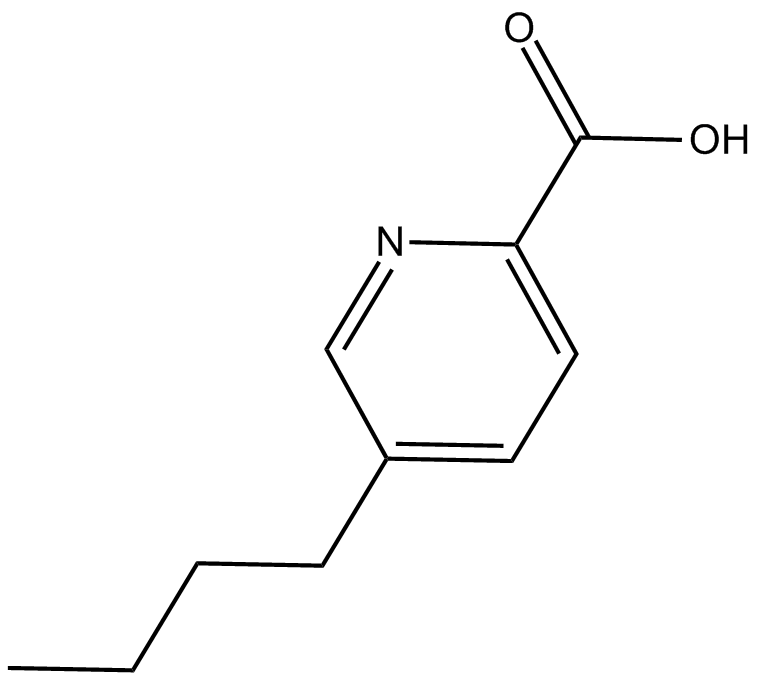
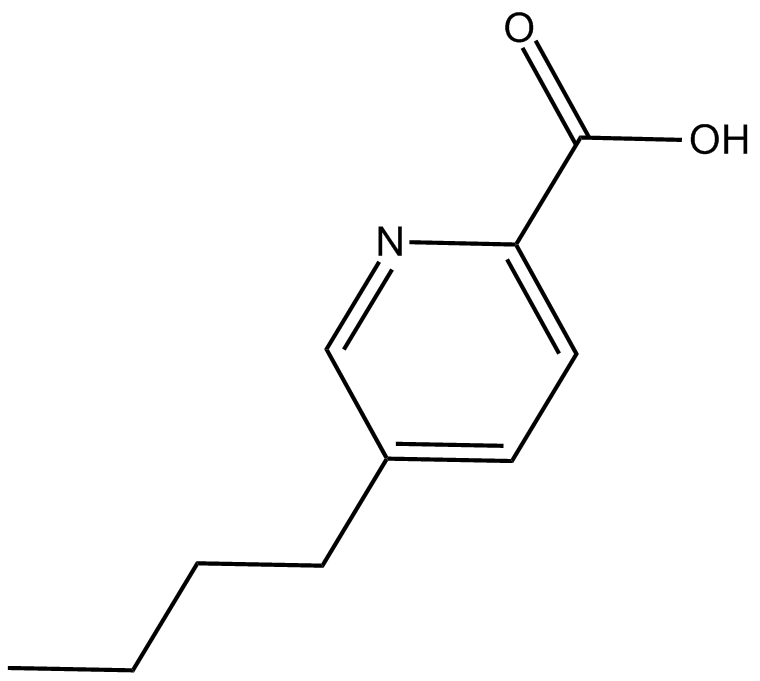
Fusaric Acid

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Fusaric acid is a mycotoxin produced by several species of Fusarium [1]. Mycotoxins are biologically active secondary fungal metabolites found as contaminants of food- and feedstuff. Mycotoxin is capable of causing disease and death in both humans and animals.
Fusaric acid is a potent inhibitor of dopamine β-hydroxylase. Fusaric acid uncompetitively inhibited the activity of dopamine β-hydroxylase with an IC50 of 30 μM in an. Fusaric acid lowered endogenous levels of norepinephrine and epinephrine in brain, spleen, heart, and adrenal glands. Fusaric acid inhibited dopamine β-hydroxylase activity in adrenal medulla in vivo [2]. Fusaric acid altered brain and pineal neurotransmitters. In the brain and pineal gland of rats, intraperitoneally (100 mg/kg) administration of fusaric acid increased the level of 5HT, 5-hydroxyindoleacetic acid (5HIAA), tyrosine, and dopamine [3].
Exposure to acute doses of fusaric acid caused vomiting and neurochemical changes in swine. Fusaric acid might act synergistically with trichothecene mycotoxins to cause vomiting and feed refusal in pigs consuming trichothecene-contaminated feedstuffs [4].
References:
[1] Hidaka H, NAGATSU T, TAKEYA K, et al. Fusaric acid, a hypotensive agent produced by fungi[J]. The Journal of antibiotics, 1969, 22(5): 228-230.
[2] Toshiharu N, Hiroyoshi H, Hiroshi K, et al. Inhibition of dopamine β-hydroxylase by fusaric acid (5-butylpicolinic acid) in vitro and in vivo[J]. Biochemical pharmacology, 1970, 19(1): 35-44.
[3] Porter J K, Bacon C W, Wray E M, et al. Fusaric acid in Fusarium moniliforme cultures, corn, and feeds toxic to livestock and the neurochemical effects in the brain and pineal gland of rats[J]. Natural Toxins, 1995, 3(2): 91-100.
[4] Smith T K, MacDonald E J. Effect of fusaric acid on brain regional neurochemistry and vomiting behavior in swine[J]. Journal of animal science, 1991, 69(5): 2044-2049.
| Storage | Store at -20°C |
| M.Wt | 179.2 |
| Cas No. | 536-69-6 |
| Formula | C10H13NO2 |
| Synonyms | 5-Butylpicolinic Acid,5-Butylpyridine-2-Carboxylic Acid,NSC 135043 |
| Solubility | ≥26.45 mg/mL in H2O with ultrasonic; ≥55.8 mg/mL in EtOH; ≥8.75 mg/mL in DMSO |
| Chemical Name | 5-butyl-2-pyridinecarboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | O=C(O)C1=CC=C(CCCC)C=N1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |