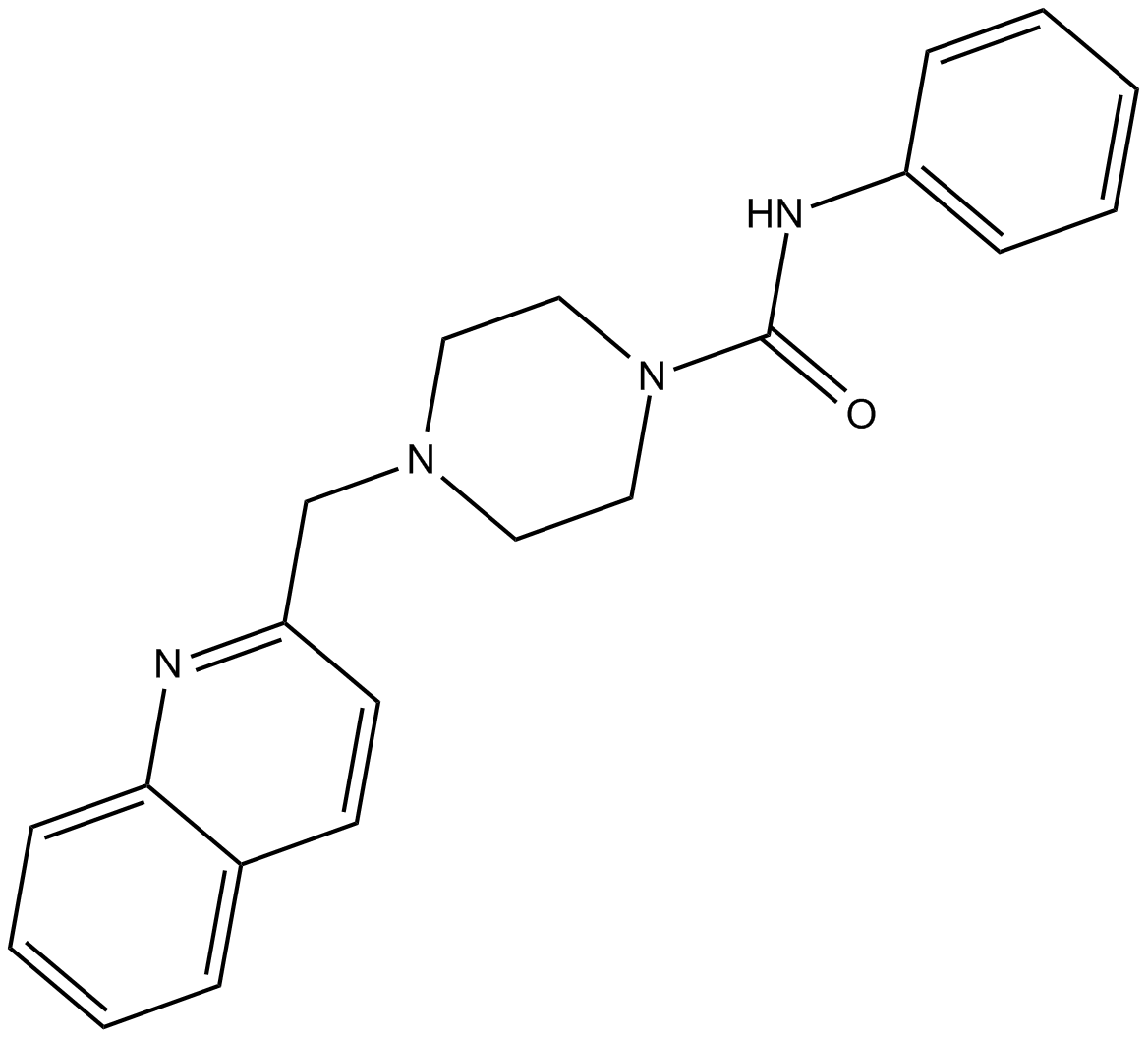
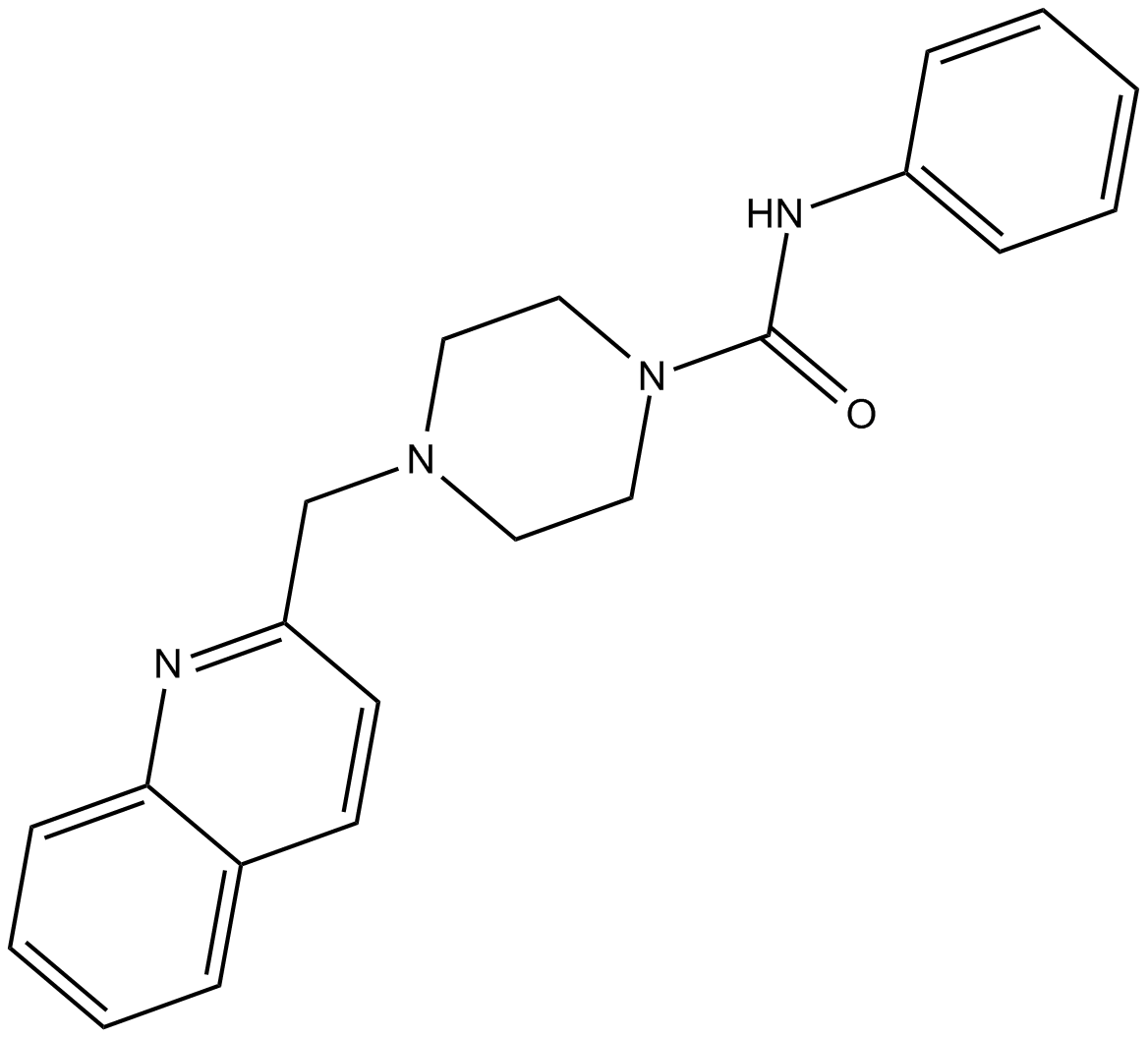
PF-622

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
PF-622 is a potent, time-dependent, irreversible FAAH inhibitor [1].
Fatty acid amide hydrolase (FAAH), belongs to a member of an unusual class of serine hydrolases, is an integral membrane enzyme involved in regulating the fatty acid amide family of lipid transmitters. Genetic or pharmacological inactivation of FAAH leads to elevated endogenous levels of fatty acid amides with analgesic, anti-inflammatory, anxiolytic, and antidepressant phenotypes. The FAAH is an attractive drug target for the treatment of pain [1].
In vitro: PF-622 inhibited the activity of FAAH in a time-dependent manner with the IC50 values of 0.99 and 0.033 μM in human recombinant FAAH for 5 and 60 minutes, respectively [1]. In various human and murine tissue proteome samples, PF-622 showed highly selectivity for FAAH in relative to other serine hydrolases, showing no discernable off-site activity up to 500 μM [1]. PF-622 at 1 μM decreased IL-2 production in both healthy subjects and in HCV patients [2].
References:
[1] Ahn K, Johnson D S, Fitzgerald L R, et al. Novel mechanistic class of fatty acid amide hydrolase inhibitors with remarkable selectivity[J]. Biochemistry, 2007, 46(45): 13019-13030.
[2] Patsenker E, Sachse P, Chicca A, et al. Elevated levels of endocannabinoids in chronic hepatitis C may modulate cellular immune response and hepatic stellate cell activation[J]. International journal of molecular sciences, 2015, 16(4): 7057-7076.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 346.4 |
| Cas No. | 898235-65-9 |
| Formula | C21H22N4O |
| Solubility | ≤0.3mg/ml in ethanol;2mg/ml in DMSO;3mg/ml in dimethyl formamide |
| Chemical Name | N-phenyl-4-(2-quinolinylmethyl)-1-piperazinecarboxamide |
| SDF | Download SDF |
| Canonical SMILES | O=C(Nc1ccccc1)N1CCN(CC1)Cc1ccc2ccccc2n1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |