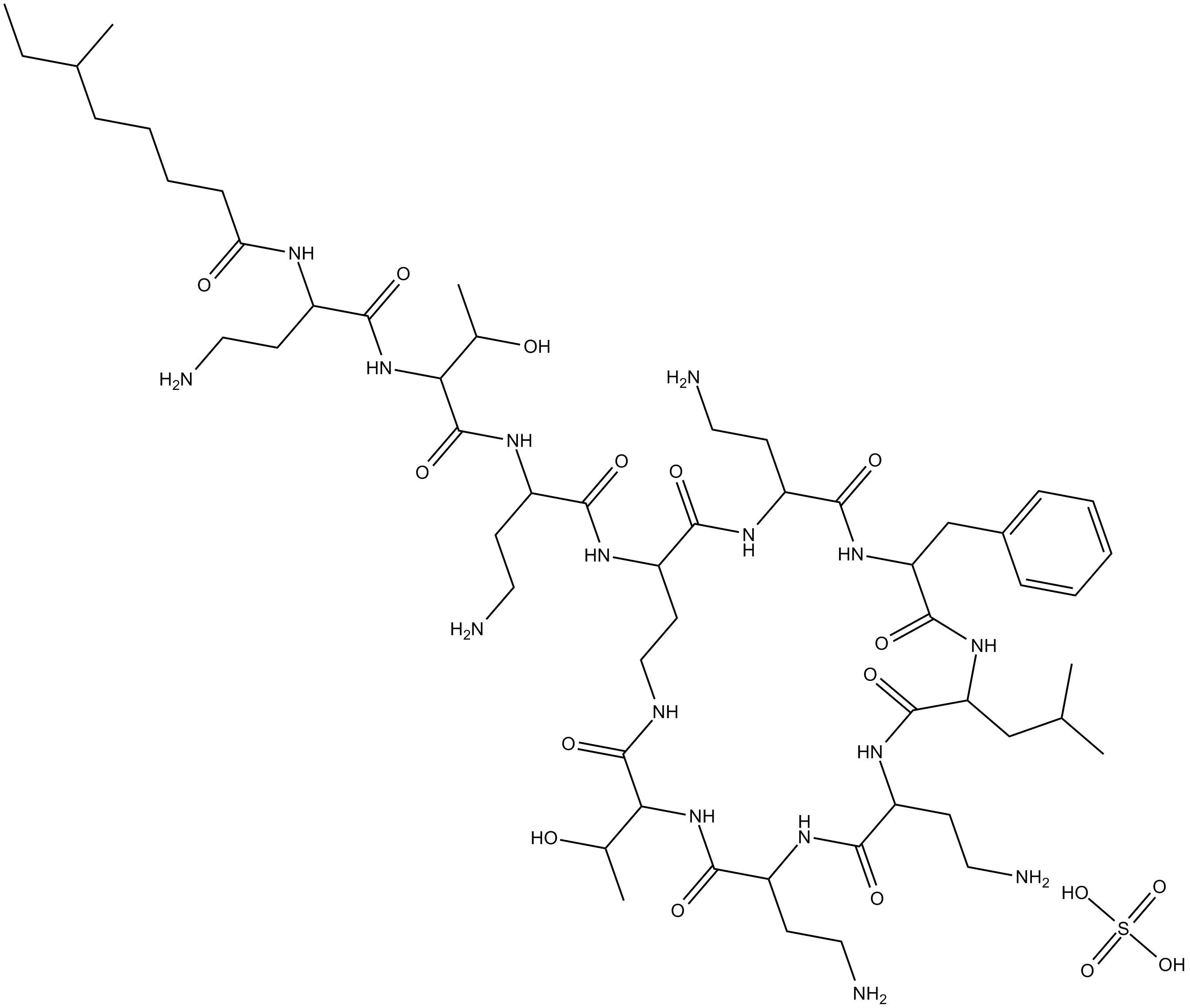
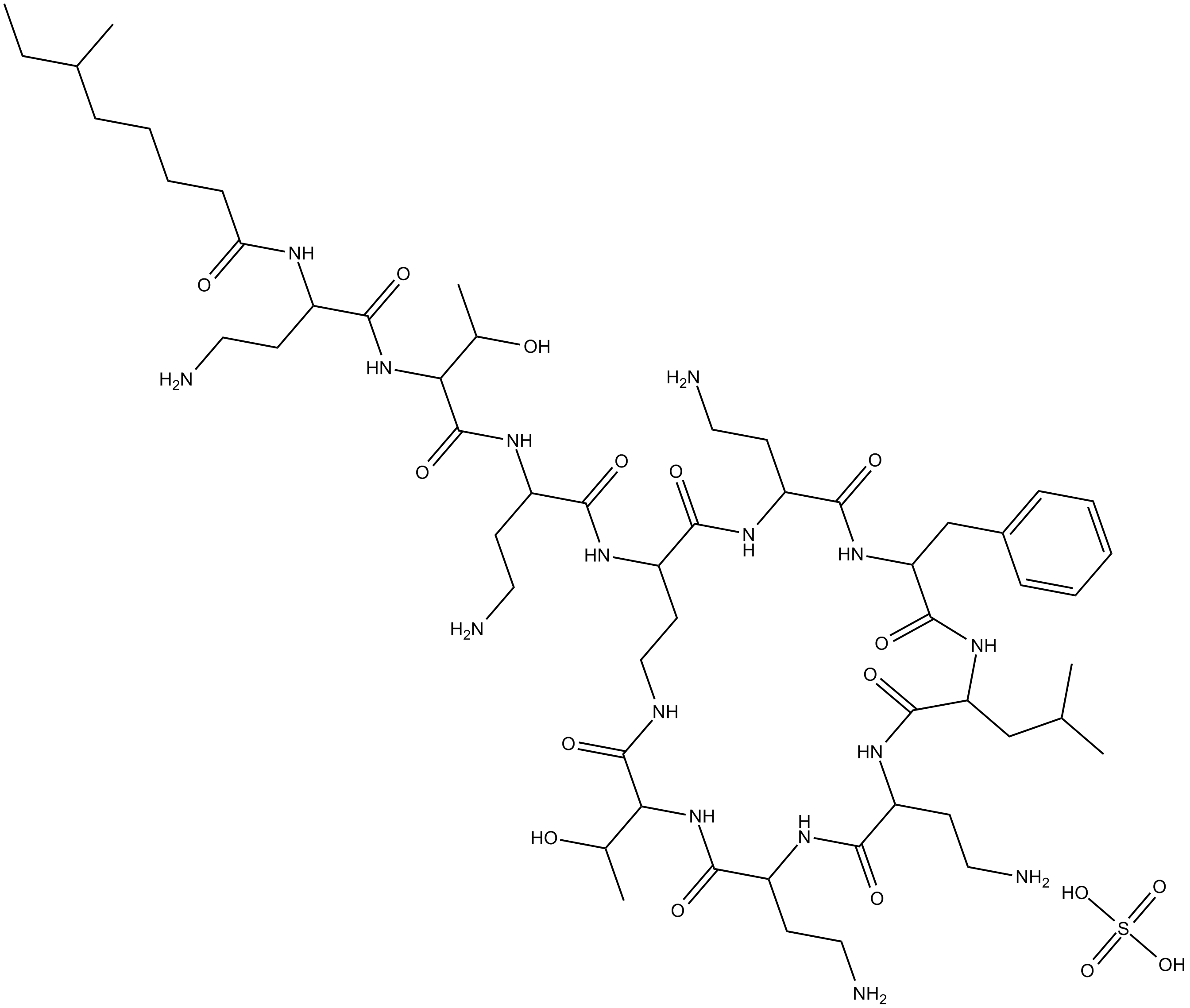
Polymyxin B (sulfate)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Polymyxin B, a mixture of polymyxins B1 and B2, is obtained from Bacillus polymyxa strains. Polymyxin B exhibits bactericidal activity against major multidrug-resistant gram-negative bacteria, most fungi and gram-positive bacteria. As basic polypeptides of about eight amino acids, polymyxins B1 and B2 have cationic detergent action on cell membranes. Moreover, polymyxin B is used for infections with gram-negative organisms, which may be nephrotoxic and neurotoxic. Polymyxin B is appropriate to treat the infections of the meninges, urinary tract, and blood stream, triggered by susceptible Pseudomonas aeruginosa strains.
In vitro: Polymyxin B elicited up-regulation of dendritic cells (DCs) maturation markers, including the increase in the in the expression of co-stimulatory molecule CD86 and HLA-class I and II molecules. Polymyxin B induced a progressive increase in the adhesion property of human DCs. In addition, polymyxin B triggered the activation of the ERK1/2 pathway and IκB-α/NF-κB pathways [1].
In vivo: Male bacteraemia ddY mice were subcutaneously treated with polymyxin B at a dose of 5, 10, 15 or 20 mg/kg for 7 days. Polymyxin B, in a dose-dependent fashion, improved the survival both of OU-01062- and OU-98039-infected mice. In polymyxin B-treated mice, except for 5 mg/kg polymyxin B, the viable cell counts had a tendency to reduce steadily in each concentration group. It was showed a rapid and marked decline of bacterial cell count between 3 to 6 h after infection [2].
References:
[1]. Valentinis, B., Bianchi, A., Zhou, D., Cipponi, A., Catalanotti, F., Russo, V., & Traversari, C. Direct Effects of Polymyxin B on Human Dendritic Cells Maturation: THE ROLE OF I B- /NF- B AND ERK1/2 PATHWAYS AND ADHESION. Journal of Biological Chemistry. 2005; 280(14): 14264-14271.
[2]. Miyajima, Y., Hiramatsu, K., Mizukami, E., Morinaga, R., Ishii, H., & Shirai, R. et al. In vitro and in vivo potency of polymyxin B against IMP-type metallo-β-lactamase-producing Pseudomonas aeruginosa. International Journal of Antimicrobial Agents. 2008; 32(5): 437-440.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 1301.6 |
| Cas No. | 1405-20-5 |
| Formula | C56H98N16O13·H2SO4 |
| Synonyms | Aerosporin,Mastimyxin |
| Solubility | ≤2mg/ml in PBS(pH7.2) |
| Chemical Name | N-(4-amino-1-((1-((4-amino-1-oxo-1-((6,9,18-tris(2-aminoethyl)-15-benzyl-3-(1-hydroxyethyl)-12-isobutyl-2,5,8,11,14,17,20-heptaoxo-1,4,7,10,13,16,19-heptaazacyclotricosan-21-yl)amino)butan-2-yl)amino)-3-hydroxy-1-oxobutan-2-yl)amino)-1-oxobutan-2 |
| SDF | Download SDF |
| Canonical SMILES | O=C(C(NC(C(CCN)NC(CCCCC(C)CC)=O)=O)C(C)O)NC(CCN)C(NC(CCNC(C(NC(C(NC(C(CCN)N1)=O)CCN)=O)C(O)C)=O)C(NC(CCN)C(NC(CC2=CC=CC=C2)C(NC(CC(C)C)C1=O)=O)=O)=O)=O.O=S(O)(O)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构