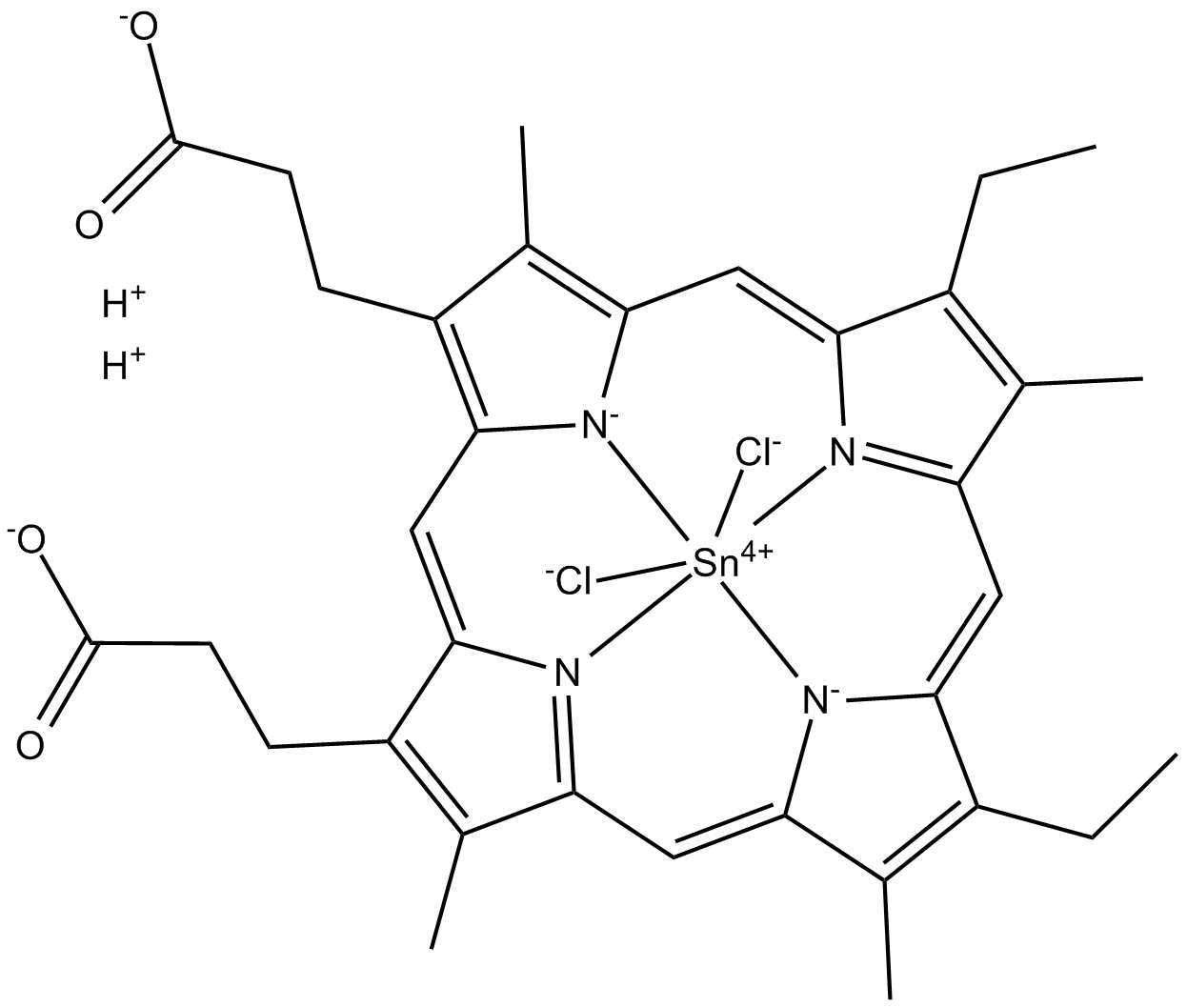
Tin Mesoporphyrin IX (chloride)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Ki = 14 nM
Tin Mesoporphyrin IX is a potent and competitive inhibitor of heme oxygenase (HO) activity.
Heme oxygenase or haem oxygenase (HO) is an enzyme catalyzing the degradation of heme. This produces biliverdin, ferrous iron, and carbon monoxide. There is limited evidence that levels of heme oxygenase are positive predictors of metabolic disease, insulin resistance, and metaflammation.
In vitro: Previous study found that Tin Mesoporphyrin IX was a potent competitive in-vitro inhibitor of enzyme activity when it was incubated with rat splenic microsomal heme oxygenase, with a Ki of 0.014 microM [1].
In vivo: Animal study showed that Tin Mesoporphyrin IX at 1 pmol/kg body wt could inhibit hepatic, renal, and splenic heme oxygenase activity in adult animals for extended periods of time. Tin Mesoporphyrin IX at 1 pmol/kg body wt also prevented the transient increase in serum bilirubin 24 h after birth in the rat neonate and significantly reduced the levels of serum bilirubin in aminolevulinic acid induced hyperbilirubinemia in the 7-day-old suckling neonate. Moreover, it was found that the tissue heme oxygenase activity decreased in both animal models of hyperbilirubinemia. Tin Mesoporphyrin IX treatment resulted in a prolonged increase in the heme saturation of hepatic tryptophan pyrrolase [1].
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Drummond, G. S.,Galbraith, R.A.,Sardana, M.K., et al. Reduction of the C2 and C4 vinyl groups of Sn-protoporphyrin to form Sn-mesoporphyrin markedly enhances the ability of the metalloporphyrin to inhibit in vivo heme catabolism. Archives of Biochemistry and Biophysics 255(1), 64-74 (1987).
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 754.3 |
| Cas No. | 106344-20-1 |
| Formula | C34H34Cl2N4O4Sn·2H |
| Synonyms | NSC 267099,SnMP |
| Solubility | ≤0.5mg/ml in DMSO;1mg/ml in dimethyl formamide |
| Chemical Name | (OC-6-13)-dichloro[7,12-diethyl-3,8,13,17-tetramethyl-21H,23H-porphine-2,18-dipropanoato(4-)-κN21,κN22,κN23,κN24]-stannate(2-), dihydrogen |
| SDF | Download SDF |
| Canonical SMILES | [Cl-][Sn+4]123([N-]4C5=C(CCC([O-])=O)C(C)=C4C=C(C(CC)=C6C)[N]1=C6C=C(C(CC)=C7C)[N-]2C7=CC8=[N]3C(C(CCC([O-])=O)=C8C)=C5)[Cl-].[H+].[H+] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |