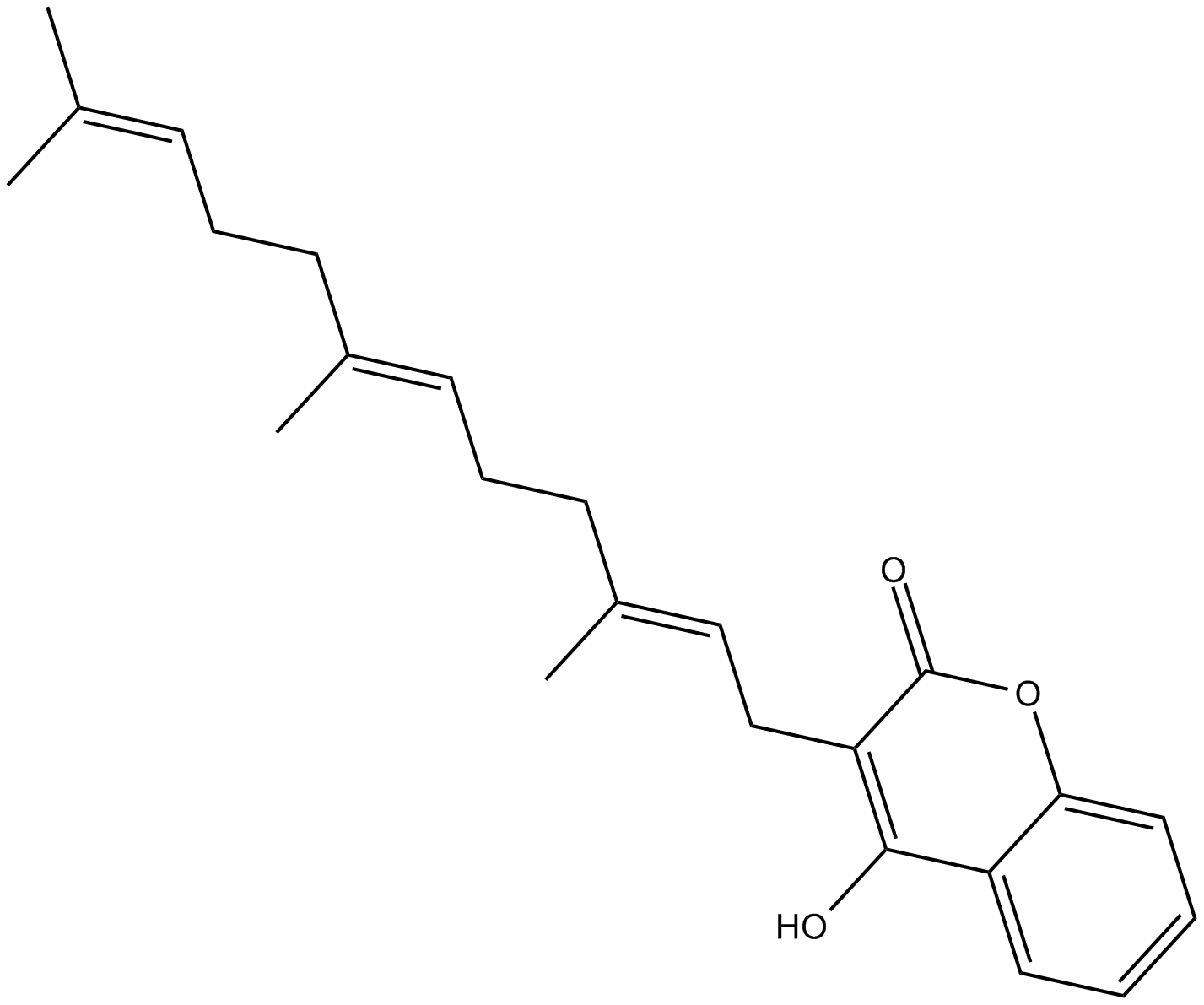
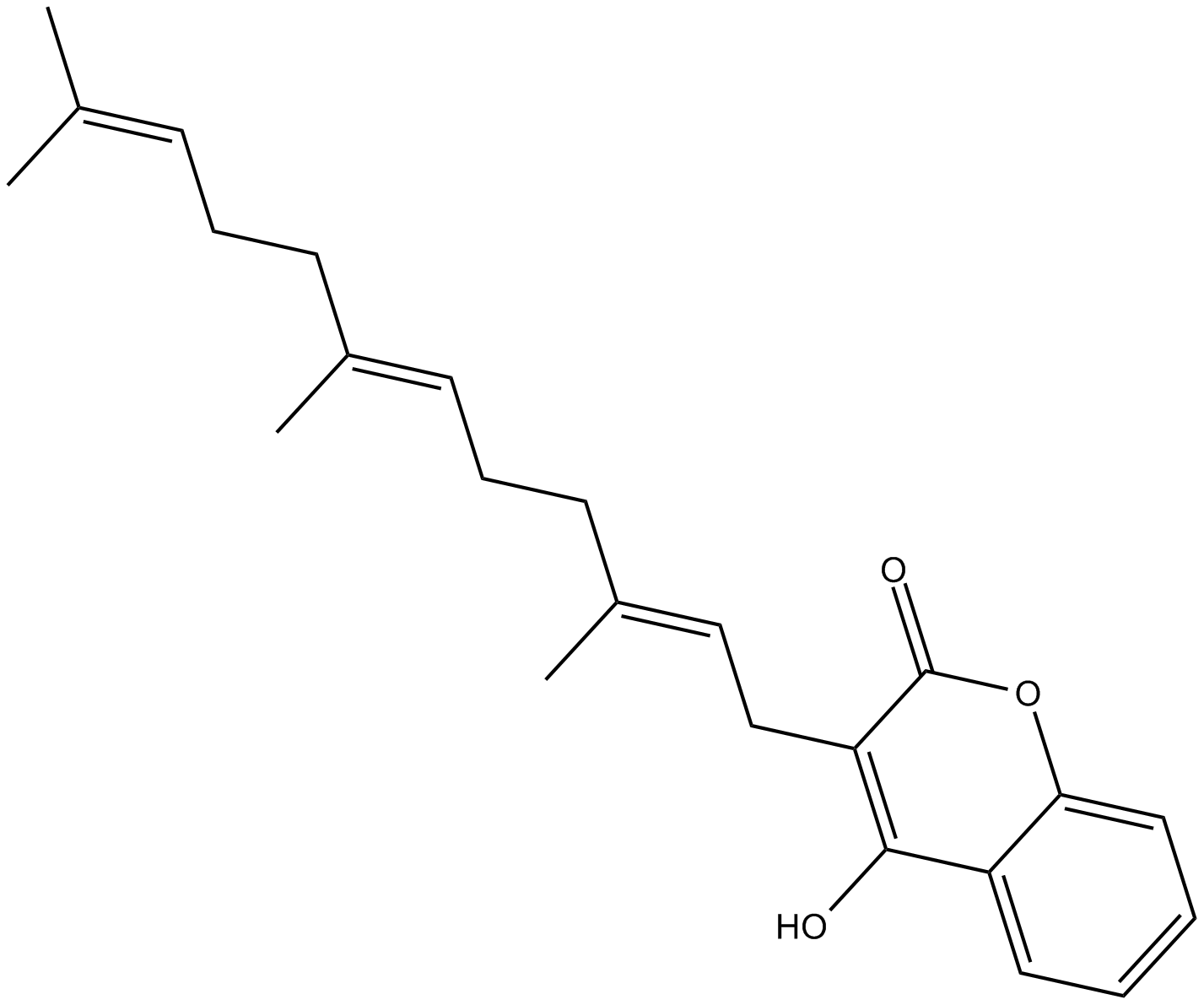
Ferulenol

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Ferulenol is a prenylated 4-hydroxycoumarin derivative from Ferula communis var. genuine with haemorrhagic action [1]. It has been demonstrated that ferulenol exihibits potent antimycobacterial activity [2].
In vitro: Ferulenol stimulated tubulin polymerization in the absence of GTP, with a less extensive polymerization profile at 100 pM concentration. Ferulenol decreased radiolabeled colchicine bound by tubulin in a dose-dependent manner. Ferulenol altered the normal nuclear morphology of MCF-7 cells. Treatment with ferulenol (100 nM and 1 μM) for 24h induced a dose-dependent reduction of cell viability [3]
In vivo: In albino mice, the acute LD50s of ferulenol by single po or ip were 2,100 and 319 mg/kg bw, respectively. Three days after ferulenol administration, dosed animals showed hypoprothrombinemia with internal and external hemorrhages. Male mice were more sensitive to intoxication than females [4].
References:
[1] Lamnaouer D, Bodo B, Martin M T, et al. Ferulenol and ω-hydroxyferulenol, toxic coumarins from Ferula communis var. genuina[J]. Phytochemistry, 1987, 26(6): 1613-1615.
[2] E. Mamoci, I. Cavoski, V. Simone, et al. Chemical composition and in vitro activity of plant extracts from Ferula communis and Dittrichia viscosa against postharvest fungi. Molecules 16(3), 2609-2625 (2011).
[3] Bocca C, Gabriel L, Bozzo F, et al. Microtubule-interacting activity and cytotoxicity of the prenylated coumarin ferulenol[J]. Planta medica, 2002, 68(12): 1135-1137.
[4] Fraigui O, Lamnaouer D, Faouzi M Y. Acute toxicity of ferulenol, a 4-hydroxycoumarin isolated from Ferula communis L[J]. Veterinary and human toxicology, 2002, 44(1): 5-7.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 366.5 |
| Cas No. | 6805-34-1 |
| Formula | C24H30O3 |
| Solubility | Soluble in DMSO |
| Chemical Name | 4-hydroxy-3-[(2E,6E)-3,7,11-trimethyl-2,6,10-dodecatrien-1-yl]-2H-1-benzopyran-2-one |
| SDF | Download SDF |
| Canonical SMILES | O=C1C(C/C=C(C)/CC/C=C(C)/CC/C=C(C)/C)=C(O)C2=CC=CC=C2O1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构