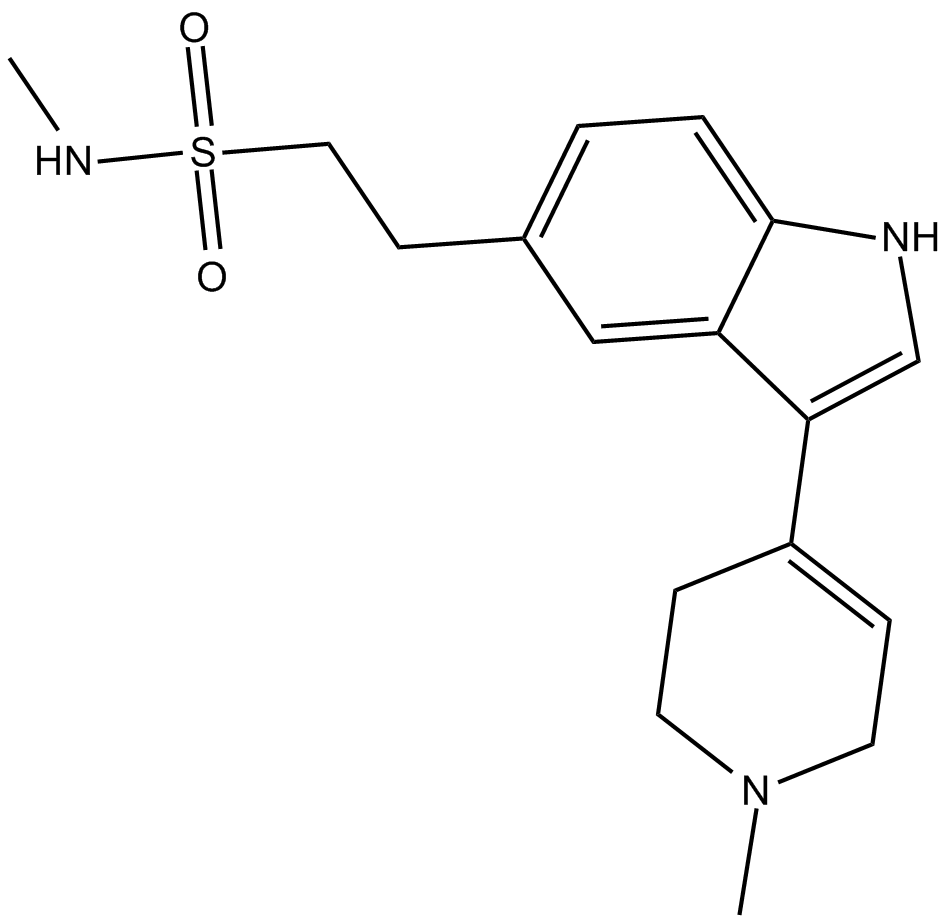
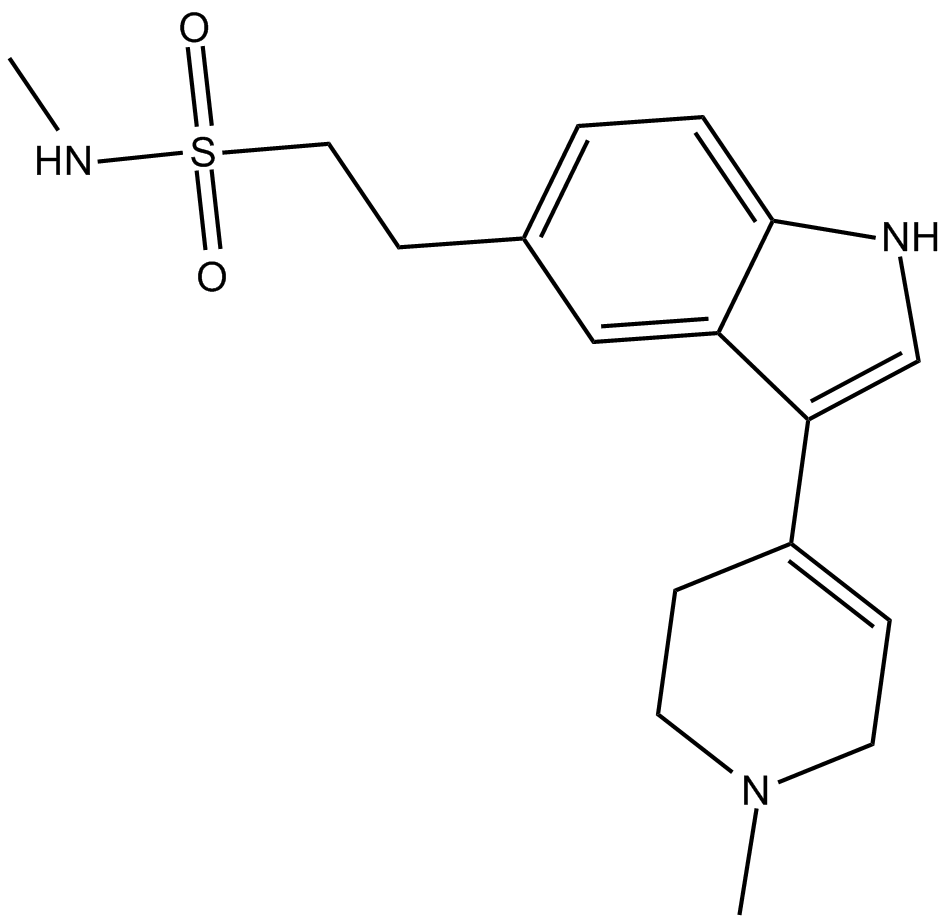
3,4-dihydro Naratriptan

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
pKi: 8.9 of Naratriptan for human 5-HT1B
3,4-dihydro Naratriptan is a selective serotonin 5-HT1B agonist.
5-HT1B receptors are widely distributed throughout the CNS with the highest concentrations found in the basal ganglia, frontal cortex, striatum, and the hippocampus. The function of the 5-HT1B receptor differs depending upon its location.
In vitro: 3,4-dihydro Naratriptan is an impurity formed during the preparation of naratriptan. Naratriptan had high affinity for human recombinant 5HT1B and 5HT1D receptors and could cause contractions of dog isolated basilar and middle cerebral artery. Naratriptan also caused small contractions of human isolated coronary arteries [1].
In vivo: In anaesthetized dogs, naratriptan caused selective vasoconstriction of the carotid arterial bed and, in anaesthetized rats, naratriptan selectively inhibited neurogenic plasma protein extravasation in the dura. In various antinociceptive tests, naratriptan had no effect even at high doses. In conscious rats and dogs, naratriptan had high oral bioavailability [1].
Clinical trial: Naratriptan has been approved for acute oral migraine therapy. In two Phase III trials of naratriptan compared with placebo, relief at four hours was obtained in 60% and 68% of patients using the 2.5-mg dose, with recurrence of headache in 24 hours in 27% and 28% of patients. Adverse effects of naratriptan were found to be similar to placebo, and its tolerability appeared superior compared with studies of other oral triptans [2].
References:
[1] Connor HE, Feniuk W, Beattie DT, North PC, Oxford AW, Saynor DA, Humphrey PP. Naratriptan: biological profile in animal models relevant to migraine. Cephalalgia. 1997 May;17(3):145-52.
[2] Dulli DA. Naratriptan: an alternative for migraine. Ann Pharmacother. 1999 Jun;33(6):704-11.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 333.4 |
| Cas No. | 121679-20-7 |
| Formula | C17H23N3O2S |
| Synonyms | Naratriptan Impurity B |
| Solubility | ≤0.1mg/ml in ethanol;10mg/ml in DMSO;10mg/ml in dimethyl formamide |
| Chemical Name | N-methyl-3-(1,2,3,6-tetrahydro-1-methyl-4-pyridinyl)-1H-indole-5-ethanesulfonamide |
| SDF | Download SDF |
| Canonical SMILES | O=S(CCC1=CC=C2C(C(C3=CCN(C)CC3)=CN2)=C1)(NC)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |