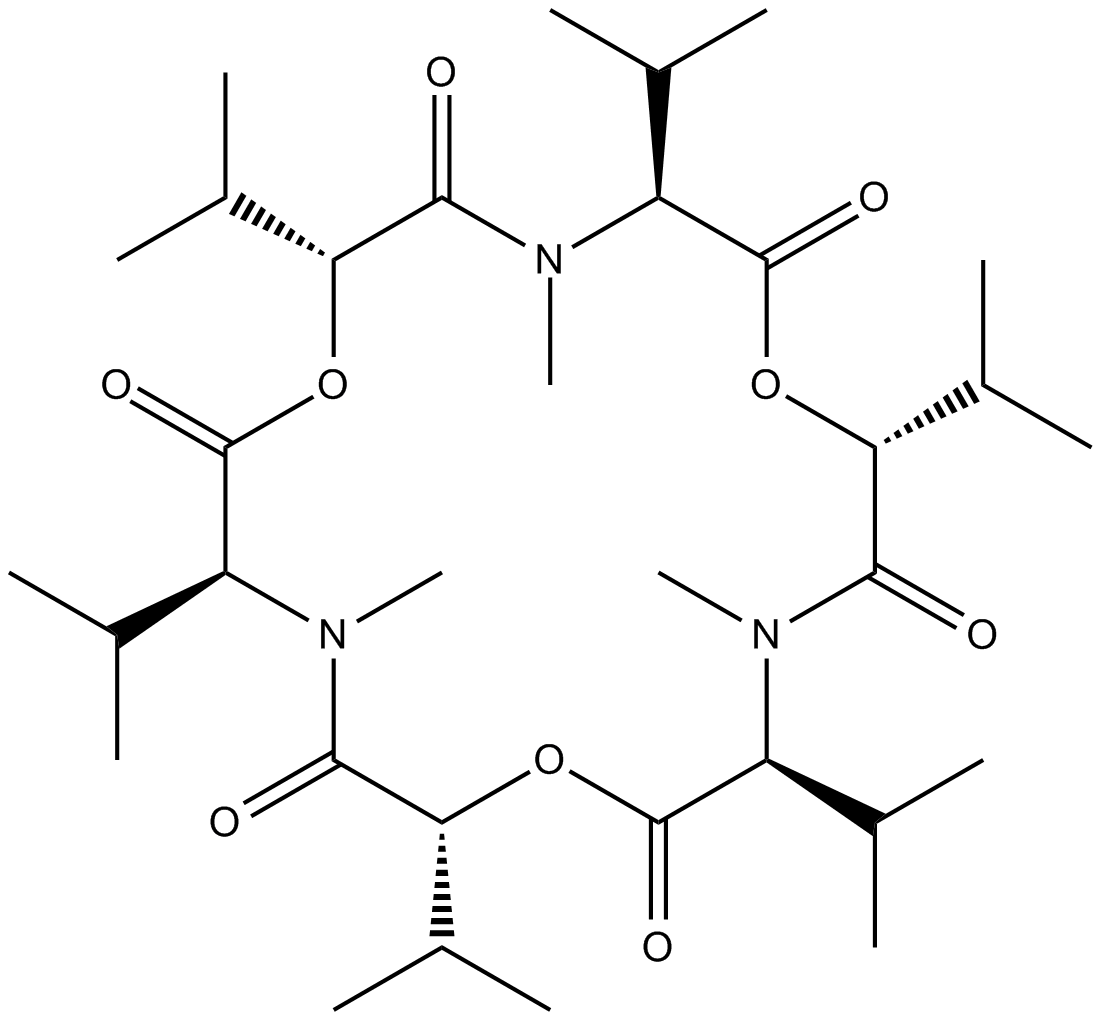
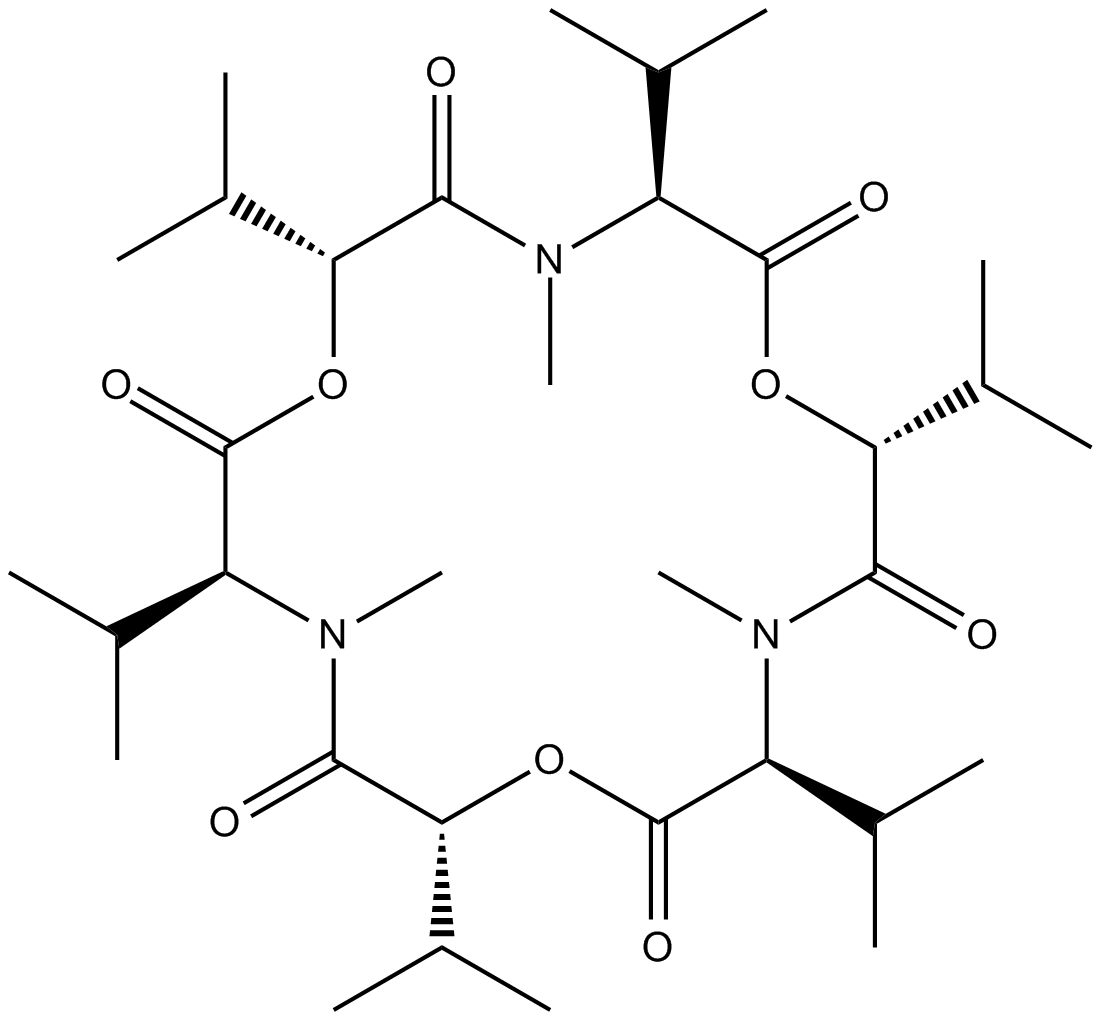
Enniatin B

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Enniatin B is an ionophore antibiotic.
Enniatins are one of the cyclohexadepsipeptides produced by various species of the genus Fusarium, and are reported to have antibiotic, ionophoric, and in-vitro hypolipidaemic activities.
In vitro: A non-toxic concentration of enniatin B could strongly inhibit a Pdr5p-mediated efflux of cycloheximide or cerulenin in Pdr5p-overexpressing cells. The mode of Pdr5p inhibition caused by enniatin B was competitive against FK506. However, enniatin B could not inhibit the function of Snq2p, a homologue of Pdr5p [1]. Another study showed that enniatin B was a relatively poor ionophore that could facilitate import of K+ and Na+ across membranes [2]. It was also found that like other enniatins, enniatin B was able to inhibit acyl-CoA: cholesterol acyltransferase [3].
In vivo: After oral administration to mice, no toxicological signs or pathological changes were observed. Moreover, enniatin B was found in all tissues and serum but not in urine, and the highest amounts was measured in liver and fat. Three phase I metabolites of enniatin B were found in liver and colon, with dioxygenated-enniatin B being most prominent [4].
Clinical trial: Up to now, enniatin B is still in the preclinical development stage.
References:
[1] K. Hiraga, S. Yamamoto, H. Fukuda, et al. Enniatin has a new function as an inhibitor of Pdr5p, one of the ABC transporters in Saccharomyces cerevisiae. Biochemical and Biophysical Research Communications 328(4), 1119-1125 (2005).
[2] M. R. Kamyar, P. Rawnduzi, C. R. Studenik, et al. Investigation of the electrophysiological properties of enniatins. Archives of Biochemistry and Biophysics429(2), 215-223 (2004).
[3] Tomoda, X. H. Huang, J. Cao, et al. Inhibition of acyl-CoA: Cholesterol acyltransferase activity by cyclodepsipeptide antibiotics. J.Antibiot.(Tokyo) 45(10),1626-1632 (1992).
[4] Rodríguez-Carrasco Y et al. Mouse tissue distribution and persistence of the food-born fusariotoxins Enniatin B and Beauvericin. Toxicol Lett. 2016 Apr 15;247:35-44.
| Physical Appearance | A powder |
| Storage | Store at -20°C |
| M.Wt | 639.8 |
| Cas No. | 917-13-5 |
| Formula | C33H57N3O9 |
| Synonyms | Antibiotic 86/88 |
| Solubility | Soluble in DMSO |
| Chemical Name | cyclo[(2R)-2-hydroxy-3-methylbutanoyl-N-methyl-L-valyl-(2R)-2-hydroxy-3-methylbutanoyl-N-methyl-L-valyl-(2R)-2-hydroxy-3-methylbutanoyl-N-methyl-L-valyl |
| SDF | Download SDF |
| Canonical SMILES | O=C(O[C@H](C(C)C)C(N([C@H](C(O[C@@H](C(N([C@H](C(O[C@@H]1C(C)C)=O)C(C)C)C)=O)C(C)C)=O)C(C)C)C)=O)[C@H](C(C)C)N(C)C1=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构