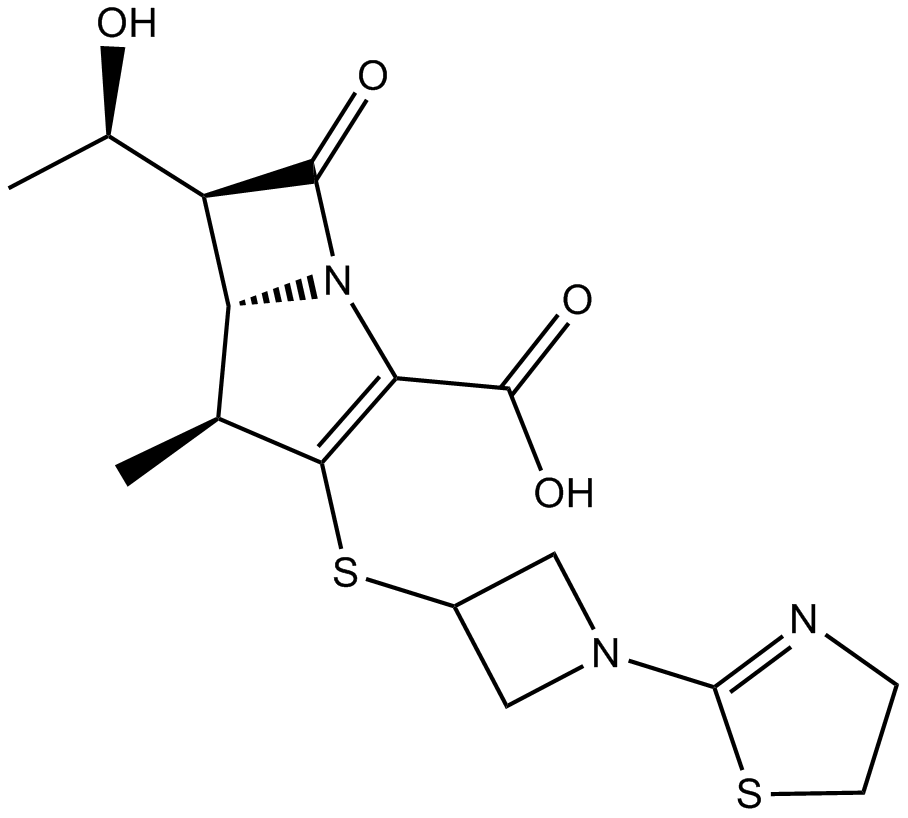
Tebipenem

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Tebipenem is an orally available carbapenem antibiotic. Tebipenem is active against a panel of clinical isolates from a variety of bacterial species (MIC50s ≤ 0.0039 ~ 8 µg/ml), including methicillin-resistant strains of Staphylococcus aureus (S. aureus) and Staphylococcus epidermidis (S. epidermidis), as well as penicillin-resistant Streptococcus pneumoniae (S. pneumonia). Tebipenem also inhibits β-lactamase in a time- and concentration-dependent manner. Tebipenem pivoxil, a derivative of tebipenem, has been under development as the first orally available carbapenem antibiotic for the treatment of respiratory and otolaryngological infections caused by drug-resistant S. pneumonia in pediatric patients.
References:
1. Hazra S, Xu H, Blanchard JS. Tebipenem, a new carbapenem antibiotic, is a slow substrate that inhibits the β-lactamase from Mycobacterium tuberculosis. Biochemistry, 2014, 53(22): 3671-3678.
2. Fujimoto K, Takemoto K, Hatano K, et al. Novel carbapenem antibiotics for parenteral and oral applications: in vitro and in vivo activities of 2-aryl carbapenems and their pharmacokinetics in laboratory animals. Antimicrobial Agents and Chemotherapy, 2013, 57(2): 697-707.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 383.49 |
| Cas No. | 161715-21-5 |
| Formula | C16H21N3O4S2 |
| Solubility | insoluble in EtOH; ≥19.15 mg/mL in H2O with gentle warming; ≥24.9 mg/mL in DMSO |
| Chemical Name | (4R,5S,6S)-3-[1-(4,5-dihydro-1,3-thiazol-2-yl)azetidin-3-yl]sulfanyl-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | CC1C2C(C(=O)N2C(=C1SC3CN(C3)C4=NCCS4)C(=O)O)C(C)O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Animal experiment:[2] | |
|
Animal models |
A mouse model of penicillin-resistant S. pneumoniae infection |
|
Dosage form |
0.32 ~ 3.2 mg/kg Administered intravenously thrice daily 1 day and 2 days after infection |
|
Applications |
Tebipenem dose-dependently decreased the number of colony forming units (CFUs) in the lungs of infected mice. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Hazra S, Xu H, Blanchard JS. Tebipenem, a new carbapenem antibiotic, is a slow substrate that inhibits the β-lactamase from Mycobacterium tuberculosis. Biochemistry, 2014, 53(22): 3671-3678. 2. Fujimoto K, Takemoto K, Hatano K, et al. Novel carbapenem antibiotics for parenteral and oral applications: in vitro and in vivo activities of 2-aryl carbapenems and their pharmacokinetics in laboratory animals. Antimicrobial Agents and Chemotherapy, 2013, 57(2): 697-707. |
|
质量控制和MSDS
- 批次:
化学结构