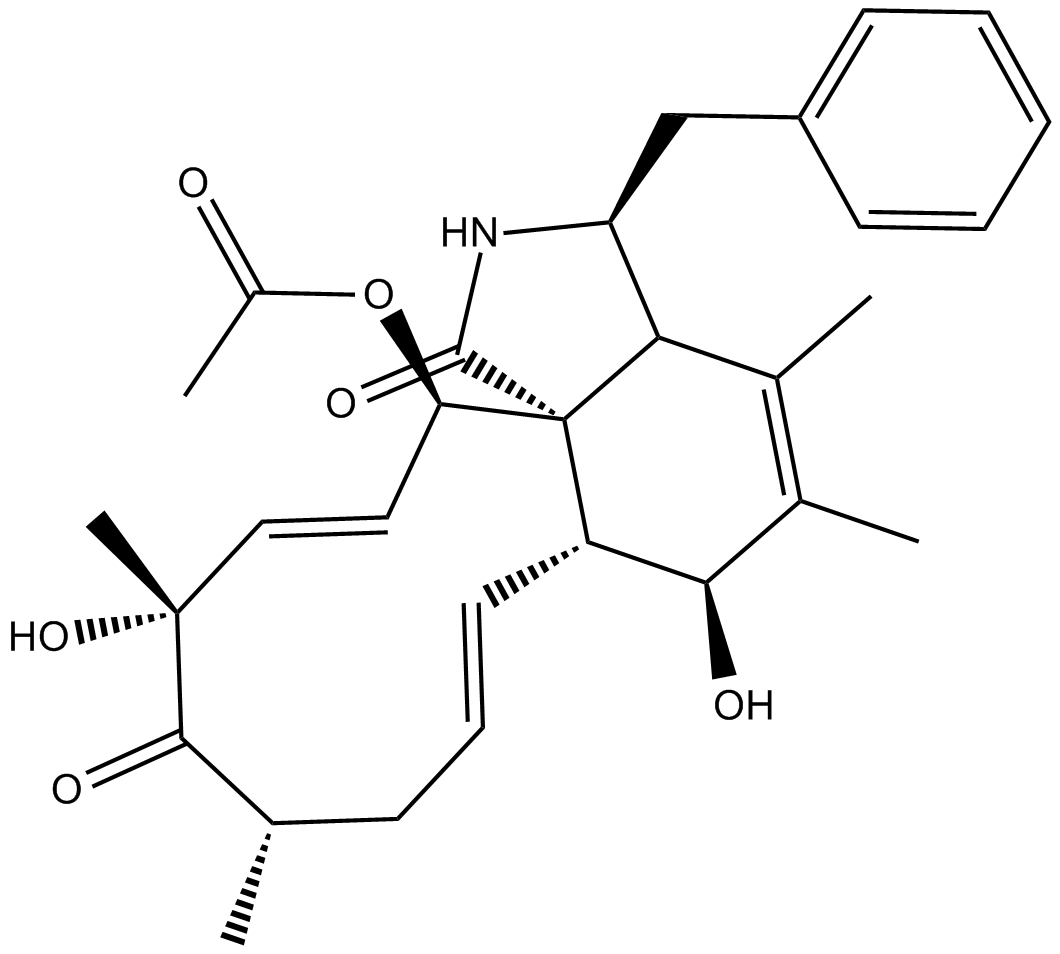
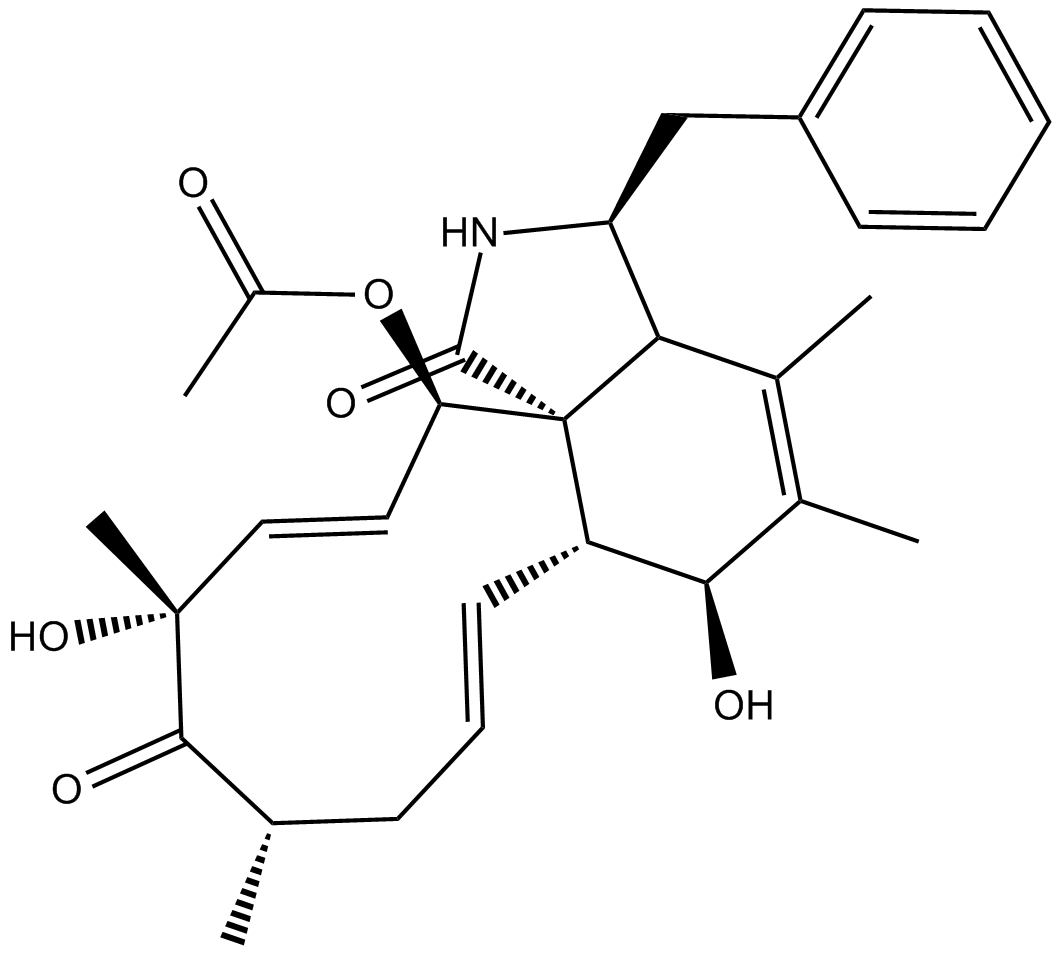
Cytochalasin C

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Cytochalasin C inhibits actin polymerization.
The cytochalasins are cell-permeable fungal metabolites inhibiting actin polymerization. This interferes with such diverse processes as cell growth, movement, phagocytosis, degranulation, as well as secretion.
In vitro: Previous study described the first results of actin assembly assays in the presence of the different cytochalasins. Acceleration of actin assembly in the presence of several cytochalasins was apparent from the more extensive assembly at shorter times. Cytochalasin C and its analogs, cytochalasin D, H, and J made up the same Group, which was characterized by fast assembly, so that the extent of assembly was reached before the first FPR data trace could be obtained, less than 4 min into the reaction. Thus, the members of this group including cytochalasin C were both strong accelerators of assembly and also inhibitors of steadystate extent of assembly.Moreover, cytochalasin C and D were found to be much stronger inhibitors than cytochalasin H and J, which were moderate and weak inhibitors, respectively. In addition, the effects of these cytochalasins on the diffusion coefficients of actin filaments at steady state was also have examined. Results showd that cytochalasin D and H had significantly higher diffusion coefficients. In contrast, cytochalasin C and cytochalasin A, B, J displayed a weak shortening activity [1].
In vivo: In zebrafish, cytochalasin D at 0.2 μM gave an approximate LD50, while cytochalasin B was fully tolerated at 5 μM, and gave an LD50 of 10 μM. Cytochalasin C was tolerated fully at 1 μM, which was ten-fold higher than the level for cytochalasin D that was tolerated [2].
Clinical trial: Up to now, there is no clinical data reported.
References:
[1] Walling, E. A.,Krafft, G.A. and Ware, B.R. Actin assembly activity of cytochalasins and cytochalasin analogs assayed using fluorescence photobleaching recovery. Archives of Biochemistry and Biophysics 264(1), 321-332 (1988).
[2] Trendowski M, Wong V, Wellington K, Hatfield S, Fondy TP. Tolerated doses in zebrafish of cytochalasins and jasplakinolide for comparison with tolerated doses in mice in the evaluation of pre-clinical activity of microfilament-directed agents in tumor model systems in vivo. In Vivo. 2014 Nov-Dec;28(6):1021-31.
| Physical Appearance | A white powder |
| Storage | Store at -20°C |
| M.Wt | 507.6 |
| Cas No. | 22144-76-9 |
| Formula | C30H37NO6 |
| Solubility | Soluble in ethanol;Soluble in methanol;Soluble in DMSO;Soluble in dimethyl formamide |
| Chemical Name | (3S,3aR,6S,6aR,7E,10S,12R,13E,15R,15aR)-15-(acetyloxy)-3,3a,6,6a,9,10,12,15-octahydro-6,12-dihydroxy-4,5,10,12-tetramethyl-3-(phenylmethyl)-1H-cycloundec[d]isoindole-1,11(2H)-dione |
| SDF | Download SDF |
| Canonical SMILES | O[C@H]1[C@H]2[C@@]3([C@@H](/C=C/[C@@](C)(O)C([C@@H](C)C/C=C/2)=O)OC(C)=O)C([C@H](CC4=CC=CC=C4)NC3=O)C(C)=C1C |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |