PPT

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
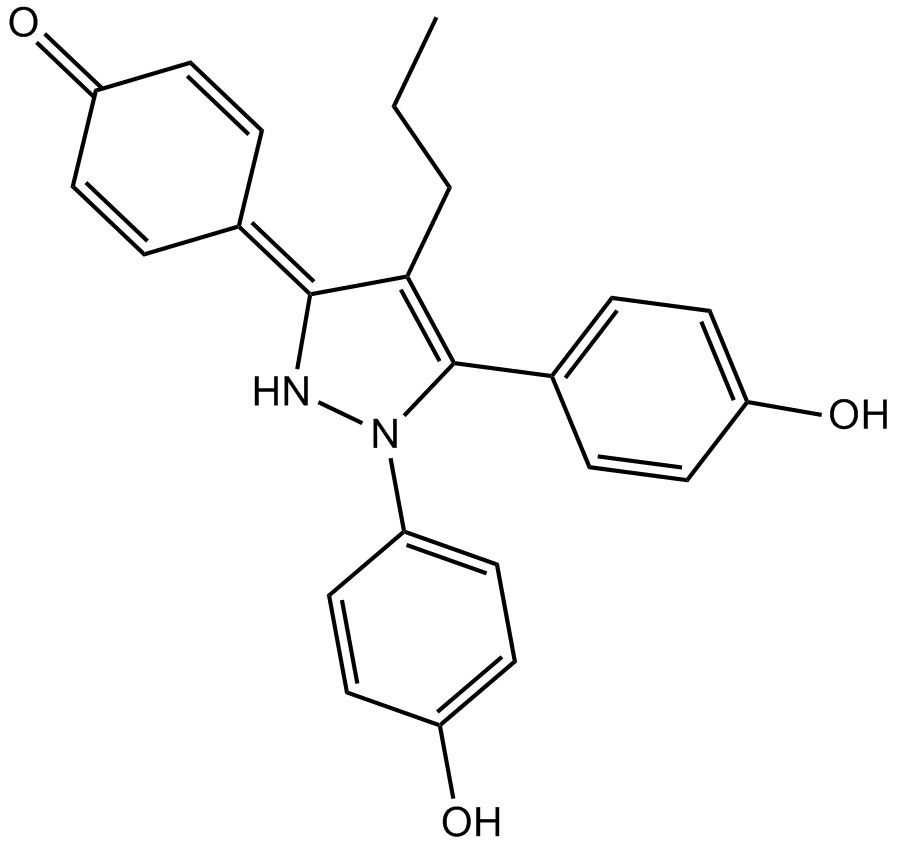
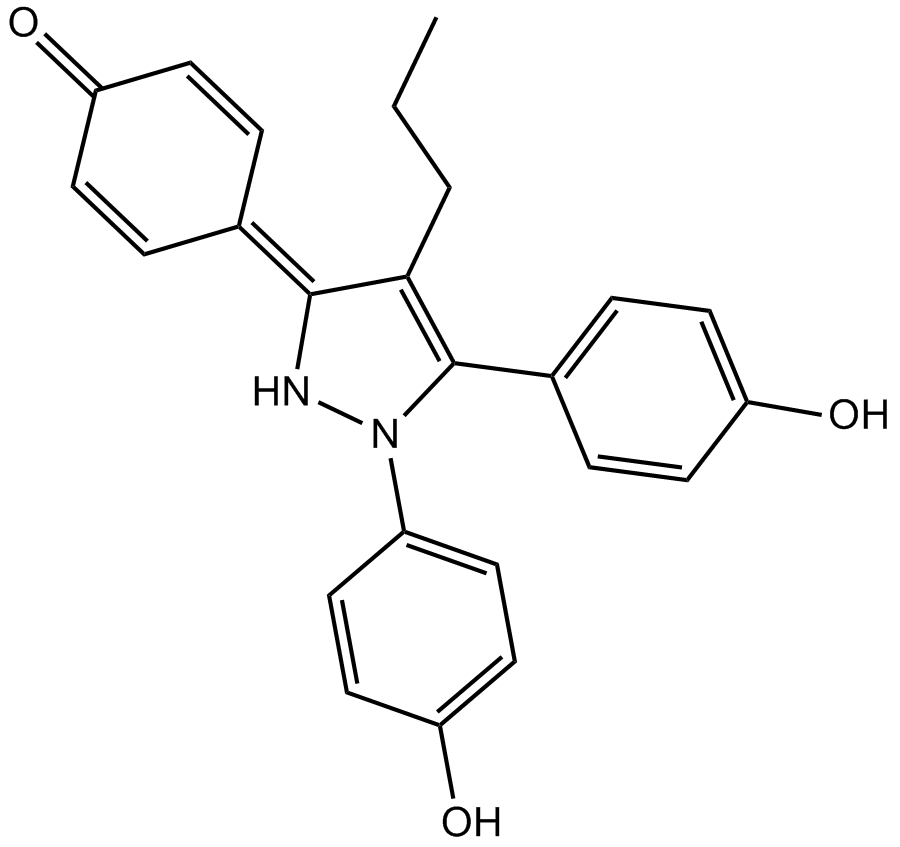
PPT, abbreviated from propyl pyrazole triol, is a potent, selective agonist of estrogen receptor α (ERα), with a reported 410-fold selectivity for ERα over ERβ. ERα is one of two main types of estrogen receptor, a nuclear receptor that is activated by the sex hormone estrogen and involved in normal developmental, physiological, and reproductive processes in vertebrates. Compared with ERβ, ERα is encoded by a distinct gene, and differs in its relative and absolute tissue distribution. Since PPT exhibits subtype-selective property for estrogen receptors, it may serve as a useful tool for exploring how estrogens work through different ER subtypes.
References:
1. Sotoca AM, van den Berg H, Vervoort J, et al. Influence of cellular ERα/ERβ ratio on the ERα-agonist induced proliferation of human T47D breast cancer cells. Toxicological Sciences, 2008, 105(2): 303-311.
2. Stauffer SR, Coletta CJ, Tedesco R, et al. Pyrazole ligands: structure-affinity/activity relationships and estrogen receptor-alpha-selective agonists. Journal of Medicinal Chemistry, 2000, 43(26): 4934-4947.
3. Harris HA, Katzenellenbogen JA, Katzenellenbogen BS. Characterization of the biological roles of the estrogen receptors, ERα and ERβ, in estrogen target tissues in vivo through the use of an ERα-selective ligand. Endocrinology, 2002, 143(11): 4172-4177.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 386.45 |
| Cas No. | 263717-53-9 |
| Formula | C24H22N2O3 |
| Solubility | ≥95.4 mg/mL in DMSO,≥48.9 mg/mL in EtOH,insoluble in H2O |
| Chemical Name | 4-(1,5-bis(4-hydroxyphenyl)-4-propyl-1H-pyrazol-3(2H)-ylidene)cyclohexa-2,5-dienone |
| SDF | Download SDF |
| Canonical SMILES | OC1=CC=C(C=C1)C(N(C(C=C2)=CC=C2O)N/3)=C(CCC)C3=C(C=C4)/C=CC4=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| Cell experiment:[3] | |
|
Cell lines |
Saos-2 cells expressing either human ERα or ERβ |
|
Reaction Conditions |
1 μM PPT for 24 h incubation |
|
Applications |
PPT did not up-regulate metallothionein-II mRNA which is regulated only by ERβ, and only up-regulated IGFBP-4 mRNA (regulated by both ERα and ERβ) in Saos-2 cells expressing ERα. |
| Animal experiment:[3] | |
|
Animal models |
Sexually immature Sprague Dawley rats |
|
Dosage form |
5 ~ 1000 μg/rat Once daily by subcutaneous injection for 3 days |
|
Applications |
In a short-term (4 d) uterotrophic assay, PPT was found to be as efficacious as 17α-ethinyl-17β-estradiol in stimulating uterine weight gain and up-regulating complement 3 gene expression. |
|
Note |
The technical data provided above is for reference only. |
|
References: 1. Sotoca AM, van den Berg H, Vervoort J, et al. Influence of cellular ERα/ERβ ratio on the ERα-agonist induced proliferation of human T47D breast cancer cells. Toxicological Sciences, 2008, 105(2): 303-311. 2. Stauffer SR, Coletta CJ, Tedesco R, et al. Pyrazole ligands: structure-affinity/activity relationships and estrogen receptor-alpha-selective agonists. Journal of Medicinal Chemistry, 2000, 43(26): 4934-4947. 3. Harris HA, Katzenellenbogen JA, Katzenellenbogen BS. Characterization of the biological roles of the estrogen receptors, ERα and ERβ, in estrogen target tissues in vivo through the use of an ERα-selective ligand. Endocrinology, 2002, 143(11): 4172-4177. |
|
质量控制和MSDS
- 批次:
化学结构