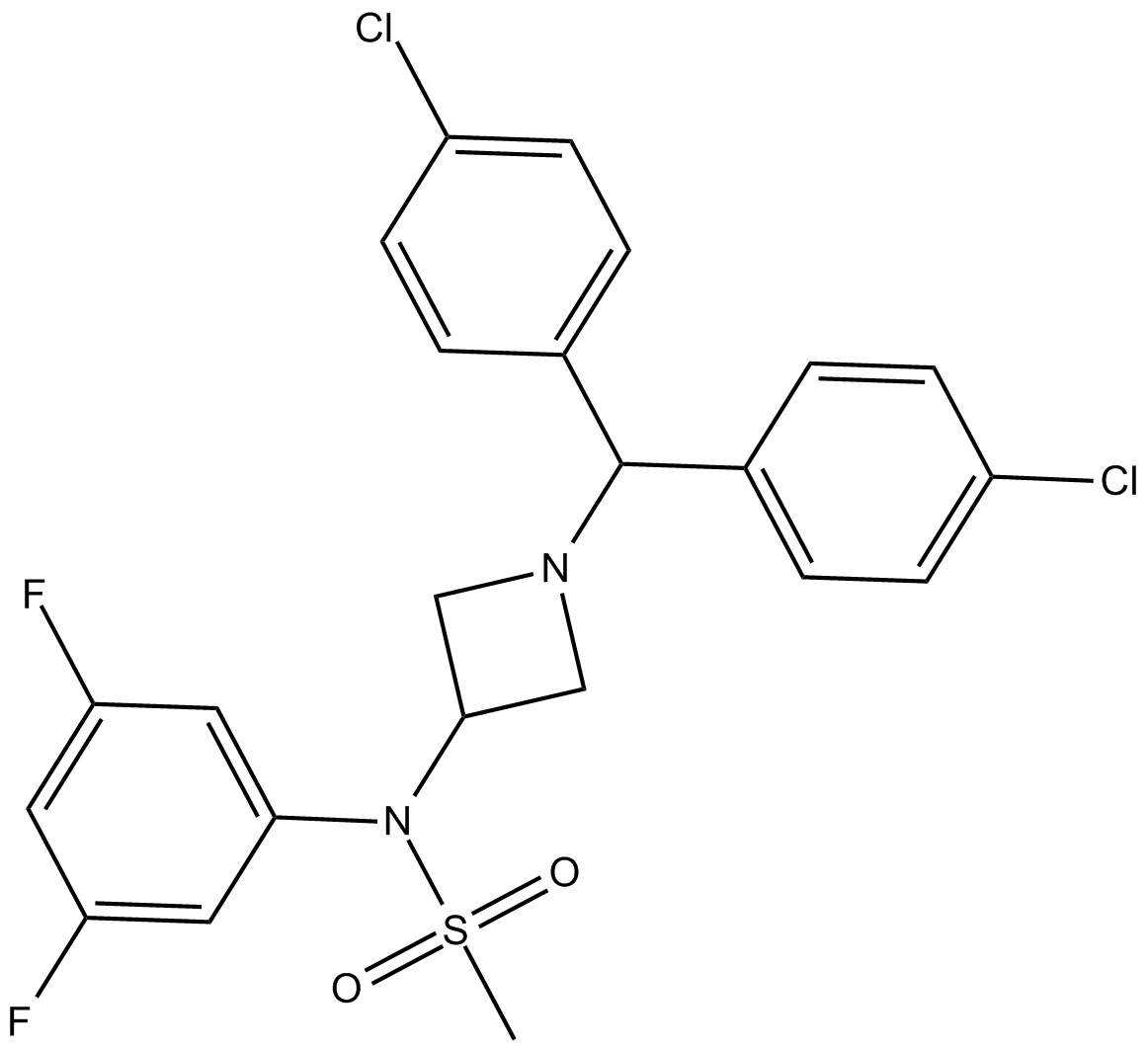
AVE-1625

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
AVE-1625 is a highly potent, selective antagonist for the CB1 receptor [1].
The cannabinoid receptor type 1 (CB1) is a G protein-coupled receptor mainly expressed in the central and peripheral nervous system. The CB1 receptor is activated by the endocannabinoid neurotransmitters anandamide and 2-arachidonoylglycerol (2-AG). The CB1 receptor has been implicated in the maintenance of homeostasis in health and disease [2]. The CB1 receptor plays vital roles in modulating neurotransmitter release by preventing the development of excessive neuronal activity, reducing pain and other inflammatory symptoms [2].
AVE-1625 antagonized the CB1 receptor activity with the Ki values of 0.16-0.44 nM [1]. Treatment with AVE-1625 (1-3 mg/kg) significantly improved the performance of rodents in working memory tasks. At 30 mg/kg, AVE-1625 reduced caloric intake by more than 50% of controls and significantly increased lipolysis from fat tissues and reduced hepatic glycogen levels in rodents. In Wistar rats, postprandially administration of AVE1625 slightly increased the basal lipolysis in a dose-dependent manner. AVE1625 caused primary effects on metabolic blood and tissue parameters as well as metabolic rate [3]. As measured by indirect calorimetry, AVE1625 immediately increased the total energy expenditure and a transiently increased glucose oxidation [3].
References:
[1] Borowsky B, Stevens R, Mark B, et al. AVE1625, a cannabinoid CBI antagonist, as a co-treatment for schizophrenia: Improvement in cognitive function and reduction of antipsychotic-side effects in animal models[C]//Neuropsychopharmacology. Macmillan building, 4 crinan st, london n1 9xw, ENGLAND: NATURE PUBLISHING GROUP, 2005, 30: S116-S117.
[2] Herkenham M, Lynn A B, Little M D, et al. Cannabinoid receptor localization in brain[J]. Proceedings of the national Academy of sciences, 1990, 87(5): 1932-1936.
[3] Herling A W, Gossel M, Haschke G, et al. CB1 receptor antagonist AVE1625 affects primarily metabolic parameters independently of reduced food intake in Wistar rats[J]. American Journal of Physiology-Endocrinology and Metabolism, 2007, 293(3): E826-E832.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 497.4 |
| Cas No. | 358970-97-5 |
| Formula | C23H20Cl2F2N2O2S |
| Synonyms | Drinabant |
| Solubility | ≤0.15mg/ml in ethanol;15mg/ml in DMSO;15mg/ml in dimethyl formamide |
| Chemical Name | N-[1-[bis(4-chlorophenyl)methyl]azetidin-3-yl]-N-(3,5-difluorophenyl)methanesulfonamide |
| SDF | Download SDF |
| Canonical SMILES | CS(=O)(N(C1CN(C(C2=CC=C(Cl)C=C2)C3=CC=C(Cl)C=C3)C1)C4=CC(F)=CC(F)=C4)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |