Nullscript

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Nullscript is an HDAC inhibitor.
Histone deacetylase inhibitors (HDIs) have been used in psychiatry and neurology as mood stabilizers and anti-epileptics, such as valproic acid. Recently, HDIs are being studied as a mitigator or treatment for neurodegenerative diseases. Moreover, there has been an effort to develop HDIs for cancer therapy.
In vitro: Nullscript, a close analog of scriptaid, was found to be inactive in transcriptional facilitation at corresponding concentrations, which confirmed a minimal requirement for the length of the linker chain expected for this class of HDAC inhibitors. In addition, nullscript was not able to induce the p6SBE-luc reporter construct, which was identified from the library using ChemFinder by its structural similarity to scriptaid [1].
In vivo: A standard in vivo model of cardiac I/RWe was utilized to examine the in vivo consequences of HDAC inhibition in the intact heart. Results showed that the treatment with scriptaid led to a nearly identical effect when compared to nullscript, with a 46.8% reduction in infarct size. Such results strongly suggested that in murine models, HDACIs could reverse the induction of ischemia-induced HDAC activity and reduced myocardial infarct size by more than 50% [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] G. H. Su, T. A. Sohn, B. Ryu, et al. A novel histone deacetylase inhibitor identified by high-throughput transcriptional screening of a compound library. Cancer Research 60, 3137-3142 (2000).
[2] Anne Granger et al. Histone deacetylase inhibition reduces myocardial ischemia-reperfusion injury in mice. FASEB J. 2008 Oct; 22(10): 3549–3560.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 298.3 |
| Cas No. | 300816-11-9 |
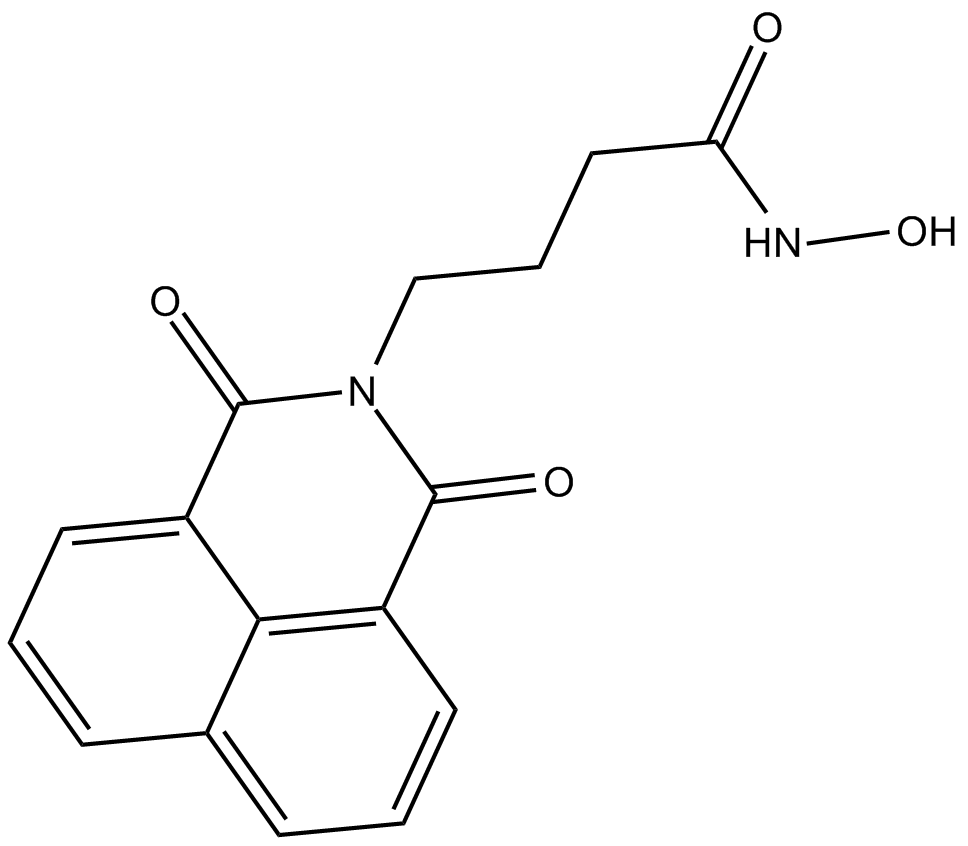
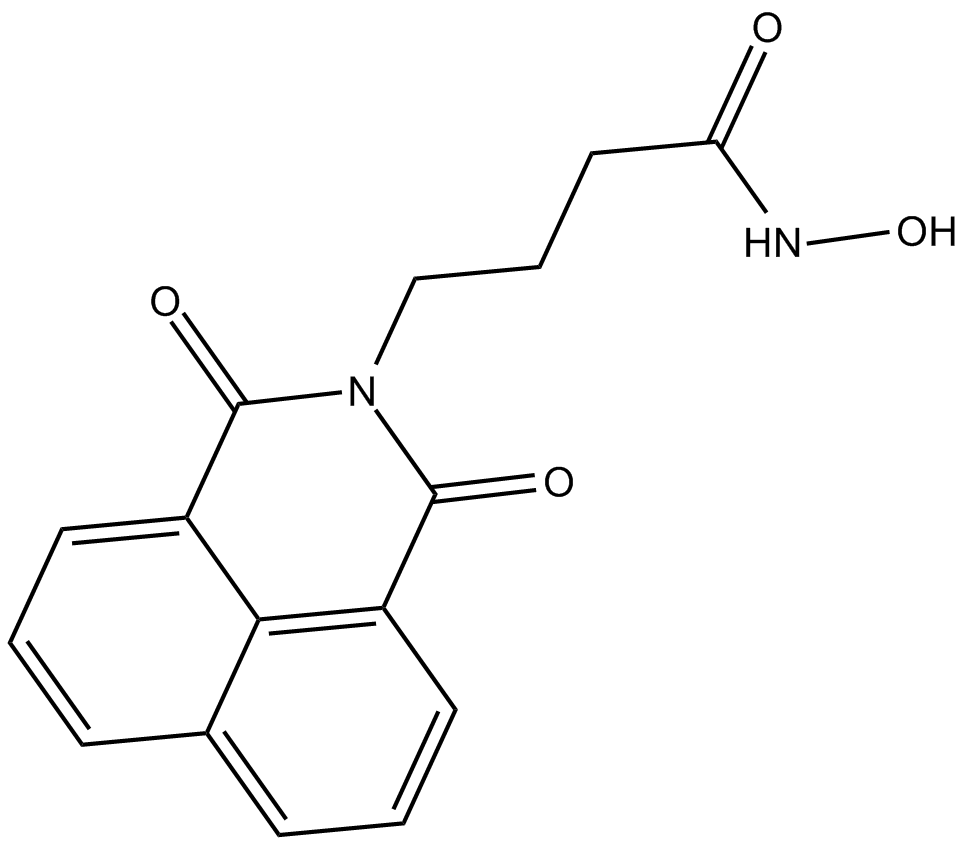
| Formula | C16H14N2O4 |
| Solubility | ≤2mg/ml in DMSO;2mg/ml in dimethyl formamide |
| Chemical Name | N-hydroxy-1,3-dioxo-1H-benz[de]isoquinoline-2(3H)-butanamide |
| SDF | Download SDF |
| Canonical SMILES | O=C(C1=CC=CC2=CC=CC3=C12)N(CCCC(NO)=O)C3=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构