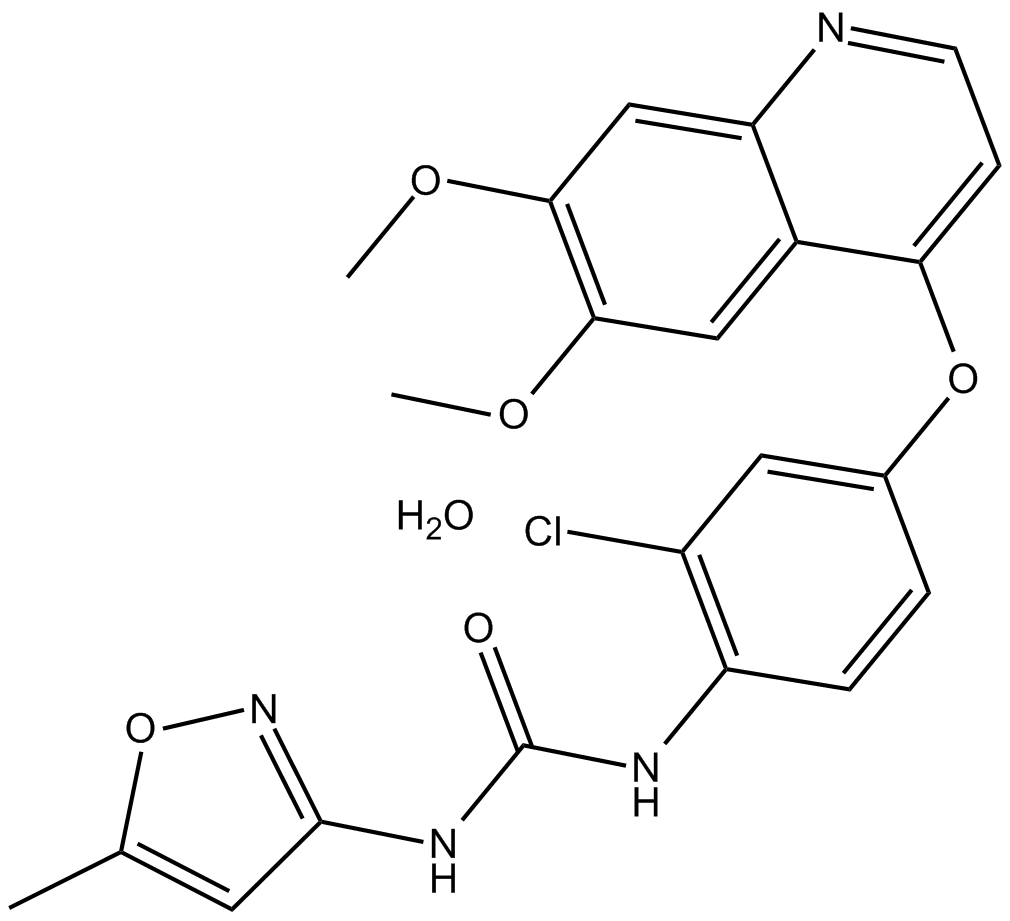
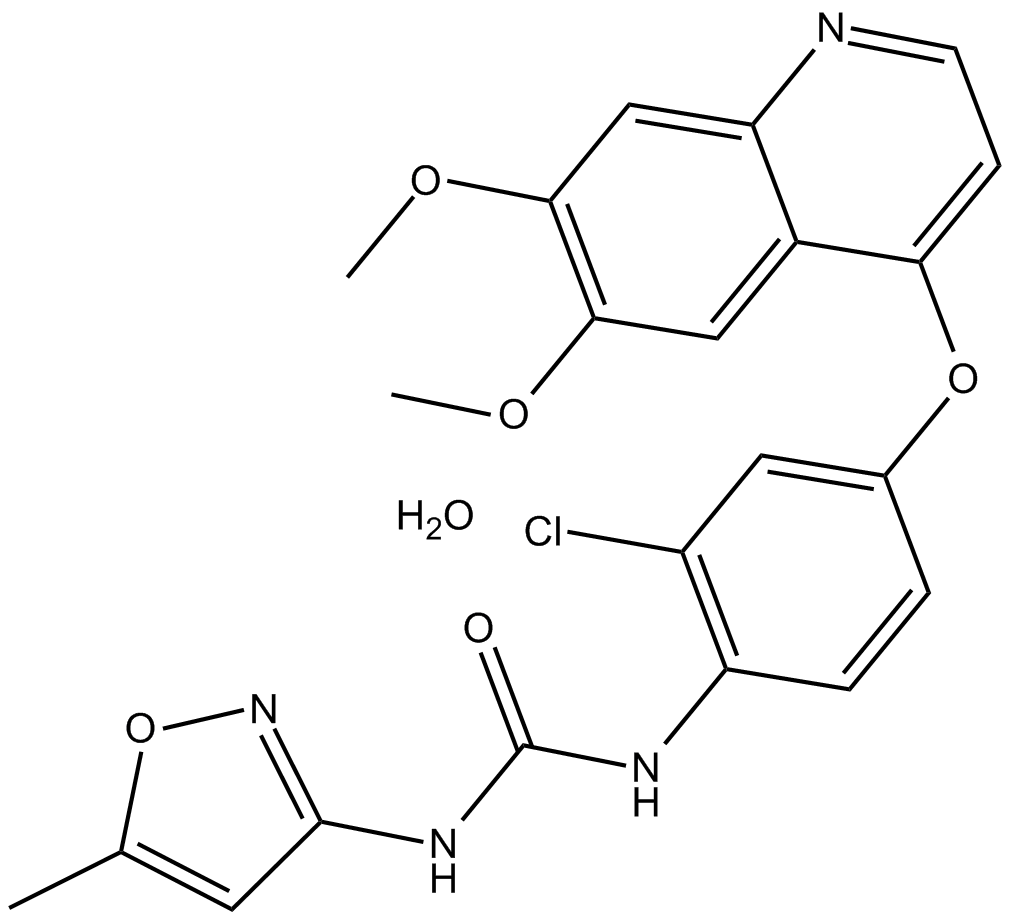
Tivozanib (hydrate)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Tivozanib, also known as AV-951 and KRN-951, is an orally active, ATP-competitive, small-molecule, quinoline-urea derivative. Tivozanib is a pan-VEGFR tyrosine kinase inhibitor.
In vitro: Tivozanib markedly inhibited the ligand-induced phosphorylation of VEGFR1\2 and 3 with the IC50 value of 30 nM\6.5 nM and 15 nM, respectively. Tivozanib also exihibited inhibitory effects on PDGFR and c-Ki with the IC50 value of 1.72 and 1.63 nmol/L, respectively. Tivozanib showed little activity against FGFR-1, Flt3, c-Met, EGFR and IGF-1R [1]. Tivozanib blocked VEGF-dependent activation of mitogen-activated protein kinases and proliferation of endothelial cells. It also inhibited VEGF-mediated migration of human umbilical vein endothelial cells [1].
In vivo: In tumor xenografts athymic rat model, p.o. administration of tivozanib decreased the micro vessel density and suppressed VEGFR2 phosphorylation levels, especially at a concentration of 1mg/kg. Tivozanib almost completely inhibited tumor xenografts growth (TGI > 85%) in athymic rats. Tivozanib displayed antitumor activity against various human tumor xenografts, such as lung, breast, colon, pancreas, ovarian and prostate cancer.[1]. In rat peritoneal disseminated tumor model, tivozanib prolonged the survival of the tumor-bearing rats with the MST of 53.5 days [2].
Clinical trials: Tivozanib has entered phase III clinical trials in patients with advanced renal cell carcinoma. Tivozanib improved progression-free survival (PFS), but not overall survival (OS). The most common adverse events were hypertension and dysphonia [3]. In patients with refractory, metastatic colorectal cancer, tivozanib has entered Multicenter phase II study [4].
References:
[1]. Nakamura K, Taguchi E, Miura T, et al. KRN951, a highly potent inhibitor of vascular endothelial growth factor receptor tyrosine kinases, has antitumor activities and affects functional vascular properties[J]. Cancer research, 2006, 66(18): 9134-9142.
[2]. Taguchi E, Nakamura K, Miura T, et al. Anti‐tumor activity and tumor vessel normalization by the vascular endothelial growth factor receptor tyrosine kinase inhibitor KRN951 in a rat peritoneal disseminated tumor model[J]. Cancer science, 2008, 99(3): 623-630.
[3]. Motzer R J, Nosov D, Eisen T, et al. Tivozanib versus sorafenib as initial targeted therapy for patients with metastatic renal cell carcinoma: results from a phase III trial[J]. Journal of clinical oncology, 2013, 31(30): 3791-3799.
[4]. Wolpin B M, Ng K, Zhu A X, et al. Multicenter phase II study of tivozanib (AV-951) and everolimus (RAD001) for patients with refractory, metastatic colorectal cancer[J]. The oncologist, 2013, 18(4): 377-378.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 472.9 |
| Cas No. | 682745-40-0 |
| Formula | C22H19ClN4O5·H2O |
| Synonyms | AV-951,KRN 951 |
| Solubility | ≤25mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | N-[2-chloro-4-[(6,7-dimethoxy-4-quinolinyl)oxy]phenyl]-N'-(5-methyl-3-isoxazolyl)-urea, monohydrate |
| SDF | Download SDF |
| Canonical SMILES | COC1=C(OC)C=C(C(OC2=CC(Cl)=C(NC(NC3=NOC(C)=C3)=O)C=C2)=CC=N4)C4=C1.O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构