F16

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
F16 is a small, cationic, lipophilic, and fluorescent molecule capable of binding preferentially to mitochondrial membranes and disrupts their function. F16 is a potential antitumor agent.
F16 affected growth in some mouse and human breast cancer cell lines. F16 resulted in a dramatic decrease in the number of cells in S phase and an increase in the percentage of cells in G1 phase [1]. Prolonged incubation with 3 μM F16 led to increased cell death of F16-sensitive cells but not of F16-resistant ones. F16 accumulation in mitochondria induced mitochondrial damage characterized by imbalance of volumetric homeostasis, failure to synthesize ATP, cytochrome c release and increased production of reactive oxy gen species [1]. F16 incubation decreased the cellular ATP pool in both parental EpH4-A6 and Bcl-2-overexpressing EpH4-A6.C13 and EpH4-A6.C18 cells in a time-dependent manner [1]. Treatment with F16 promoted early release of cytochrome c in transformed EpH4-A6 cells. Treatment with F16 (0.3-3 μM) resulted in the characteristic apoptotic DNA laddering in the EpH4-A6 cells. F16-Induced mitochondrial dysfunction triggers apoptosis or necrosis. F16 induced necrosis in various cell lines resistant to apoptosis [2].
References:
[1] Fantin V R, Berardi M J, Scorrano L, et al. A novel mitochondriotoxic small molecule that selectively inhibits tumor cell growth[J]. Cancer cell, 2002, 2(1): 29-42.
[2] Fantin V R, Leder P. F16, a mitochondriotoxic compound, triggers apoptosis or necrosis depending on the genetic background of the target carcinoma cell[J]. Cancer research, 2004, 64(1): 329-336.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 362.2 |
| Cas No. | 36098-33-6 |
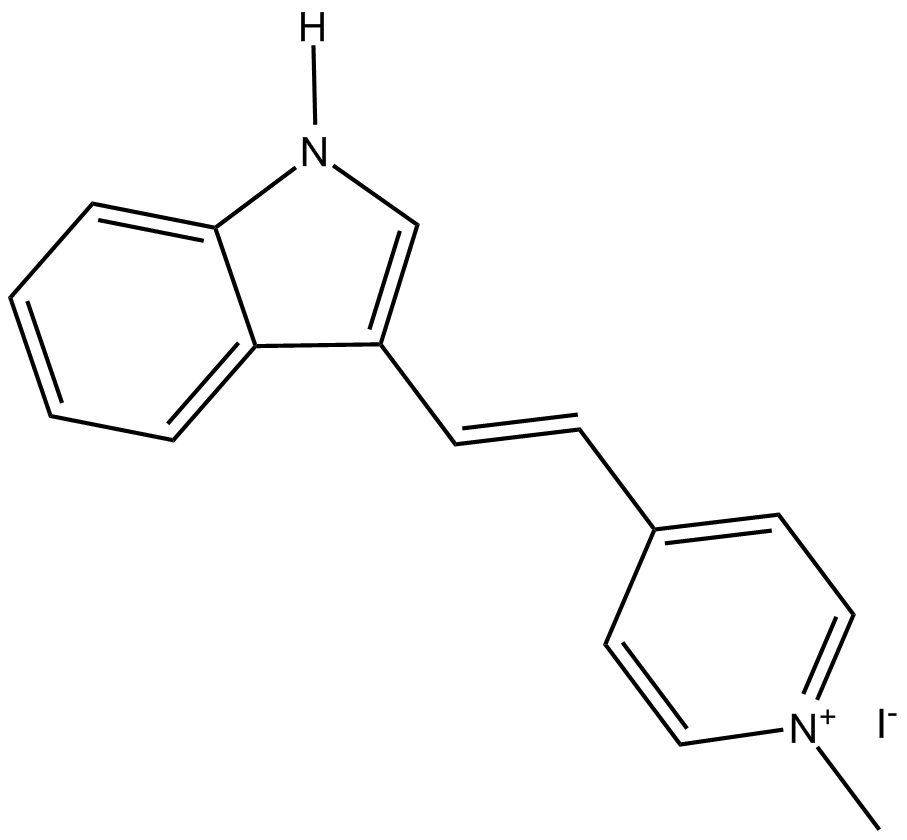
| Formula | C16H15N2·I |
| Solubility | ≤500μg/ml in ethanol;20mg/ml in DMSO;25mg/ml in dimethyl formamide |
| Chemical Name | 4-[(1E)-2-(1H-indol-3-yl)ethenyl]-1-methyl-pyridinium iodide |
| SDF | Download SDF |
| Canonical SMILES | [H]N1C=C(/C=C/C2=CC=[N+](C)C=C2)C3=CC=CC=C31.[I-] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |