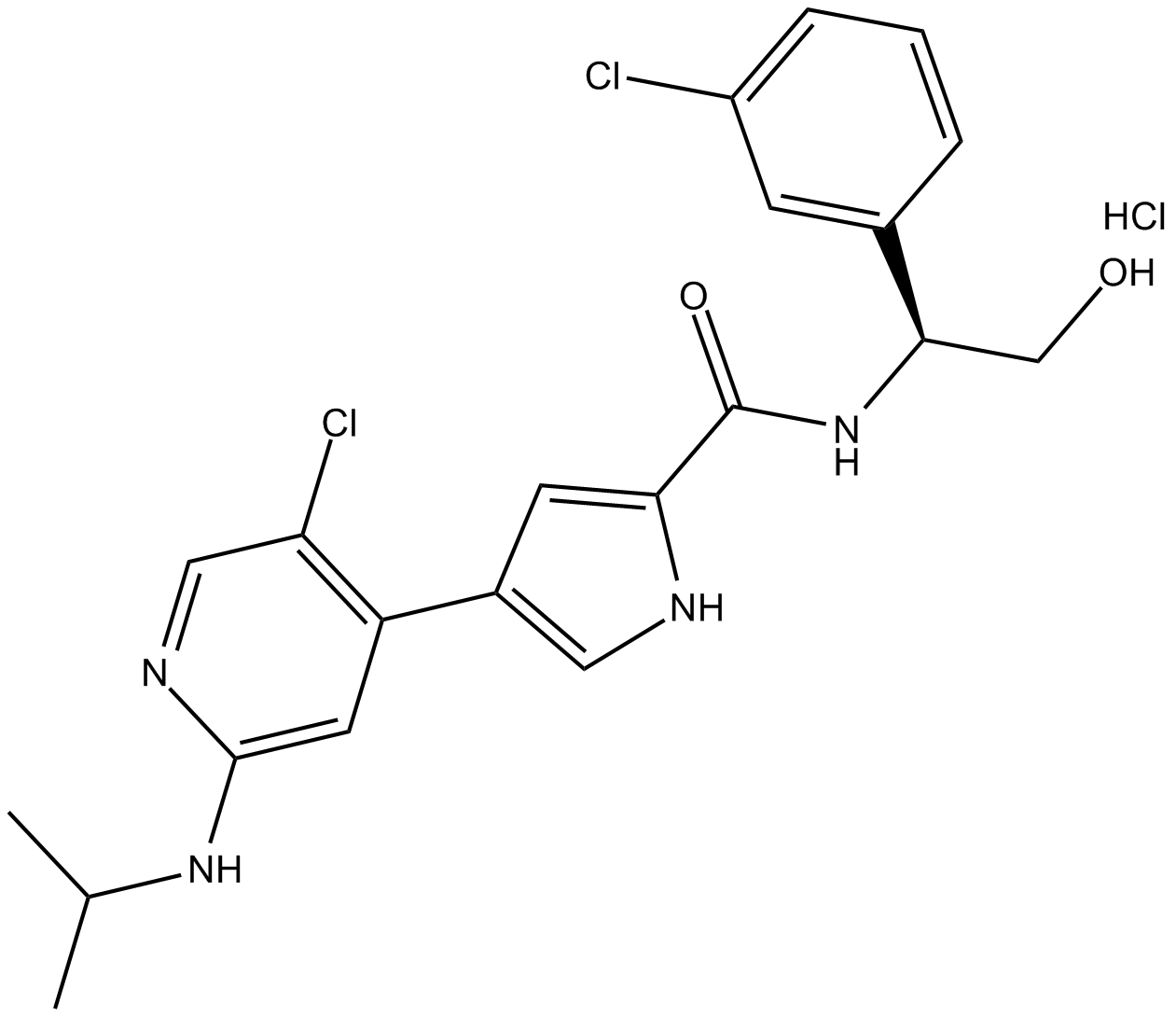
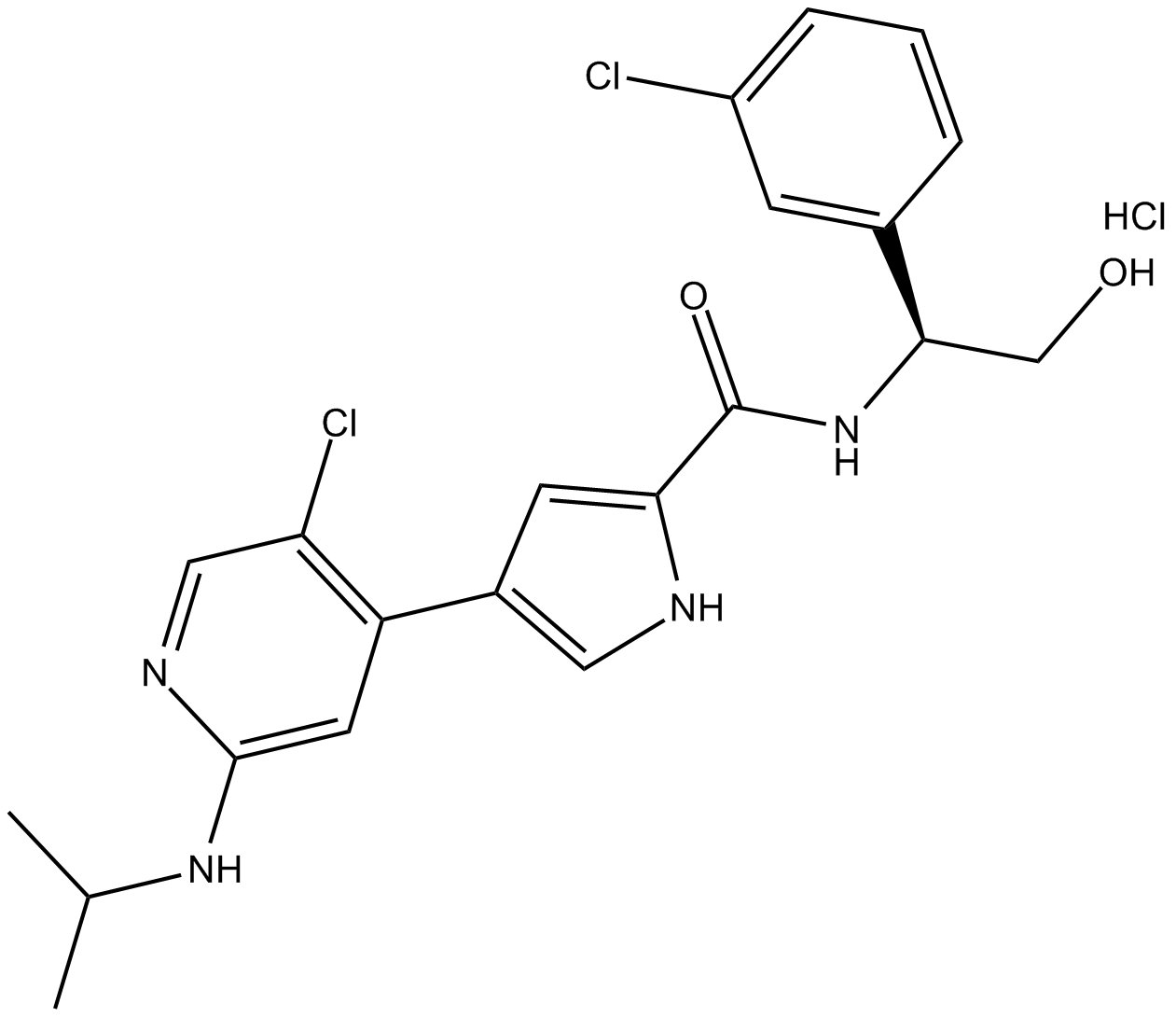
Ulixertinib (hydrochloride)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Ulixertinib, also named as BVD-523, is a novel and reversible inhibitor of ERK1/2. Ulixertinib potently and selectively inhibits the activity of ERK1 and ERK2 kinases in a reversible, ATP-competitive fashion [1].
The Ras-dependent extracellular signal-regulated kinase (ERK)1/2 mitogen-activated protein (MAP) kinase pathway plays a central role in cell proliferation control. In normal cells, sustained activation of ERK1/ERK2 is necessary for G1- to S-phase progression and is associated with induction of positive regulators of the cell cycle and inactivation of antiproliferative genes. The RAF-MEK-ERK1/2 signal pathway plays a dominant role in promoting cell survival [2].
In vitro: In two lymphoma cell lines (SUDHL-10 and Raji), treatment with ulixertinib significantly reduced the expression of ERK1/2 phosphorylation in a dose-dependent manner. Treatment with 0.4 nM ulixertinib decreased the percentage of G2-M phase cells in the SUDHL-10 cells. In the Raji cells, treated with ulixertinib at 0.4 and 1.0 nM increased the percentage of G0-G1 phase cells and decreased S phase cells [1]. Treatment of ulixertinib at the dose of 0.1, 0.4 and 1.0 nM for 48 h dose-dependently increased the number of early apoptotic SUDHL- 10 and Raji cells [1]. In SUDHL-10 and Raji cells, ulixertinib reduced mRNA and protein expression of VEGFR2 and Bcl-2 genes and increased the expression of Bax and caspase-3 genes [1].
In vivo: BVD-523 inhibited tumor growth in BRAF-mutant melanoma and colorectal xenografts as well as in KRAS-mutant colorectal and pancreatic models. BVD-523 treatment in combination with dabrafenib inhibited tumor growth in a BRAF-mutant melanoma model [3]. Single-agent BVD-523 inhibited the growth of a patient-derived tumor xenograft harboring cross-resistance to dabrafenib, trametinib, and the combination treatment following clinical progression on a MEK inhibitor [3].
Clinical trials: In patients (pts) with advanced solid tumors, BVD-523 (ulixertinib) achieved pharmacologically relevant exposure and manageable tolerability at its MTD of 600 mg twice a day [4].
References:
[1] Ding W J, Tao W P, Zeng T, et al. Selective ERK inhibitor ulixertinib inhibits proliferation and induces apoptosis in lymphoma cell lines[J]. INTERNATIONAL JOURNAL OF CLINICAL AND EXPERIMENTAL MEDICINE, 2016, 9(6): 10955-10962.
[2] Meloche S, Pouysségur J. The ERK1/2 mitogen-activated protein kinase pathway as a master regulator of the G1-to S-phase transition[J]. Oncogene, 2007, 26(22): 3227-3239.
[3] Germann U, Furey B, Roix J, et al. The selective ERK inhibitor BVD-523 is active in models of MAPK pathway-dependent cancers, including those with intrinsic and acquired drug resistance[J]. 2015.
[4] Infante J R, Janku F, Tolcher A W, et al. Dose escalation stage of a first-in-class phase I study of the novel oral ERK 1/2 kinase inhibitor BVD-523 (ulixertinib) in patients with advanced solid tumors[J]. 2015
.| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 469.8 |
| Cas No. | 1956366-10-1 |
| Formula | C21H22Cl2N4O2·HCl |
| Synonyms | BVD-523,VRT-752271 |
| Solubility | insoluble in H2O; ≥20.15 mg/mL in DMSO; ≥6.88 mg/mL in EtOH |
| Chemical Name | 4-[5-chloro-2-[(1-methylethyl)amino]-4-pyridinyl]-N-[(1S)-1-(3-chlorophenyl)-2-hydroxyethyl]-1H-pyrrole-2-carboxamide, monohydrochloride |
| SDF | Download SDF |
| Canonical SMILES | CC(C)NC1=CC(C2=CNC(C(N[C@H](CO)C3=CC(Cl)=CC=C3)=O)=C2)=C(Cl)C=N1.Cl |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构