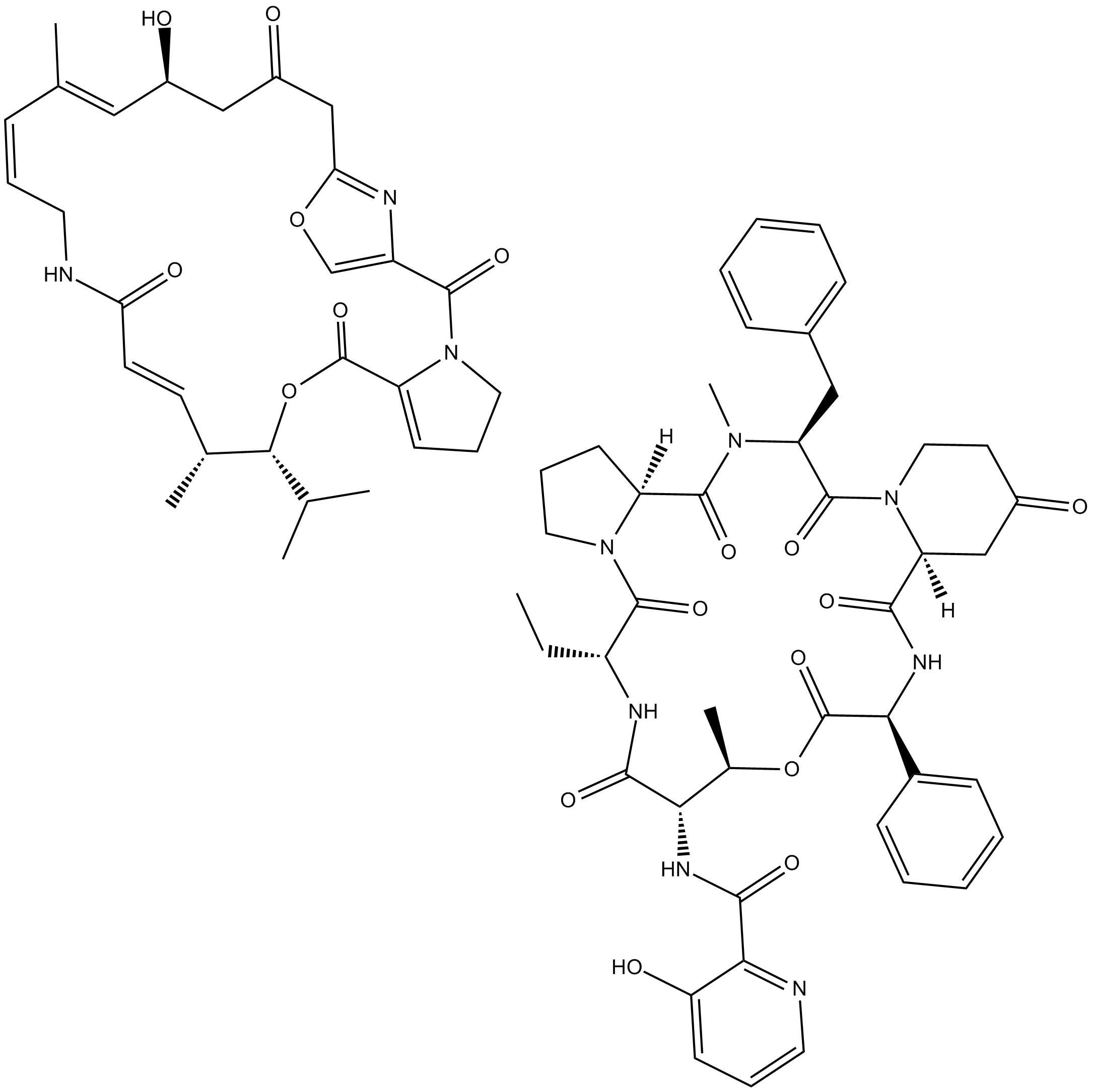
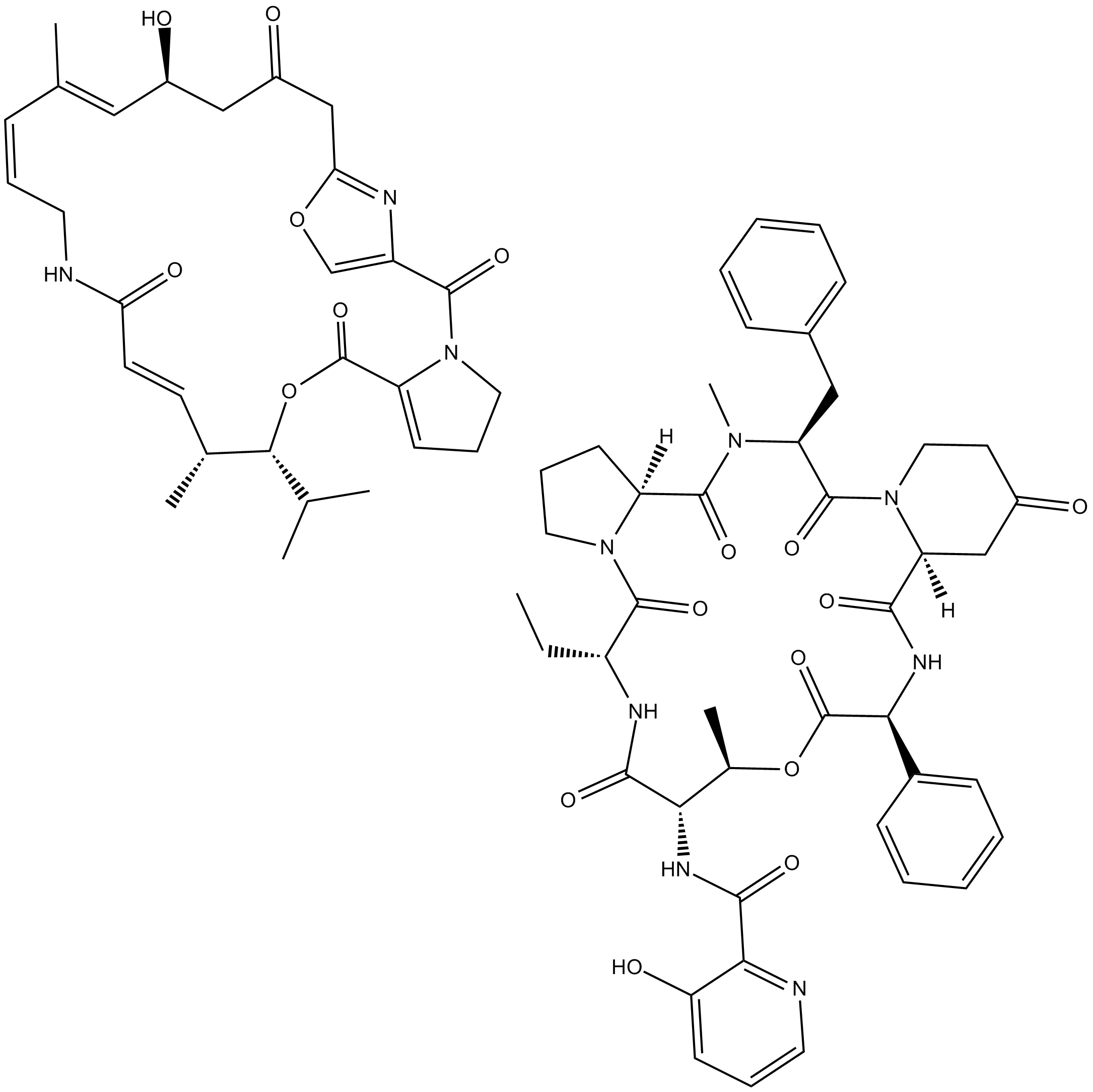
Virginiamycin Complex

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Virginiamycin Complex is a a complex containing two streptogramin antibiotics.
Virginiamycin complex contains two streptogramin antibiotics, virginiamycin M1 (75%) and virginiamycin S1 (25%), produced by S. virginiae.
In vitro: Previous study found that the M1 and S1 components of virginiamycin (VM and VS) could inhibit protein synthesis in bacteria--reversibly when a single component was present and irreversibly when both were present. In cell-free systems, each factor bound to the large ribosomal subunit, and the affinity of ribosomes for VS was enhanced in the presence of VM. In addition, the binding of labeled VM to ribosomes yielded particles unable to perform poly(U)-directed polyphenylalanine synthesis. Moreover, the association constant for the binding of VS to these particles was equal to that incubated with a mixture of VM and VS [1].
In vivo: A radiochemical method was developed to estimate cholyltaurine hydrolase potentials and rates of cholyltaurine hydrolysis in chicken intestinal homogenates. This method was used to monitor the effects of antibiotic feed additives on cholyltaurine hydrolase activity. Results showed that virginiamycin could improve the rate of weight gain and feed conversion and decrease cholyltaurine hydrolase activity in ileal homogenates relative [2].
Clinical trial: So far, no clinical study has been conducted.
References:
1. Parfait, R., and Cocito, C. Lasting damage to bacterial ribosomes by reversibly bound virginiamycin M. Proceedings of the National Academy of Sciences of the United States of America 77(9), 5492-5496 (1980).
2. Feighner, S.D., and Dashkevicz, M.P. Subtherapeutic levels of antibiotics in poultry feeds and their effects on weight gain, feed efficiency, and bacterial cholyltaurine hydrolase activity. Applied and Environmental Microbiology 53(2), 331-336 (1987).
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 1396.6 |
| Cas No. | 11006-76-1 |
| Formula | C43H49N7O10·C28H35N3O10 |
| Synonyms | Mikamycin,RP 7293,Stapyocine,Streptogramin |
| Solubility | ≤15mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | N-((6R,9S,10R,13S,15aS,22S,24aS)-22-benzyl-6-ethyl-10,23-dimethyl-5,8,12,15,17,21,24-heptaoxo-13-phenyldocosahydro-12H-pyrido[2,1-f]pyrrolo[2,1-l][1]oxa[4,7,10,13,16]pentaazacyclononadecin-9-yl)-3-hydroxypicolinamide compound with (12Z,6R,7R,8E,13Z,15E,17 |
| SDF | Download SDF |
| Canonical SMILES | O=C1CCN(C([C@@H](N(C)C([C@@]23[H])=O)CC4=CC=CC=C4)=O)[C@@](C(N[C@@H](C5=CC=CC=C5)C(O[C@H](C)[C@H](NC(C6=NC=CC=C6O)=O)C(N[C@H](CC)C(N2CCC3)=O)=O)=O)=O)([H])C1.O=C(/C=C/[C@@H](C)[C@@H](C(C)C)OC(C7=CCCN7C(C8=COC(C9)=N8)=O)=O)NC/C=C\C(C)=C\[C@@H](O)CC9=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |