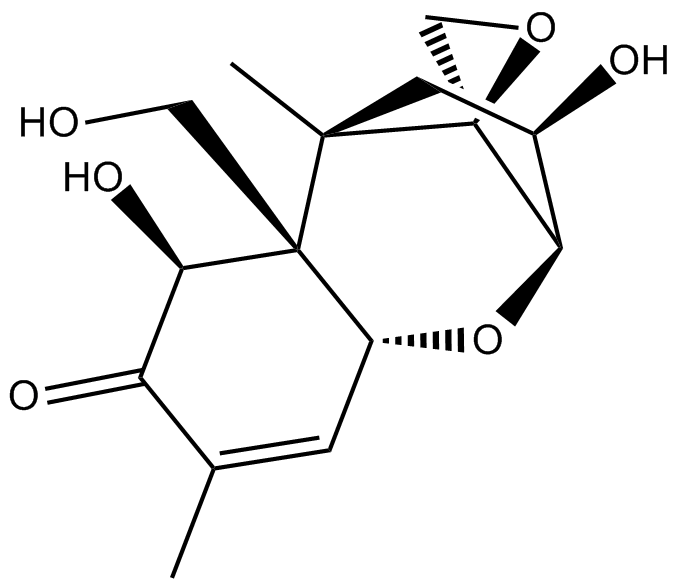
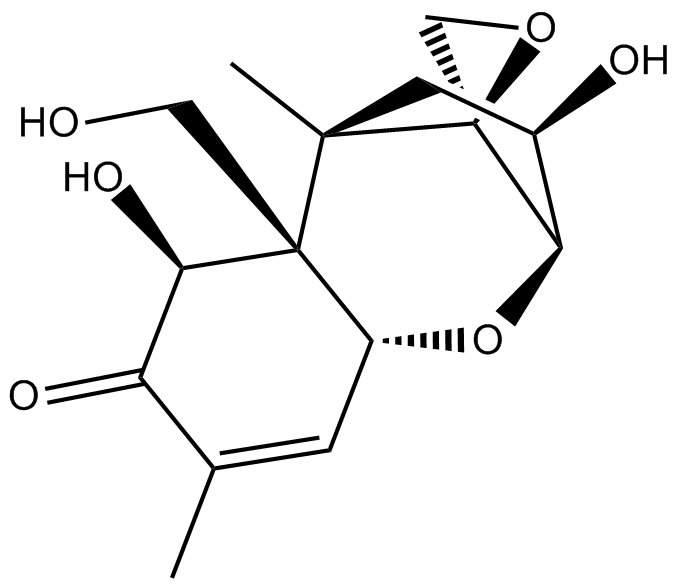
Deoxynivalenol

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Deoxynivalenol, also known as vomitoxin, is a foodborne mycotoxin, acting as a potent inhibitor of protein synthesis. It stimulates the pro-inflammatory response, causes ribotoxic stress, cytotoxicity and apoptosis, as well as results in the impairment of multiple physiological functions, such as the intestinal barrier, growth, immune regulation and reproduction [1].
In immortalized human kidney epithelial cells, deoxynivalenol induced cytotoxicity, with EC50 value of 1.1 µM [2]. In cloned macrophages and monocytes, the Jurkat human T-cell model, and cloned B cells, deoxynivalenol triggered apoptosis at concentrations ranging from 100 to 1000 ng/ml [3].
In swine, 50 µg/kg deoxynivalenol (i.p. or oral) could elicit emesis. Rapid onset of vomiting in pigs also occurred upon ingestion of feed contaminated with 19.7 ppm deoxynivalenol. Swine consuming 2 and 4 ppm deoxynivalenol exhibited a dose-related decrease in weight gain within the first 8 weeks of feeding, with 4 ppm causing decreased feed intake, weight gain, and efficiency of feed utilization throughout the experiment [3].
References:
[1]. Vidal A, Claeys L, Mengelers M, et al. Humans significantly metabolize and excrete the mycotoxin deoxynivalenol and its modified form deoxynivalenol-3-glucoside within 24 hours. Scientific Reports, 2018, 8(1): 5255.
[2]. Bretz M, Beyer M, Cramer B, et al. Thermal degradation of the Fusarium mycotoxin deoxynivalenol. Journal of Agricultural and Food Chemistry, 2006, 54(17): 6445-6451.
[3]. Pestka J J. Deoxynivalenol: mechanisms of action, human exposure, and toxicological relevance. Archives of Toxicology, 2010, 84(9): 663-679.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 296.32 |
| Cas No. | 51481-10-8 |
| Formula | C15H20O6 |
| Solubility | Soluble in DMSO |
| Chemical Name | (2R,2'S,3R,5R,5aR,6S,9aR)-3,6-dihydroxy-5a-(hydroxymethyl)-5,8-dimethyl-2,3,4,5,5a,6-hexahydrospiro[2,5-methanobenzo[b]oxepine-10,2'-oxiran]-7(9aH)-one |
| SDF | Download SDF |
| Canonical SMILES | O[C@H]1[C@H]2O[C@H](C=C(C)C3=O)[C@@]([C@@H]3O)(CO)[C@](C1)(C)[C@@]24OC4 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- View current batch:
-
Purity = 98.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
化学结构