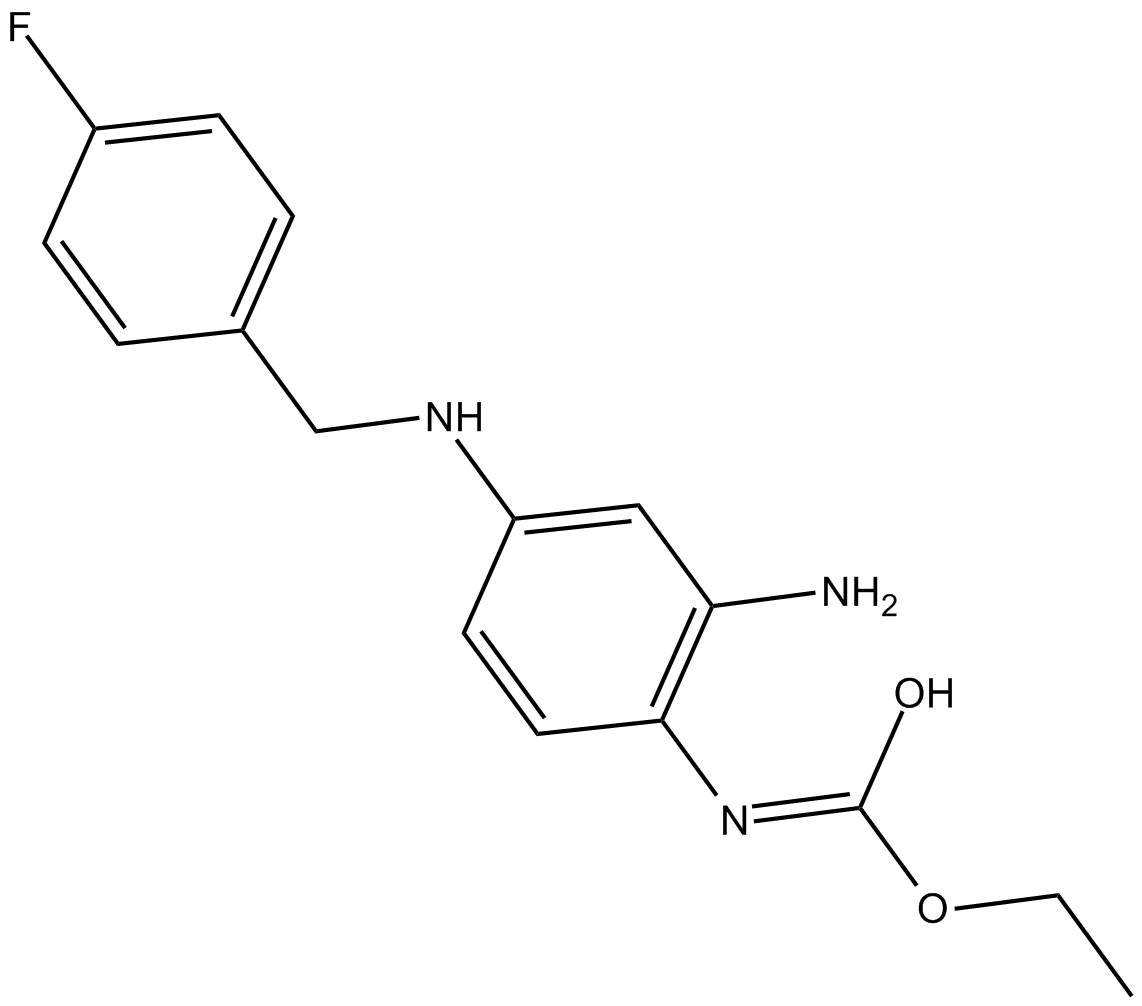
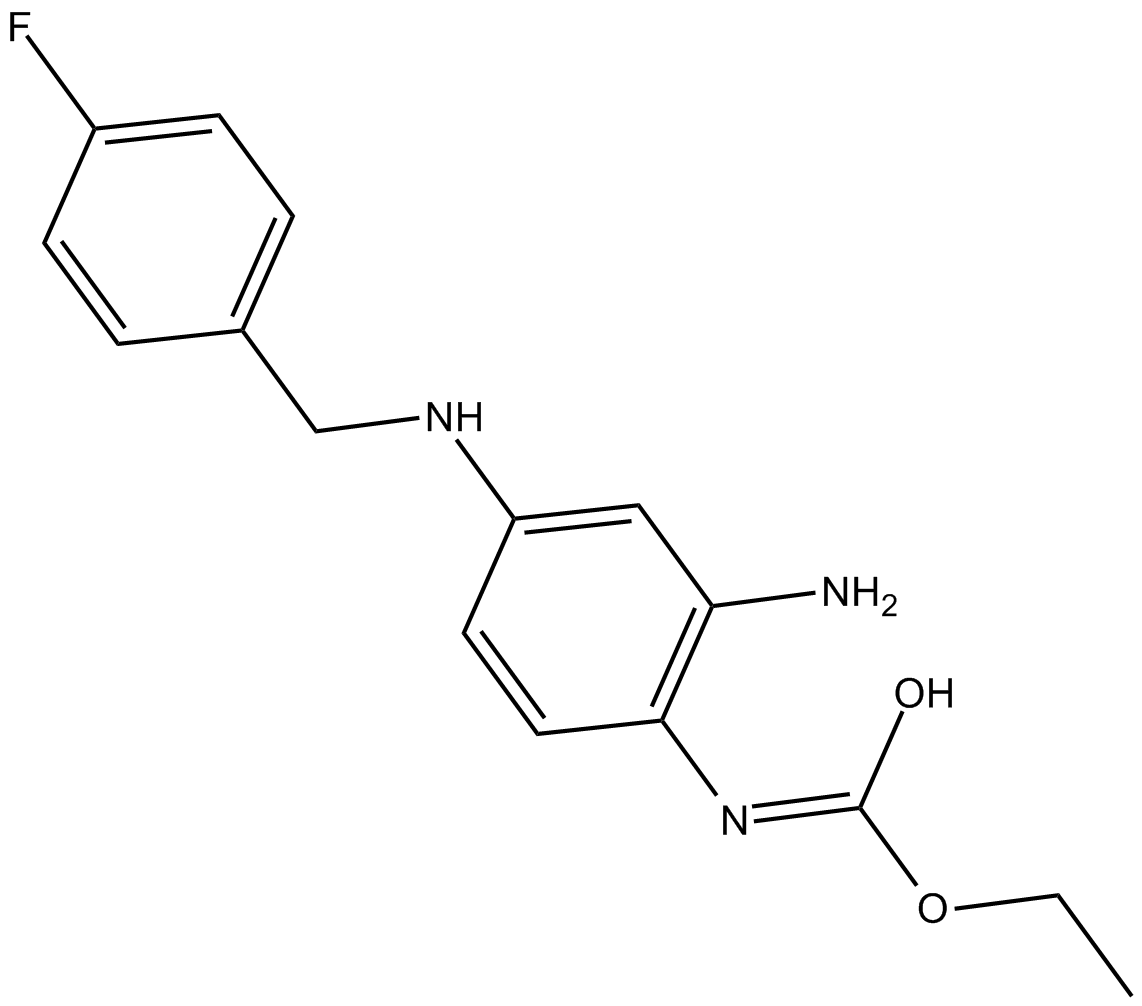
Retigabine

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Retigabine is a positive allosteric modulator of KCNQ2-5 (KV7.2-7.5) ion channels, with EC50 values being 1.4, 1.6 and 5.2 μM for KCNQ3/5, KCNQ2/3 and KCNQ4, respectively. Retigabine is the first neuronal potassium channel opener used for the treatment of epilepsy. KCNQ2-5 channels, predominantly expressed in neurons, are important determinants of cellular excitability. The occurrence of human genetic mutations in KCNQ2/3 channels are associated with the syndrome of benign familial neonatal convulsions.
References:
1. Gunthorpe MJ, Large CH, Sankar R. The mechanism of action of retigabine (ezogabine), a first-in-class K+ channel opener for the treatment of epilepsy. Epilepsia, 2012, 53(3): 412-424.
2. Stas JI, Bocksteins E, Jensen CS, et al. The anticonvulsant retigabine suppresses neuronal KV2-mediated currents. Scientific Reports, 2016, 6: 35080.
3. De Sarro G, Di Paola ED, Conte G, et al. Influence of retigabine on the anticonvulsant activity of some antiepileptic drugs against audiogenic seizures in DBA/2 mice. Naunyn-Schmiedeberg's Archives of Pharmacology, 2001, 363(3): 330-336.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 303.33 |
| Cas No. | 150812-12-7 |
| Formula | C16H18FN3O2 |
| Solubility | insoluble in H2O; ≥11.1 mg/mL in EtOH with ultrasonic; ≥12.95 mg/mL in DMSO |
| Chemical Name | (E)-ethyl hydrogen (2-amino-4-((4-fluorobenzyl)amino)phenyl)carbonimidate |
| SDF | Download SDF |
| Canonical SMILES | CCO/C(O)=N/C1=C(N)C=C(NCC2=CC=C(F)C=C2)C=C1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Cell experiment:[2] | |
|
Cell lines |
HEK293 cells transfected with KV2.1 |
|
Reaction Conditions |
0.1, 0.3, 1 or 3 μM retigabine for 4 h incubation |
|
Applications |
Retigabine reduced the KV2.1 current density in a concentration-dependent manner. The current density was significantly reduced by approximately 2.5-fold after exposure to 1 and 3 μM retigabine. Thus, KV2.1 was identified as a new molecular target for retigabine, with the potential to further explain retigabine's neuroprotective properties. |
| Animal experiment:[3] | |
|
Animal models |
DBA/2 mice |
|
Dosage form |
0.5 ~ 20 mg/kg Intraperitoneal administration |
|
Applications |
Retigabine exhibited an additive effect when administered in combination with classical anticonvulsants, most notably diazepam, phenobarbital, phenytoin as well as valproate. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Gunthorpe MJ, Large CH, Sankar R. The mechanism of action of retigabine (ezogabine), a first-in-class K+ channel opener for the treatment of epilepsy. Epilepsia, 2012, 53(3): 412-424. 2. Stas JI, Bocksteins E, Jensen CS, et al. The anticonvulsant retigabine suppresses neuronal KV2-mediated currents. Scientific Reports, 2016, 6: 35080. 3. De Sarro G, Di Paola ED, Conte G, et al. Influence of retigabine on the anticonvulsant activity of some antiepileptic drugs against audiogenic seizures in DBA/2 mice. Naunyn-Schmiedeberg's Archives of Pharmacology, 2001, 363(3): 330-336. |
|
Quality Control & MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure