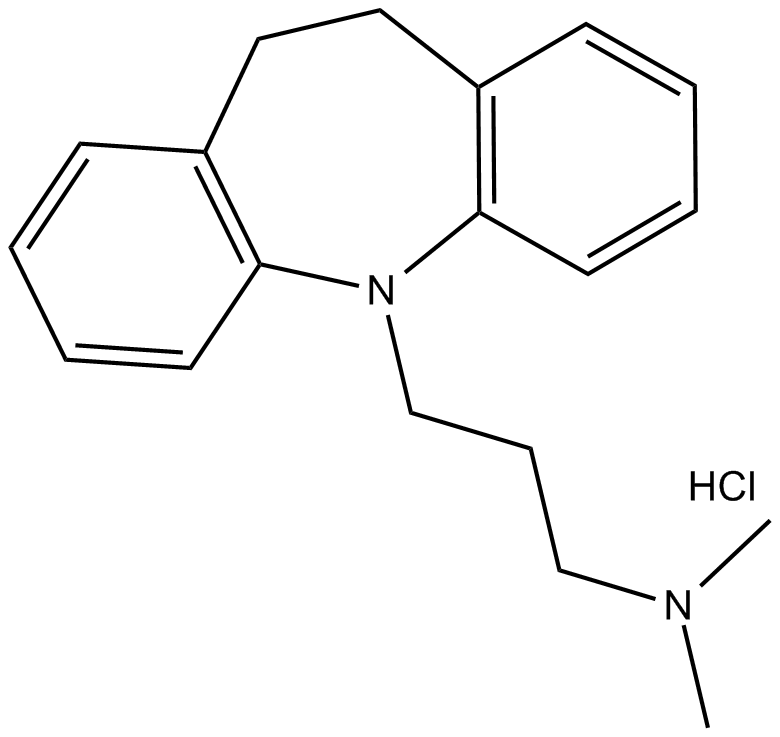
Imipramine (hydrochloride)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Imipramine (hydrochloride) is a tricyclic antidepressant and is mainly used in the treatment of major depression and enuresis [1].
Antidepressants are antagonists of many neurotransmitter receptors in human brain [3].
Imipramine is the first tricyclic antidepressant that acts mainly as an inhibitor of serotonin and norepinephrine transporters [2]. In radioligand binding assays, imipramine inhibited serotonin and norepinephrine transporters with KD values of 1.4 and 37 nM, respectively [2]. Imipramine is also inhibited histamine H1 receptor, muscarinic acetylcholine receptor and α1-adrenergic receptor with Kd values of 37, 46, and 32 nM, respectively [4].
In rodents, imipramine abolished the depressive syndrome produced by the acute administration of reserpine. Imipramine also possessed central anticholinergic activity and attenuate the activity of the centrally acting muscarinic agents tremorine and oxotremorine. Imipramine inhibited the presynaptic uptake of NA and 5-HT, and relatively weak against DA [1].
References:
[1]. Spencer PS. Review of the pharmacology of existing antidepressants. Br J Clin Pharmacol. 1977;4Suppl 2:57S-68S.
[2]. Tatsumi M, Groshan K, Blakely RD, et al. Pharmacological profile of antidepressants and related compounds at human monoamine transporters. Eur J Pharmacol. 1997 Dec 11;340(2-3):249-58.
[3]. Cusack B, Nelson A, Richelson E. Binding of antidepressants to human brain receptors: focus on newer generation compounds. Psychopharmacology (Berl). 1994 May;114(4):559-65.
| Storage | Store at -20°C |
| M.Wt | 316.9 |
| Cas No. | 113-52-0 |
| Formula | C19H24N2·HCl |
| Synonyms | Melipramine,Tofranil |
| Solubility | ≥12.5 mg/mL in DMSO; ≥22.3 mg/mL in H2O; ≥22.9 mg/mL in EtOH |
| Chemical Name | 10,11-dihydro-N,N-dimethyl-5H-dibenz[b,f]azepine-5-propanamine, monohydrochloride |
| SDF | Download SDF |
| Canonical SMILES | CN(C)CCCN1C2=C(C=CC=C2)CCC3=C1C=CC=C3.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构