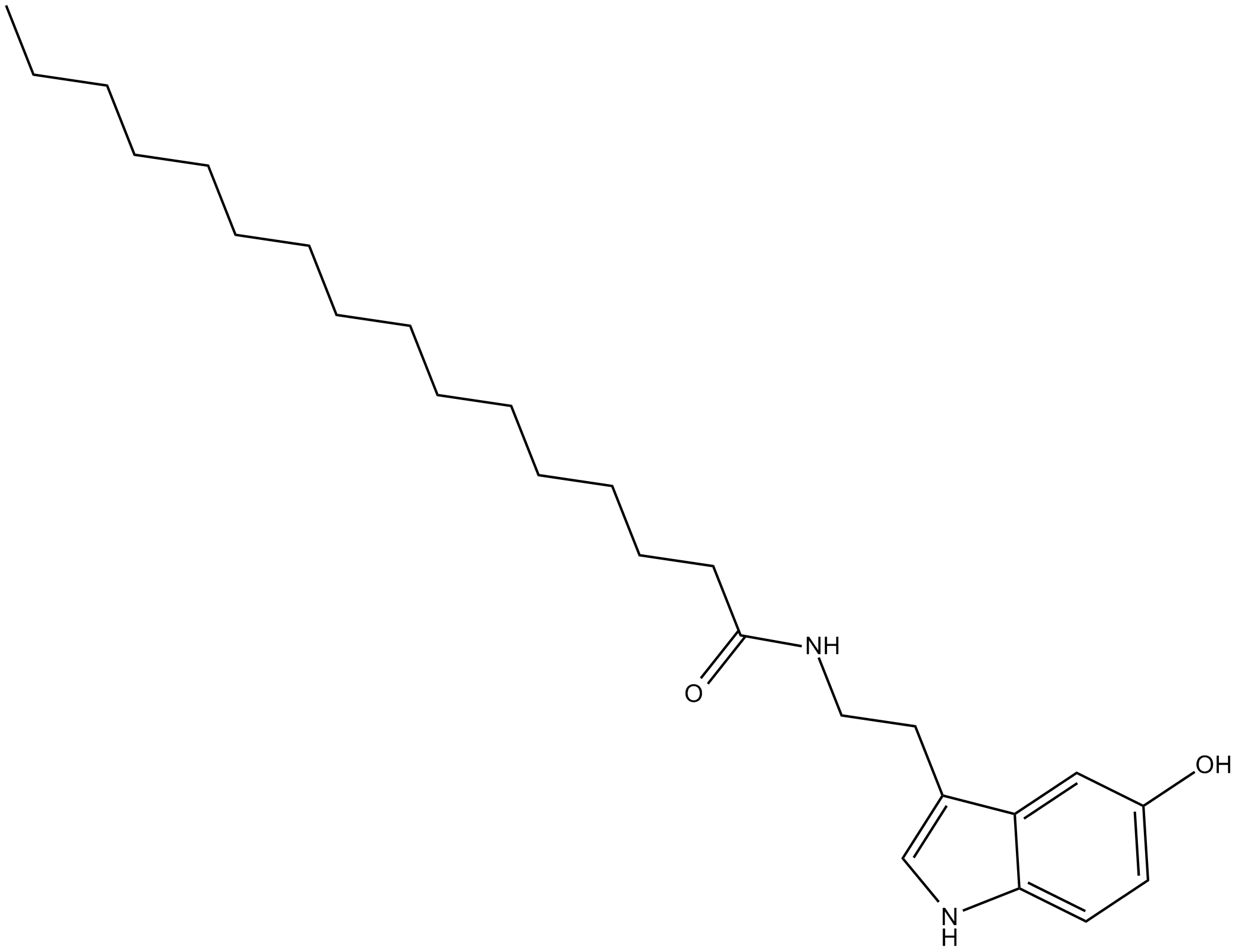
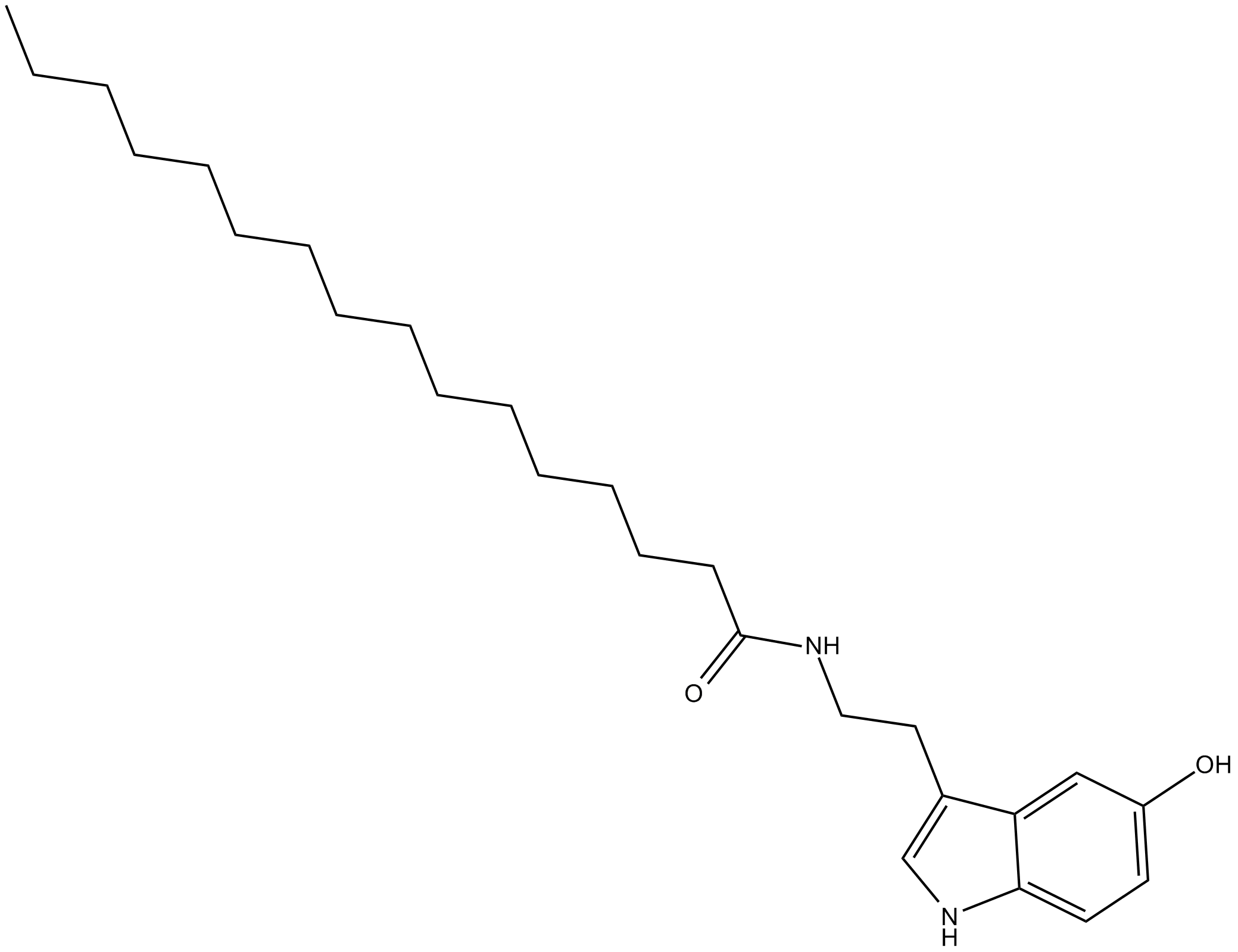
Palmitoyl Serotonin

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Palmitoyl Serotonin is a TRPV1 antagonist with IC50 value of 0.76 μM for human TRPV1 [1].
The transient receptor potential vanilloid-type 1 (TRPV1) channel is a nonselective cation channel that may be activated by a variety of exogenous and endogenous physical and chemical stimuli. TRPV1 is decreased in the injured nerve fibers but increased in those proximal to the site damage. TRPV1 is a potential new target for the development of analgesic and anti-inflammatory drugs [1].
Palmitoyl serotonin is a hybrid molecule patterned after arachidonoyl serotonin. Arachidonoyl serotonin is a dual antagonist of TRPV1 and fatty acid amide hydrolase (FAAH) with IC50 values of 0.27 and 8 μM, respectively. Arachidonoyl serotonin was highly effective against both acute and chronic peripheral pain [1][2]. In TRPV1 and FAAH assays, Palmitoyl serotonin inhibited anandamide hydrolysis mediated by FAAH and capsaicin-induced intracellular Ca2+ elevation in HEK293 cells overexpressing the human recombinant TRPV1 receptor with IC50 values of > 50 μM and 0.76 μM, respectively. However, the effects of replacing the arachidonoyl portion with the saturated 16-carbon palmitoyl moiety had not been studied [1].
References:
[1]. Ortar G, Cascio MG, De Petrocellis L, et al. New N-arachidonoylserotonin analogues with potential "dual" mechanism of action against pain. J Med Chem. 2007 Dec 27;50(26):6554-69.
[2]. Maione S, De Petrocellis L, de Novellis V, et al. Analgesic actions of N-arachidonoyl-serotonin, a fatty acid amide hydrolase inhibitor with antagonistic activity at vanilloid TRPV1 receptors. Br J Pharmacol. 2007 Mar;150(6):766-81.
| Physical Appearance | A neat solid |
| Storage | Store at -20°C |
| M.Wt | 414.6 |
| Cas No. | 212707-51-2 |
| Formula | C26H42N2O2 |
| Solubility | ≤30mg/ml in ethanol;15mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | N-[2-(5-hydroxy-1H-indol-3-yl)ethyl]-hexadecanamide |
| SDF | Download SDF |
| Canonical SMILES | CCCCCCCCCCCCCCCC(NCCC1=CNC2=CC=C(O)C=C12)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构