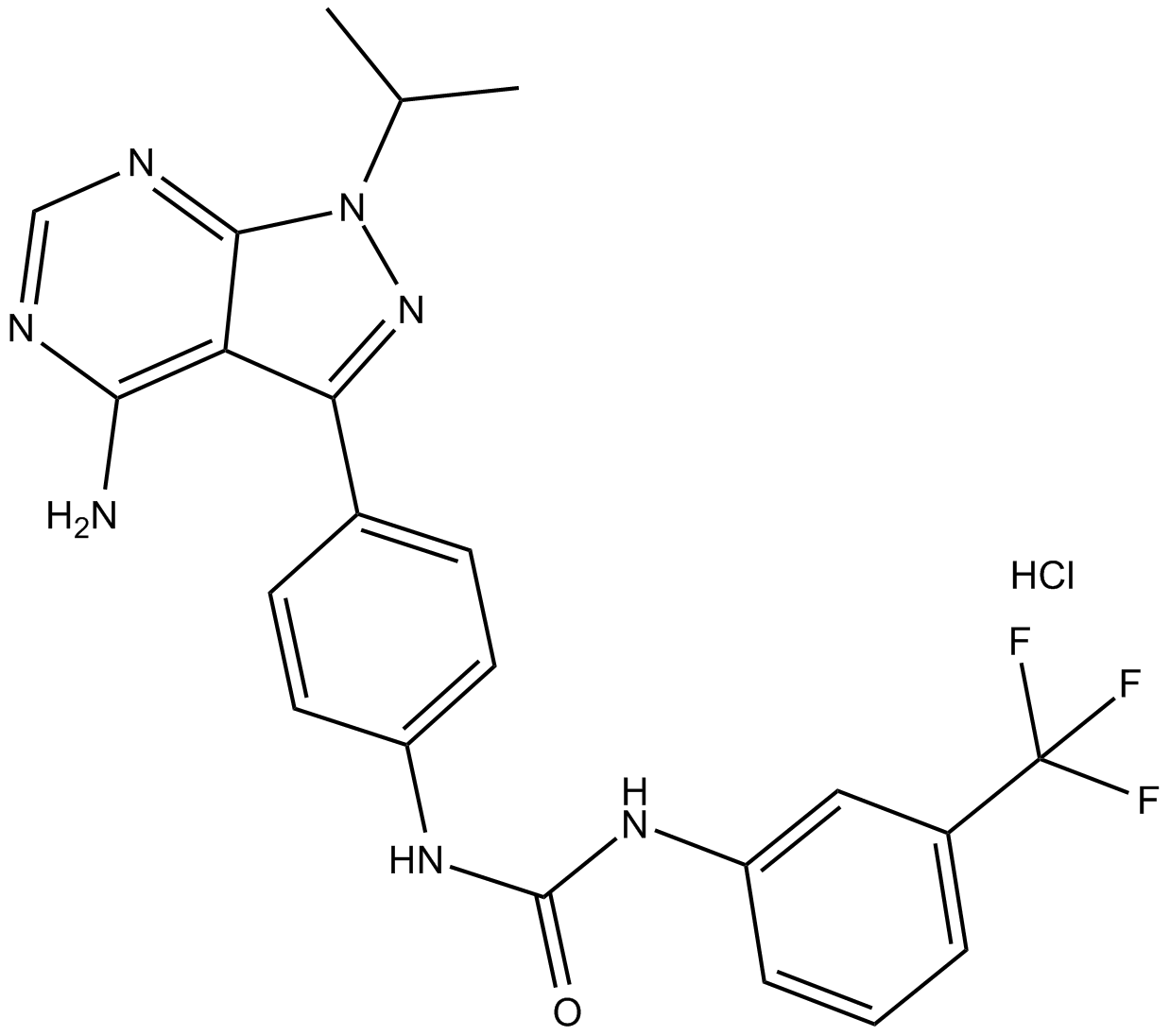
AD57 (hydrochloride)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 2 nM: blocks the receptor tyrosine kinase RET in Drosophila.
AD57, as a polypharmacological cancer therapeutic, is designed to regulate multiple targets related to cancer. AD57 effectively suppresses tyrosine kinase RET, weakens the activity of numerous other kinases, and interferes with kinases downstream of RET, including Raf, Src, and S6K, providing further efficacy in preventing signaling leading to invasion, proliferation, and metabolism related to cancer. Tyrosine kinase RET is probably sufficient to initiate a series of transformation events including medullary thyroid carcinoma, multiple endocrine neoplasias type 2A (MEN2A) and 2B (MEN2B).
In vitro: AD57 was demonstrated to be able to inhibit tyrosine kinase RET. But the efficacy of AD57 did not correlate solely with the blockade of RET, suggesting that the targeting of additional kinases is necessary for its biological efficacy. AD57 potently dampened the pathway-related human kinases B-Raf, mTOR, S6K, and Src [1].
In vivo: Male nu nu mice, injected subcutaneously with MEN2A cell lines, were administered AD57 (20 mg/kg) by oral gavage (per os) once daily, five times a week. AD57 inhibited the activity of relevant target kinases at 1 μM compared with the vehicle-treated nude mice transplanted with MEN2A cells [1].
Reference:
[1]. Dar, A., Das, T., Shokat, K., & Cagan, R. Chemical genetic discovery of targets and anti-targets for cancer polypharmacology. Nature. 2012; 486(7401): 80-84.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 491.9 |
| Formula | C22H20F3N7O·HCl |
| Solubility | ≤5mg/ml in ethanol;10mg/ml in DMSO;14mg/ml in dimethyl formamide |
| Chemical Name | N-[4-[4-amino-1-(1-methylethyl)-1H-pyrazolo[3,4-d]pyrimidin-3-yl]phenyl]-N'-[3-(trifluoromethyl)phenyl]-urea, monohydrochloride |
| SDF | Download SDF |
| Canonical SMILES | CC(C)N1C2=NC=NC(N)=C2C(C3=CC=C(NC(NC4=CC(C(F)(F)F)=CC=C4)=O)C=C3)=N1.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构