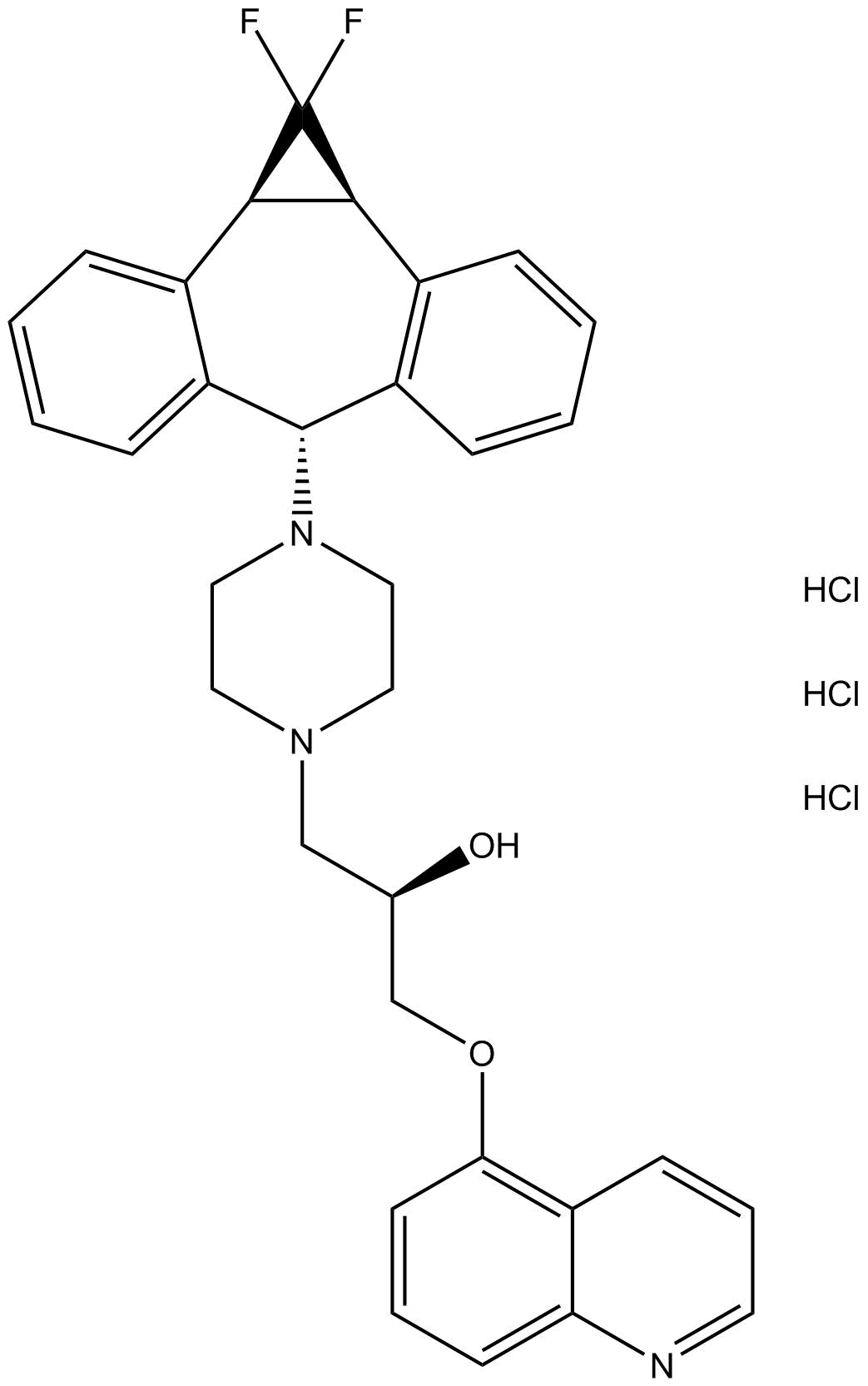
Zosuquidar

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Zosuquidar是一种新型有效的P-糖蛋白调节剂[1]。P-糖蛋白(P-gp)在脑、肝脏、小肠和肿瘤细胞中表达。在肿瘤细胞中,P-糖蛋白(P-gp)作为外排泵发挥功能,负责多药耐药性。肿瘤中P-糖蛋白(Pgp)过表达导致对结构上不相关的溶瘤剂的多药耐药性(MDR)[2]。
体外实验:在CEM/VLB100细胞中,LY335979(0.1 μM)完全恢复细胞对vinblastine、doxorubicin (Dox)、etoposide和Taxol的敏感性。在CEM/VLB100质膜中,LY335979阻断[3H]叠氮基光亲和标记M(r)约170,000的 Pgp,并竞争性抑制[3H]vinblastine与Pgp的结合平衡,Ki值约为0.06 μM[3]。在所有表达P-gp的白血病细胞系中,包括K562/HHT40、K562/HHT90、K562/DOX和HL60/DNR中,Zosuquidar完全或部分恢复药物敏感性。在具有活性P-gp的原代AML母细胞中,Zosuquidar增强anthracyclines (daunorubicin、idarubicin、mitoxantrone)和 gemtuzumab ozogamicin (Mylotarg)的细胞毒性[4]。
在体实验:在携带P388/ADR鼠白血病细胞的小鼠中,LY335979与Dox或etoposide组合治疗显著延长小鼠寿命,药代动力学没有明显改变。在MDR人非小细胞肺癌裸鼠异种移植模型中,LY335979增强Taxol的抗肿瘤活性[3]。
临床试验:在未治疗的非霍奇金淋巴瘤患者中,进行I/II期临床试验研究zosuquidar的安全性和耐受性。在口服三个500 mg剂量的zosuquidar与CHOX组合给药,药物毒性最小,并且没有观察到CHOP相关毒性的增强[5]。在I期研究的晚期实体瘤患者中,zosuquidar(100-300 mg/m2)可以抑制vinorelbine的清除率至适度程度[6]。
参考文献:
Cripe L D, Uno H, Paietta E M, et al. Zosuquidar, a novel modulator of P-glycoprotein, does not improve the outcome of older patients with newly diagnosed acute myeloid leukemia: a randomized, placebo-controlled trial of the Eastern Cooperative Oncology Group 3999[J]. Blood, 2010, 116(20): 4077-4085.
Chaudhary P M, Roninson I B. Expression and activity of P-glycoprotein, a multidrug efflux pump, in human hematopoietic stem cells[J]. Cell, 1991, 66(1): 85-94.
Dantzig A H, Shepard R L, Cao J, et al. Reversal of P-glycoprotein-mediated multidrug resistance by a potent cyclopropyldibenzosuberane modulator, LY335979[J]. Cancer Research, 1996, 56(18): 4171-4179.
Tang R, Faussat A M, Perrot J Y, et al. Zosuquidar restores drug sensitivity in P-glycoprotein expressing acute myeloid leukemia (AML)[J]. BMC cancer, 2008, 8(1): 1.
Morschhauser F, Zinzani P L, Burgess M, et al. Phase I/II trial of a P-glycoprotein inhibitor, Zosuquidar. 3HCl trihydrochloride (LY335979), given orally in combination with the CHOP regimen in patients with non-Hodgkin's lymphoma[J]. Leukemia & lymphoma, 2007, 48(4): 708-715.
Lê L H, Moore M J, Siu L L, et al. Phase I study of the multidrug resistance inhibitor zosuquidar administered in combination with vinorelbine in patients with advanced solid tumours[J]. Cancer chemotherapy and pharmacology, 2005, 56(2): 154-160.
| Storage | Store at -20°C |
| M.Wt | 527.6 |
| Cas No. | 167354-41-8 |
| Formula | C32H31F2N3O2 |
| Synonyms | LY335979;LY 335979;LY-335979 |
| Solubility | Soluble in DMSO |
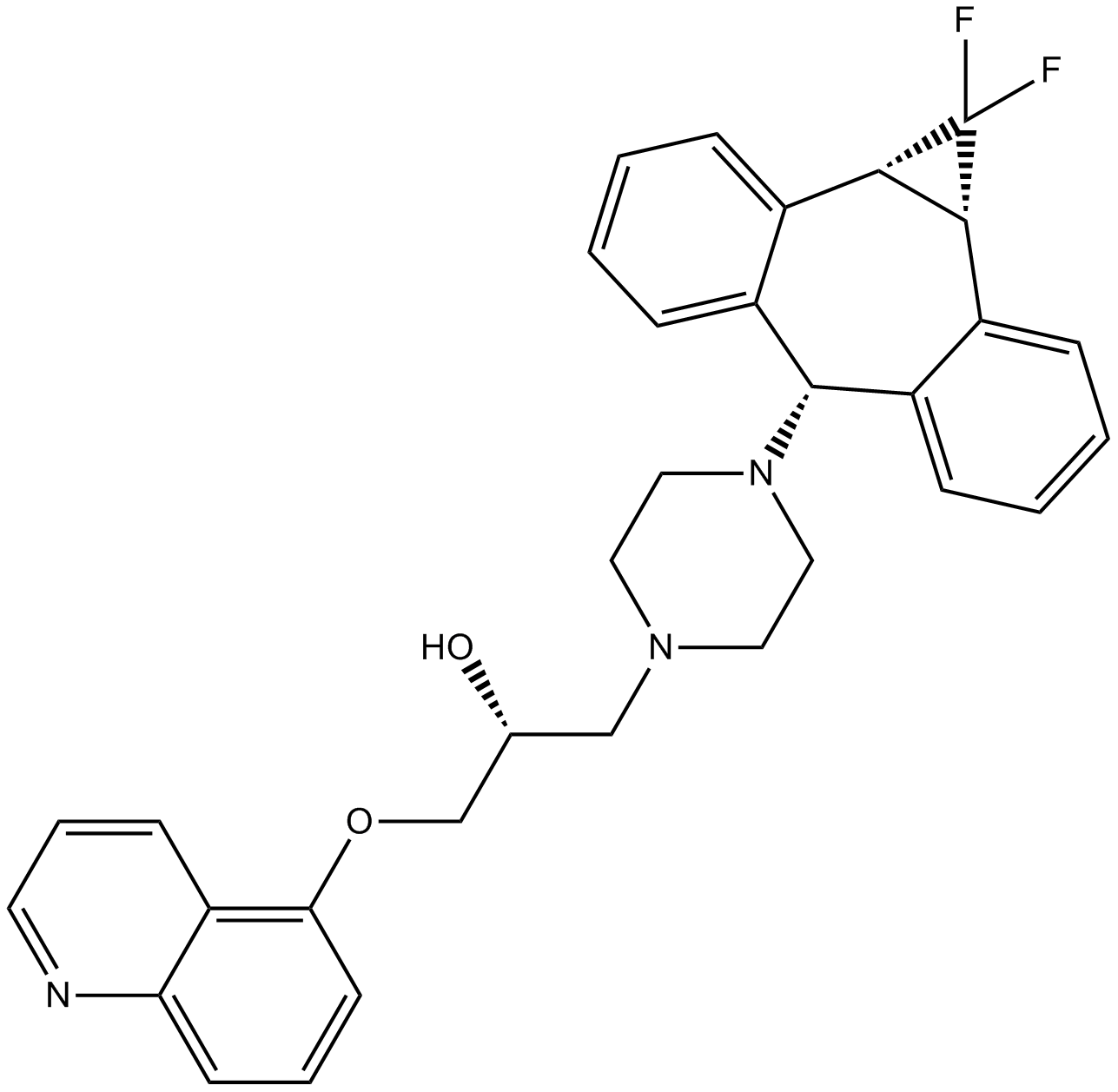
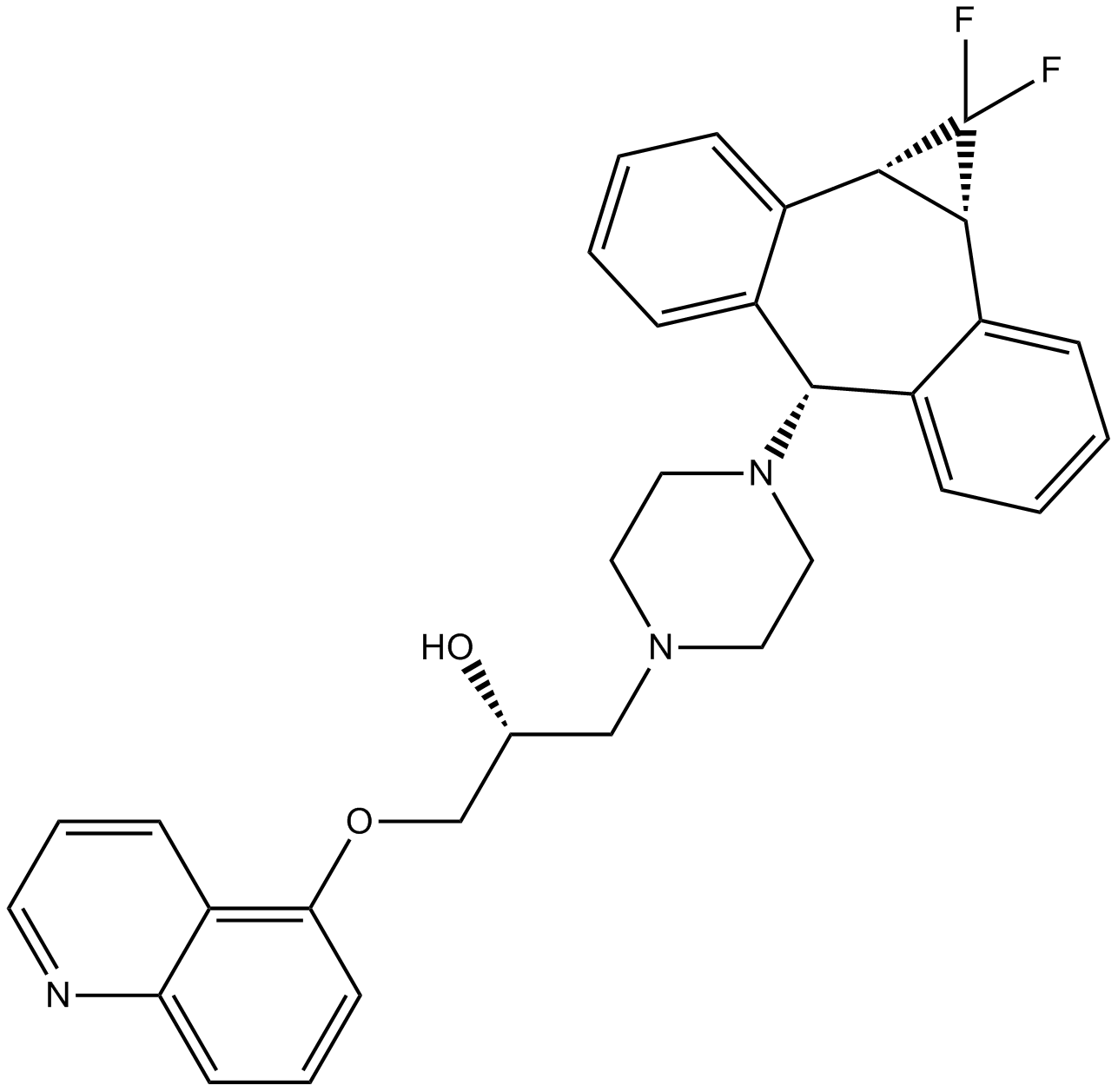
| Chemical Name | (2R)-1-(4-((1aR,10bS)-1,1-difluoro-1,1a,6,10b-tetrahydrodibenzo[a,e]cyclopropa[c][7]annulen-6-yl)piperazin-1-yl)-3-(quinolin-5-yloxy)propan-2-ol |
| SDF | Download SDF |
| Canonical SMILES | FC1([C@@](C2=C([H])C([H])=C([H])C([H])=C23)([H])[C@]1([H])C4=C([H])C([H])=C([H])C([H])=C4C3([H])N5C([H])([H])C([H])([H])N(C([H])([H])[C@@](C([H])([H])OC6=C([H])C([H])=C([H])C7=C6C([H])=C([H])C([H])=N7)([H])O[H])C([H])([H])C5([H])[H])F |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构