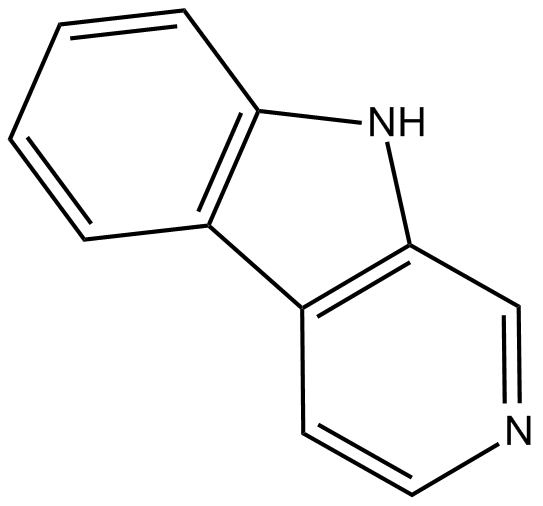
Norharmane

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Norharmane acts as a co-mutagen. Norharmane is a natural β-carboline first isolated from plants of the Zygophyllaceae family. It is a heterocyclic amine (HCA) that may also be found in fried meats, tobacco smoke, and coffee. Harman and norharmane are aminocarbolines known as “co-mutagens” because they do not show mutagenicity to Salmonella serovar typhimurium [1][2][3].
Norharmane acts as a co-mutagen. Norharmane is widely distributed in our environment. It was not mutagenic to Salmonella typhimurium TA98 and TA100 either with or without S9 mix, but became mutagenic to S.typhimurium TA98 with S9 mix when non-mutagenic aromatic amines like aniline or o- or m-toluidine are added. Also, Norharmane and aromatic amines induced the formation of DNA adduct in S.typhimurium TA98 [3].
Harman and norharmane with concentrations of 0.275 uM and 0.9 uM respectively might inhibited MAO-A and MAO-B. In adult male rats, a novel cocktail of cigarette smoke constituents, containing five minor alkaloids (nornicotine, cotinine, myosmine, anatabine, and anabasine), two β-carbolines (harman and norharmane), and acetaldehyde, did not significantly enhance nicotine self-administration [2].
References:
[1]. Jamali MA, Zhang Y, Teng H, et al. Inhibitory Effect of Rosa rugosa Tea Extract on the Formation of Heterocyclic Amines in Meat Patties at Different Temperatures. Molecules. 2016 Jan 30;21(2):173.
[2]. Smith TT, Schaff MB, Rupprecht LE, et al. Effects of MAO inhibition and a combination of minor alkaloids, β-carbolines, and acetaldehyde on nicotine self-administration in adult male rats. Drug Alcohol Depend. 2015 Oct 1;155:243-52.
[3]. Mori M1, Totsuka Y, Fukutome K, et al. Formation of DNA adducts by the co-mutagen norharman with aromatic amines. Carcinogenesis. 1996 Jul;17(7):1499-503.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 168.2 |
| Cas No. | 244-63-3 |
| Formula | C11H8N2 |
| Synonyms | 2-Azacarbazole,β-Carboline,2,9-Diazafluorene,NSC 84417 |
| Solubility | insoluble in H2O; ≥32.8 mg/mL in DMSO; ≥8.82 mg/mL in EtOH |
| Chemical Name | 9H-Pyrido[3,4-b]indole |
| SDF | Download SDF |
| Canonical SMILES | C1(C=CC=C2)=C2C(C=CN=C3)=C3N1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构