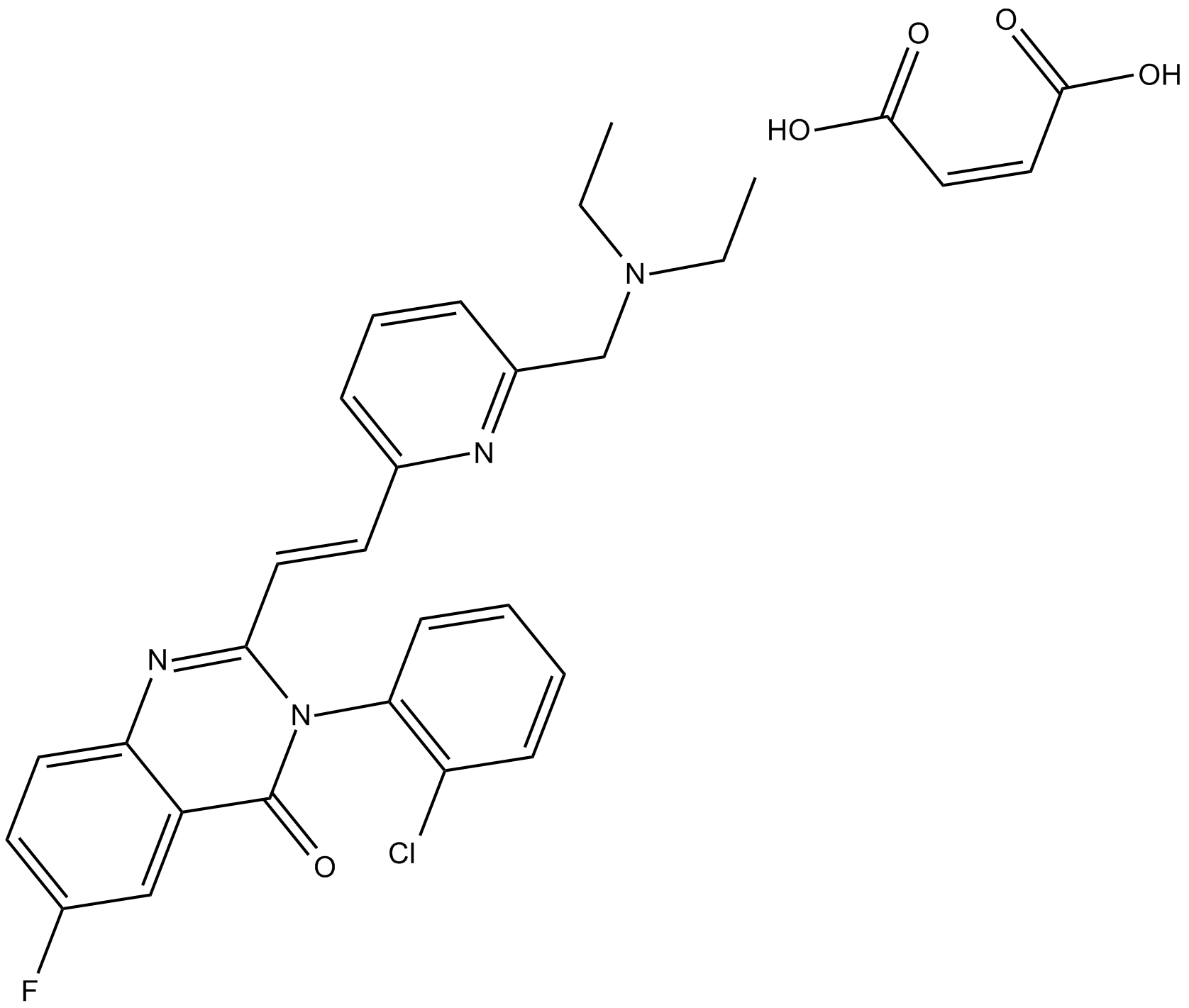
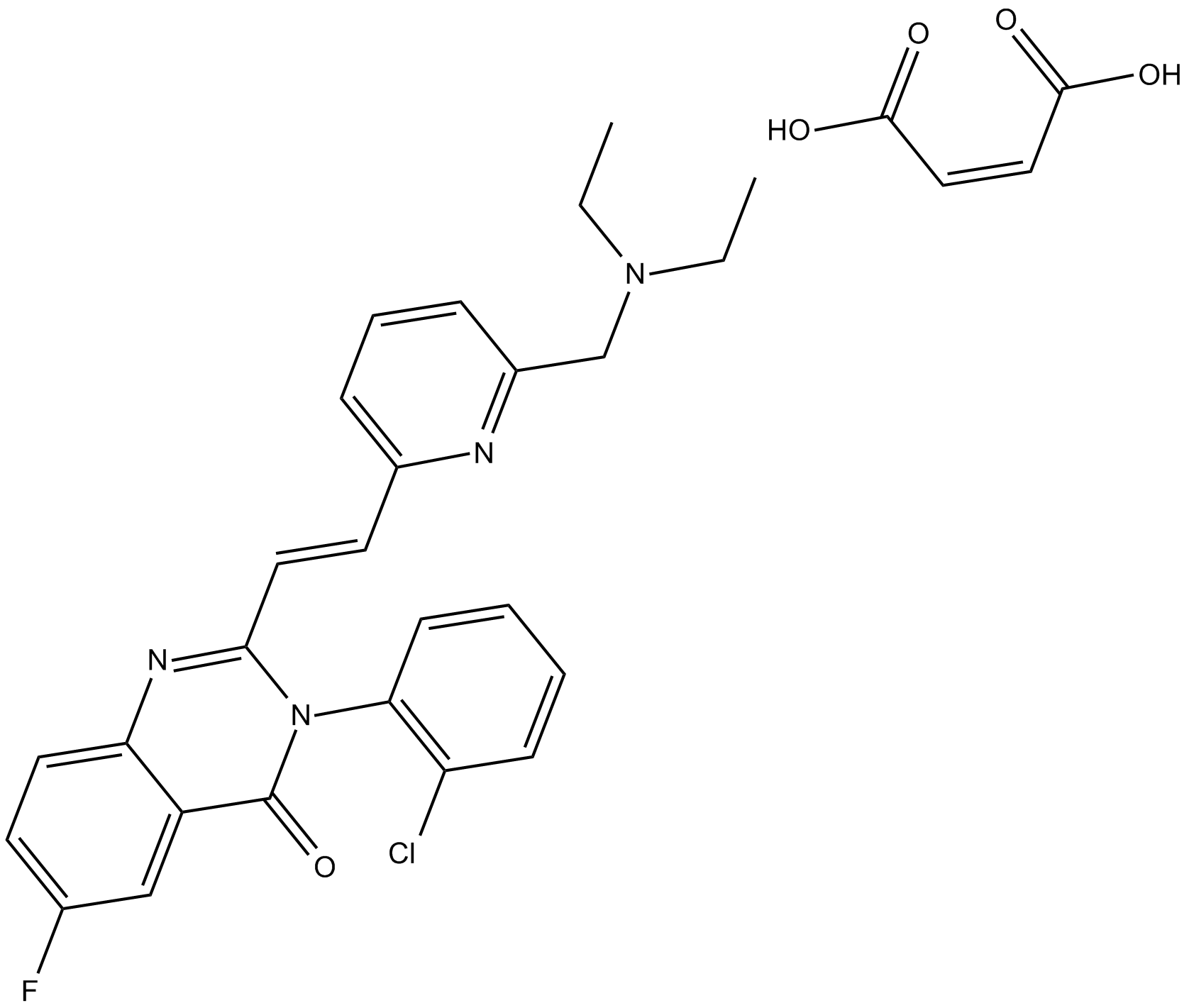
CP-465022 (maleate)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 25 nM for AMPA receptor-mediated currents in rat cortical neurons
CP-465022 is an AMPA antagonist.
The inhibition of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor has been hypothesized to lead to neuroprotective efficacy after cerebral ischemia on the basis of the activity in ischemia models of a variety of compounds with varying selectivity for AMPA over other glutamate receptor subtypes.
In vitro: CP-465022 inhibited AMPA receptor-mediated currents in rat cortical neurons and such inhibition was found to be noncompetitive with agonist concentration. CP-465022 was selective for AMPA over kainate and N-methyl-D-aspartate receptors. However, CP-465022 was found to be equipotent for AMPA receptors composed of different AMPA receptor subunit combinations, which indicated that CP-465022 is equivalently potent for inhibition of AMPA receptor-mediated responses in different types of neurons expressing different AMPA receptor subunits [1].
In vivo: Animal study showed that CP-465022 could potently and efficaciously inhibit AMPA receptor-mediated hippocampal synaptic transmission and the induction of seizures. However, at comparable doses, CP-465022 failed to prevent CA1 neuron loss after brief global ischemia or to reduce infarct volume after temporary middle cerebral artery occlusion [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Lazzaro JT, Paternain AV, Lerma J, Chenard BL, Ewing FE, Huang J, Welch WM, Ganong AH, Menniti FS. Functional characterization of CP-465,022, a selective, noncompetitive AMPA receptor antagonist. Neuropharmacology. 2002 Feb;42(2):143-53.
[2] Menniti FS, Buchan AM, Chenard BL, Critchett DJ, Ganong AH, Guanowsky V, Seymour PA, Welch WM. CP-465,022, a selective noncompetitive AMPA receptor antagonist, blocks AMPA receptors but is not neuroprotective in vivo. Stroke. 2003 Jan;34(1):171-6.
| Storage | Store at RT |
| M.Wt | 579 |
| Cas No. | 199656-46-7 |
| Formula | C26H24ClFN4O·C4H4O4 |
| Solubility | Soluble in DMSO |
| Chemical Name | 3-(2-chlorophenyl)-2-[2-[6-[(diethylamino)methyl]-2-pyridinyl]ethenyl]-6-fluoro-4(3H)-quinazolinone-(2Z)-2-butenedioate |
| SDF | Download SDF |
| Canonical SMILES | FC1=CC=C2C(C(N(C3=C(Cl)C=CC=C3)C(/C=C/C4=CC=CC(CN(CC)CC)=N4)=N2)=O)=C1.OC(/C=C\C(O)=O)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构