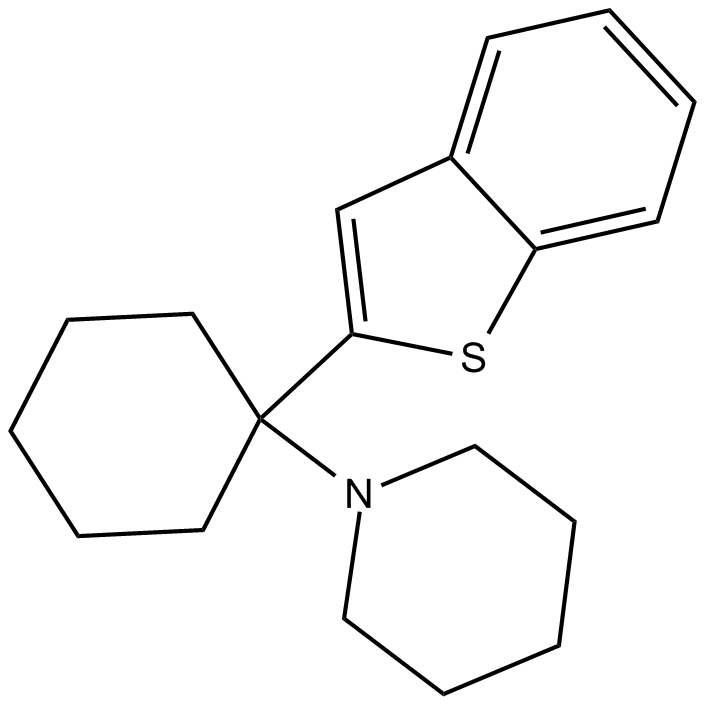
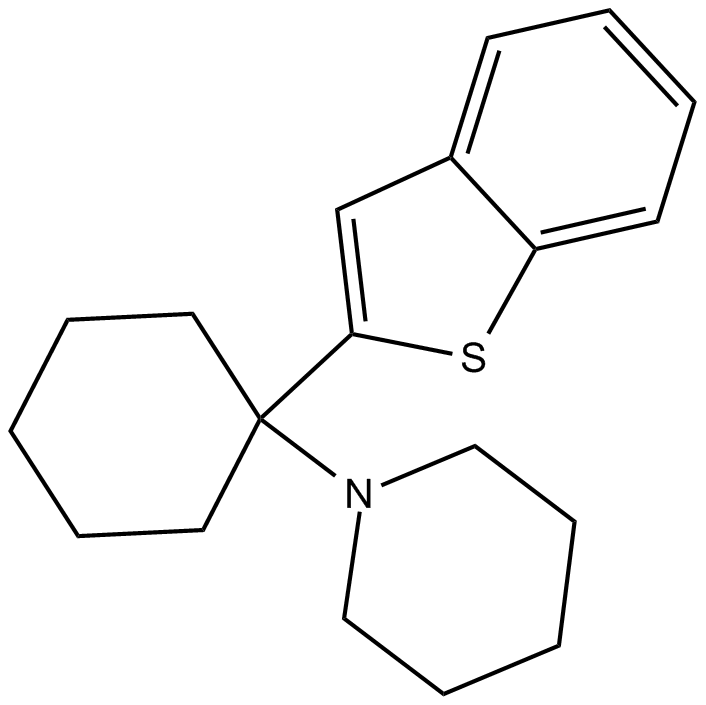
Benocyclidine

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Benocyclidine is an inhibitor of dopamine reuptake [1, 2].
Dopamine plays important roles in the brain and body. In the brain, dopamine functions as a neurotransmitter involved in reward-motivated behavior, motor control and controlling the release of various hormones. Dysfunctions of the dopamine system have been associated several important diseases including Parkinson's disease, attention deficit hyperactivity disorder, schizophrenia, and addiction [3]. Dopamine also functions in blood vessels, kidneys, pancreas, the digestive system, the immune system, and the blood vessels.
Benocyclidine (BTCP) was a derivative of phencyclidine (PCP) with a benzothiophenyl group instead of a phenyl ring. BTCP potently inhibited dopamine (DA) uptake with the IC50 of 7-8 nM. BTCP showed low affinity for the PCP receptor with the IC50 of 6 μM [1, 2]. In the striatum, BTCP binding was dose-dependently inhibited by unlabeled BTCP and nomifensine with the ID50 of 6.34 mg/kg and 11.06 mg/kg, respectively. BTCP bound to the dopamine uptake complex in the mouse brain in vivo [4].
References:
[1] Vignon J, Pinet V, Cerruti C, et al. [3H] N-[1-(2-Benzo (b) thiophenyl) cycohexyl] piperidine ([3H] BTCP): a new phencyclidine analog selective for the dopamine uptake complex[J]. European journal of pharmacology, 1988, 148(3): 427-436.
[2] Chaudieu I, Vignon J, Chicheportiche M, et al. Role of the aromatic group in the inhibition of phencyclidine binding and dopamine uptake by PCP analogs[J]. Pharmacology Biochemistry and Behavior, 1989, 32(3): 699-705.
[3] Seeman P. Brain dopamine receptors[J]. Pharmacological Reviews, 1980, 32(3): 229-313.
[4] Maurice T, Vignon J, Kamenka J M, et al. In vivo labelling of the mouse dopamine uptake complex with the phencyclidine derivative [3 H] BTCP[J]. Neuroscience letters, 1989, 101(2): 234-238.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 299.47 |
| Cas No. | 112726-66-6 |
| Formula | C19H25NS |
| Solubility | Soluble in DMSO |
| Chemical Name | 1-(1-(benzo[b]thiophen-2-yl)cyclohexyl)piperidine |
| SDF | Download SDF |
| Canonical SMILES | C1CCC(CC1)(C2=CC3=CC=CC=C3S2)N4CCCCC4 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |