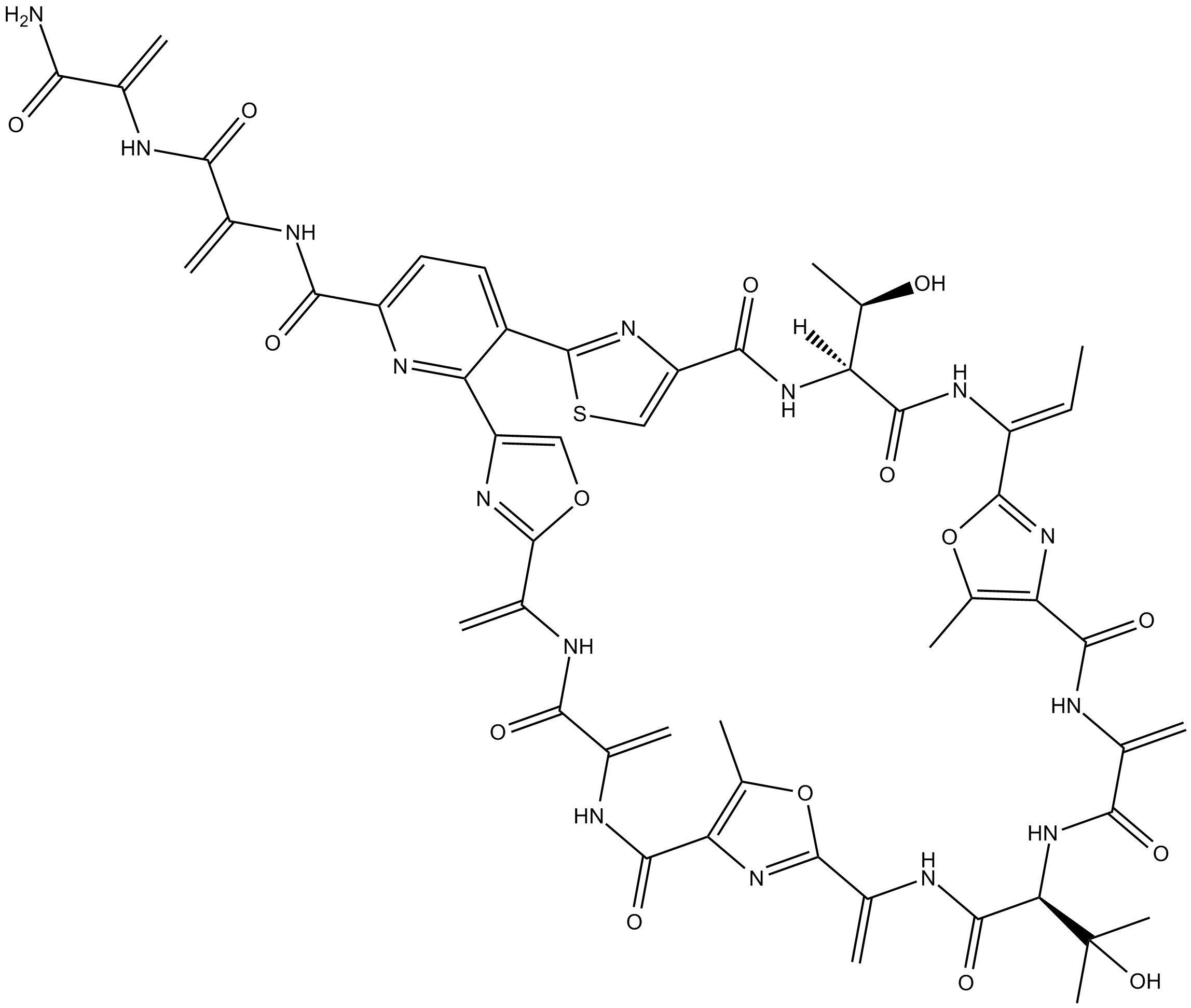
Berninamycin A

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Berninamycin A is a macrocyclic thiopeptide antibiotic first isolated from S. bernensis [1]. Thiopeptides are sulfur containing highly modified macrocyclic antibiotics with a central pyridine/tetrapyridine/dehydropiperidine ring with up to three thiazole substituents on positions 2, 3 and 6. Thiazole antibiotics thiostrepton behaves as proteasome inhibitor in mammalian tumor cells. Berninamycin showed no proteasome inhibitory activity [2]. It has been reported that the action mode of berninamycin on bacterial protein synthesis was related to that of a dissimilar compound thiostrepton. The antibiotics could bind to the complex of 23S RNA with protein L11 and both affect multiple functions of the ribosomal A site [3]. Berninamycin A was involved in inducing the transcriptional activator TipA in Streptomyces [4].
References:
[1] Lau R C M, Rinehart K L. Berninamycins B, C, and D, minor metabolites from Streptomyces bernensis[J]. The Journal of antibiotics, 1994, 47(12): 1466-1472.
[2] Pandit B, Bhat U G, Gartel A L. Proteasome inhibitory activity of thiazole antibiotics[J]. Cancer biology & therapy, 2011, 11(1): 43-47.
[3] J. Thompson, E. Cundliffe and M. J. R. Stark. The mode of action of berninamycin and mechanism of resistance in the producing organism, Streptomyces bernensis. J.Gen.Microbiol. 128(4), 875-884 (1982).
[4]. M. L. Chiu, M. Folcher, T. Katoh, et al. Broad spectrum thiopeptide recognition specificity of the Streptomyces lividans TipAL protein and its role in regulating gene expression. The Journal of Biological Chemisty 274(29), 20578-20586 (1999).
| Physical Appearance | A white solid |
| Storage | Store at -20°C |
| M.Wt | 1146.1 |
| Cas No. | 58798-97-3 |
| Formula | C51H51N15O15S |
| Solubility | Soluble in DMSO |
| Chemical Name | L-threonyl-2-[(1Z)-1-amino-1-propen-1-yl]- 5-methyl-4-oxazolecarbonyl-2,3-didehydroalanyl-3-methyl-L-threonyl-2-(1-aminoethenyl)- 5-methyl-4-oxazolecarbonyl-2,3-didehydroalanyl-6-[2-(1-aminoethenyl)- 4-oxazolyl]-5-(4-carboxy-2-thiazolyl)-2-pyridinecarbony |
| SDF | Download SDF |
| Canonical SMILES | O=C(NC(C(NC(C(N)=O)=C)=O)=C)C1=CC=C(C2=NC(C(N[C@]([C@@H](C)O)([H])C(N/C(C3=NC4=C(C)O3)=C\C)=O)=O)=CS2)C(C5=COC(C(NC(C(NC(C6=C(C)OC(C(NC([C@@H](NC(C(NC4=O)=C)=O)C(C)(O)C)=O)=C)=N6)=O)=C)=O)=C)=N5)=N1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构