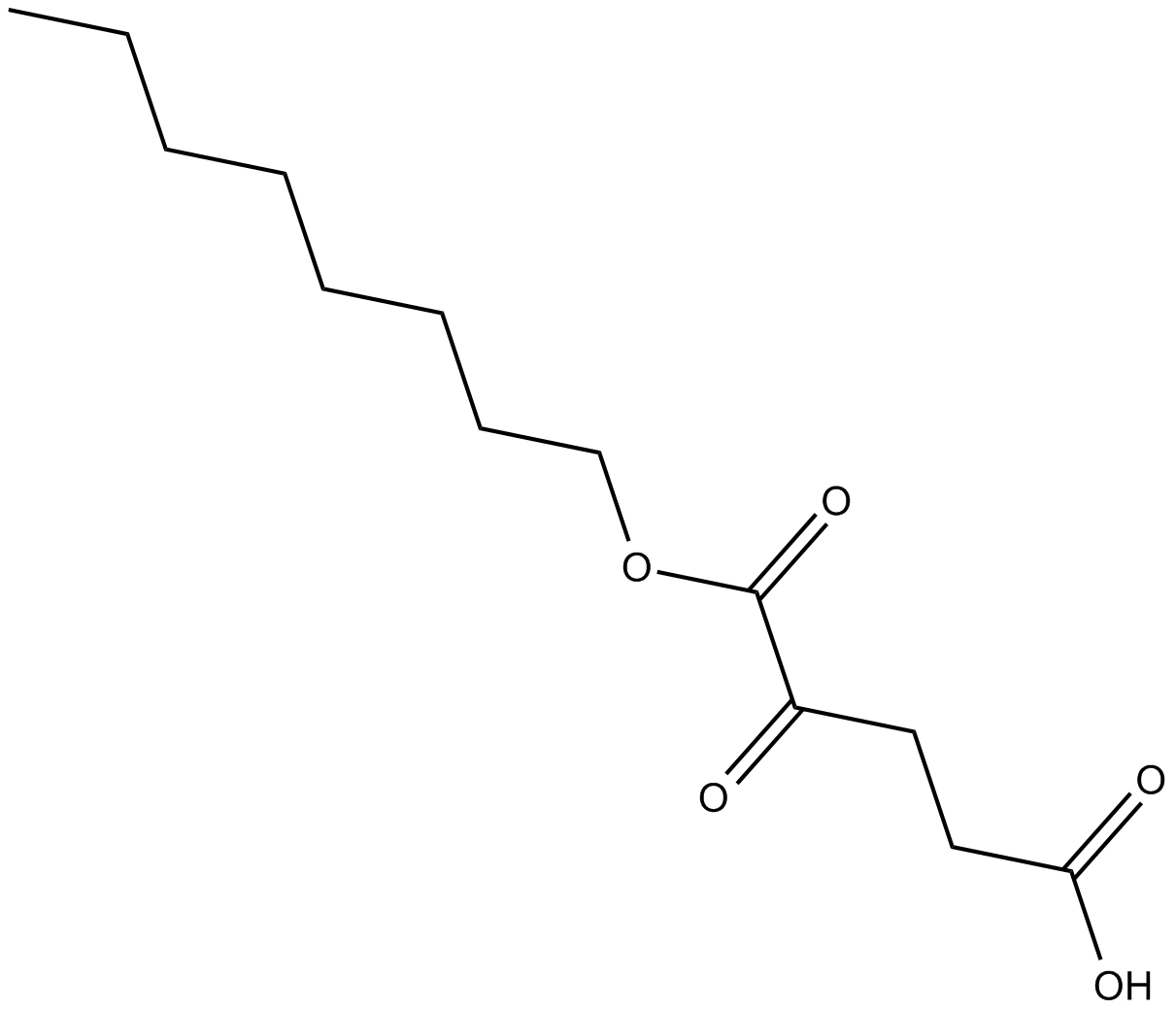
Octyl-α-ketoglutarate

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Octyl-α-ketoglutarate is a stable, cell-permeable form of α-ketoglutarate, a substrate of prolyl hydroxylases (PHD) [1][2].
The high turnover of HIFα is initiated by prolyl hydroxylases (PHD), which hydroxylate proline residues on the oxygen-dependent degradation (ODD) domain of HIFα, facilitating ubiquitination and degradation. To catalyze proline hydroxylation, PHDs convert molecular oxygen and α-ketoglutarate to carbon dioxide and succinate [2]. Loss of IDH1 activity would reduce cellular levels of α-KG [3].
Octyl-α-ketoglutarate is a cell-permeating α-ketoglutarate derivative, which built up rapidly and preferentially in cells with a dysfunctional TCA cycle. Octyl-α-ketoglutarate increased intracellular levels of freeα-ketoglutaric acid by approximately fourfold. In HEK293-derived cell lines expressing both a GFP-ODD fusion protein and HA-tagged pVHL, Octyl-α-ketoglutarate reactivated PHD activity inhibited by succinate or fumarate. Octyl-α-ketoglutarate restored hydroxylation and targeted HIF1 for ubiquitylation and proteasomally mediated degradation [2]. Octyl-α-ketoglutarate inhibited the HIF-1α induction caused by IDH1 knockdown in HeLa cells or overexpression of IDH1R132H mutant in U-87MG cells, suggesting a reduction in IDH1 activity caused a reduction in α-KG levels that in turn led to stabilization of HIF-1α [3].
References:
[1]. Gottlieb E, Tomlinson IP. Mitochondrial tumour suppressors: a genetic and biochemical update. Nat Rev Cancer. 2005 Nov;5(11):857-66.
[2]. MacKenzie ED, Selak MA, Tennant DA, et al. Cell-permeating alpha-ketoglutarate derivatives alleviate pseudohypoxia in succinate dehydrogenase-deficient cells. Mol Cell Biol. 2007 May;27(9):3282-9.
[3]. Zhao S, Lin Y, Xu W, et al. Glioma-derived mutations in IDH1 dominantly inhibit IDH1 catalytic activity and induce HIF-1alpha. Science. 2009 Apr 10;324(5924):261-5.
| Physical Appearance | A solution in acetate. To change the solvent, simply evaporate the acetate containing under a gentle stream of nitrogen and immediately add the solvent of choice. |
| Storage | Store at -20°C |
| M.Wt | 258.3 |
| Cas No. | 876150-14-0 |
| Formula | C13H22O5 |
| Synonyms | α-KG octyl ester |
| Solubility | ≤20mg/ml in ethanol;10mg/ml in DMSO;10mg/ml in dimethyl formamide |
| Chemical Name | 2-oxo-pentanedioic acid, 1-octyl ester |
| SDF | Download SDF |
| Canonical SMILES | OC(CCC(C(OCCCCCCCC)=O)=O)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |