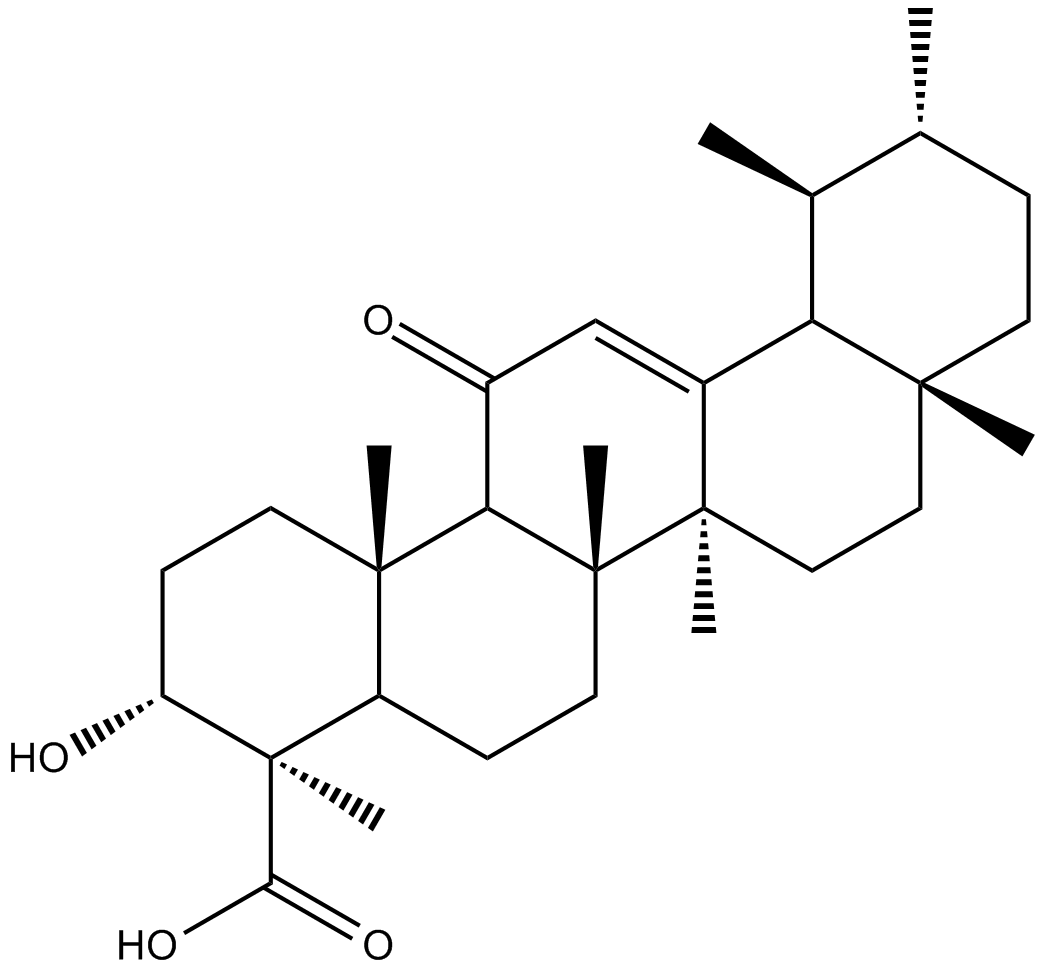
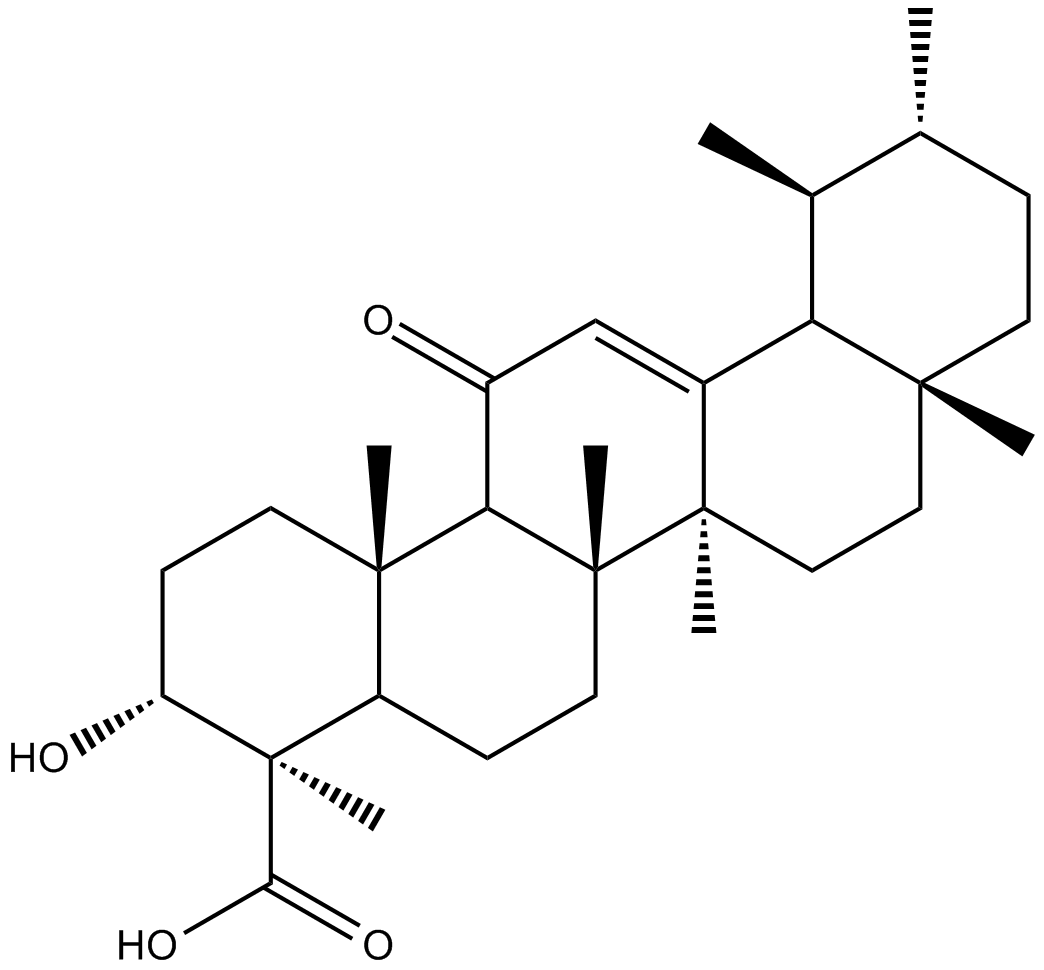
11-keto-β-Boswellic Acid

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 35.8 μM: inhibits MCF-7 (human breast adenocarcinoma) [1].
37.9 μM: blocks A2780 (cis-platin resistant ovarian cancer cells) [1].
11-keto-β-Boswellic Acid, known as KBA, is a naturally occurring pentacyclic triterpene isolated from the gum resin of the tree Boswellia serrata. KBA is non-redox, specific leukotriene synthesis inhibitors through the inhibition of 5-lipoxygenase (5-LOX) which has anti-arthritic and anti-inflammatory activities. 5-LOX catalyzes essential fatty acids substrates into leukotrienes and a variety of other biologically active products. KBA is a novel activator of nuclear factor erythroid-2-related factor 2 (Nrf2), which protects against cerebral ischemic injury.
In vitro: KBA, concentration dependently, decreased the formation of leukotriene B4 and the synthesis of all 5-LOX products from endogenous arachidonic acid in rat peritoneal neutrophils. In contrast, KBA exerted no remarkable effects on the 12-lipoxygenase and cyclooxygenase activities. [2].
In vivo: Adult male Sprague–Dawley rats were injected KAB intraperitoneally at a dose of 25 mg/kg for 48 hours. KAB remarkably decreased infarct volumes as well as apoptotic cells at 1 h, and then increased neurologic scores when applied 48 h. Moreover, posttreatment with KBA induced the decrease of malondialdehyde levels and the increase of protein Nrf2 and heme oxygenase-1 expression in brain tissues, indicating that the Nrf2/HO-1 pathway was involved in the neuroprotection of KBA against oxidative stress-induced ischemic injury [3].
References:
[1]. Csuk, R., Barthel-Niesen, A., Barthel, A., Schffer, R., & Al-Harrasi, A. 11-Keto-boswellic acid derived amides and monodesmosidic saponins induce apoptosis in breast and cervical cancers cells. European Journal of Medicinal Chemistry. 2015; 100: 98-105.
[2]. Safayhi, H., Mack, T. H. O. M. A. S., Sabieraj, J. O. A. C. H. I. M., Anazodo, M. I., Subramanian, L. R., & Ammon, H. P. Boswellic acids: novel, specific, nonredox inhibitors of 5-lipoxygenase. Journal of Pharmacology and Experimental Therapeutics. 1992; 261(3):1143-1146.
[3]. Ding, Y., Chen, M., Wang, M., Li, Y., & Wen, A. Posttreatment with 11-Keto-β-Boswellic Acid Ameliorates Cerebral Ischemia–Reperfusion Injury: Nrf2/HO-1 Pathway as a Potential Mechanism. Molecular Neurobiology. 2014; 52(3): 1430-1439.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 470.4 |
| Cas No. | 17019-92-0 |
| Formula | C30H46O4 |
| Synonyms | 11-oxo-β-Boswellic acid,KBA |
| Solubility | ≤5mg/ml in ethanol;25mg/ml in DMSO;25mg/ml in dimethyl formamide |
| Chemical Name | (3α,4β)-3-hydroxy-11-oxo-urs-12-en-23-oic acid |
| SDF | Download SDF |
| Canonical SMILES | O=C1C=C2[C@](CC[C@]3(C)C2[C@@H](C)[C@H](C)CC3)(C)[C@]4(C)CCC5[C@@](C)(C(O)=O)[C@H](O)CC[C@]5(C)C41 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |