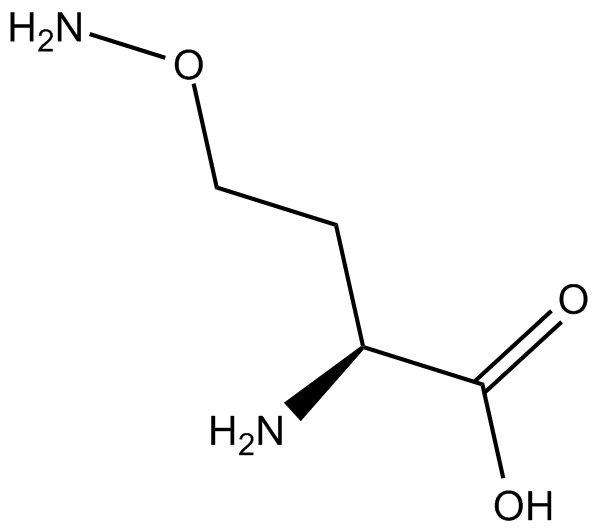
L-Canaline

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
L-Canaline is a well-known irreversible inhibitor of ornithine aminotransferase (OAT). The natural L-enantiomer reacts by oxime formation with pyridoxal 5′-phosphate in the active site of the enzyme [1]. L-Canaline is naturally found in plants such as legumes, and has been involved in the metabolism of L-canavanine, an aminooxy analog of arginine [2].
Ornithine aminotransferase (OAT) is a mitochondrial enzyme involved in catalyzing the interaction of L-ornithine and α-ketoglutarate to produce glutamic-y-semialdehyde and glutamate [3].
In vitro: Canaline strongly inhibited the activity of pyridoxal-dependent enzymes, including amino acid decarboxylases, 5-hydroxytryptophan decarboxylase, aminotransferases, tyrosine aminotransferase, ornithine transcarbamylase and plasma diamino-oxidase. The canaline inhibition was due to complex formation between canaline and the pyridoxal coenzyme. l-canaline is one of the most potent inhibitors of pyridoxal enzymes. The IC50 value of l-canaline against Ornithine aminotransferase was 3 ×10-6M [4].
In vivo: Intraperitoneal administration of 500 mg of DL-canaline/kg body wt. only produced a transient inhibition of OAT in brain and liver by 65-70%, suggesting that DL-canaline was not a useful tool in studies of biological consequences of OAT inhibition. [1].
References:
[1] Bolkenius F N, Kndgen B, Seiler N. DL-canaline and 5-fluoromethylornithine. Comparison of two inactivators of ornithine aminotransferase[J]. Biochemical Journal, 1990, 268(2): 409-414.
[2] Rosenthal G A, Rhodes D. L-Canavanine transport and utilization in developing jack bean, Canavalia ensiformis (L.) DC.[Leguminosae][J]. Plant physiology, 1984, 76(2): 541-544.
[3] Peraino C, Bunville L G, Tahmisian T N. Chemical, physical, and morphological properties of ornithine aminotransferase from rat liver[J]. Journal of Biological Chemistry, 1969, 244(9): 2241-2249.
[4] Rahiala E L, Kekomki M, Jnne J, et al. Inhibition of pyridoxal enzymes by L-canaline[J]. Biochimica et Biophysica Acta (BBA)-Enzymology, 1971, 227(2): 337-343.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 134.1 |
| Cas No. | 496-93-5 |
| Formula | C4H10N2O3 |
| Solubility | ≤1mg/ml in DMSO |
| Chemical Name | O-amino-L-homoserine |
| SDF | Download SDF |
| Canonical SMILES | N[C@@H](CCON)C(O)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
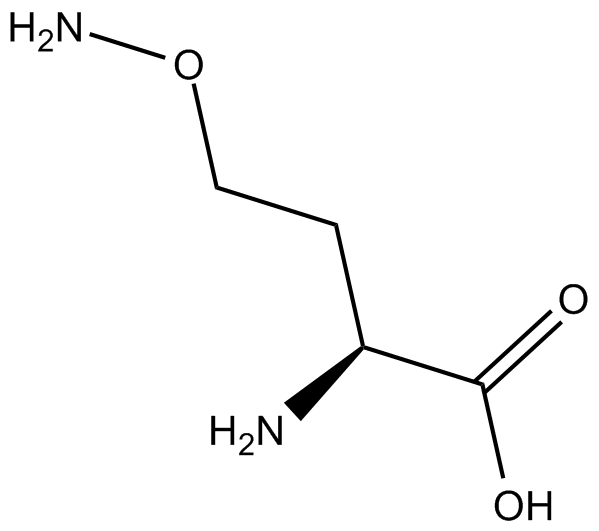
化学结构
相关生物数据