p-nitro-Pifithrin-α

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
p-nitro-Pifithrin-α, a cell-permeable cyclic analog of pifithrin-α, is an inhibitor of p53 activity [1].
The p53 tumor suppressor gene product can induce apoptotic cell death and plays a dominant role in apoptosis, genomic stability, and inhibition of angiogenesis. The p53 has been considered to be an oncogene and the wild-type gene product actually functions as a tumour suppressor gene. p53 mutations play an important role in the development of many common human malignancies [2].
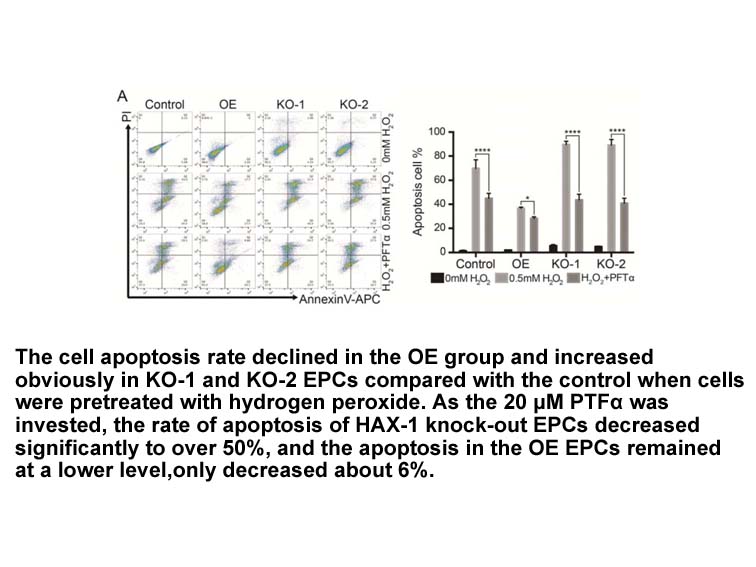
In Vitro: In p53-/- cortical neuron, p-nitro-Pifithrin-α exihibited a p53 inhibitory activity in preventing p53-induced death[1]. p-nitro-Pifithrin-α did not prevent cortical neuronal death induced by p40Met, showing the remarkable specificity in the inhibitory action of p-nitro-Pifithrin-α on p53. p-nitro-Pifithrin-α (300 nM) prevented p53-triggered increase in protein levels of p21/WAF1, indicating that p-nitro-Pifithrin-α behaved as p53 posttranscriptional activity inhibitors. p-nitro-Pifithrin-α at a dose of 30 nM was sufficient to prevent the increase of p21/WAF1 levels [1]. p-nitro-Pifithrin-α was slowly converted into a more potent cyclized form, p-nitro cyclic pifithrin-α, when incubated in biological media (t1/2= 8 h)
In human proximal tubular cells, p-nitro-Pifithrin-α (10 μM) suppressed p53-mediated TGF-β1 expression [3].
In vivo: In a mouse model of non-alcoholic fatty liver disease, p-nitro-Pifithrin-α attenuated steatosis and liver injury in mice fed a high-fat diet [4].
References:
[1] Pietrancosta N, Moumen A, Dono R, et al. Imino-tetrahydro-benzothiazole derivatives as p53 inhibitors: discovery of a highly potent in vivo inhibitor and its action mechanism[J]. Journal of medicinal chemistry, 2006, 49(12): 3645-3652.
[2] Nigro J M, Baker S J, Preisinger A C, et al. Mutations in the p53 gene occur in diverse human tumour types[J]. Nature, 1989, 342(6250): 705-708.
[3] Shimizu H, Yisireyili M, Nishijima F, et al. Indoxyl sulfate enhances p53-TGF-β1-Smad3 pathway in proximal tubular cells[J]. American journal of nephrology, 2013, 37(2): 97-103.
[4] Derdak Z, Villegas K A, Harb R, et al. Inhibition of p53 attenuates steatosis and liver injury in a mouse model of non-alcoholic fatty liver disease[J]. Journal of hepatology, 2013, 58(4): 785-791.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 398.3 |
| Cas No. | 389850-21-9 |
| Formula | C15H15N3O3S·HBr |
| Synonyms | p-nitro-PFT-α |
| Solubility | ≤1mg/ml in DMSO;1mg/ml in dimethyl formamide |
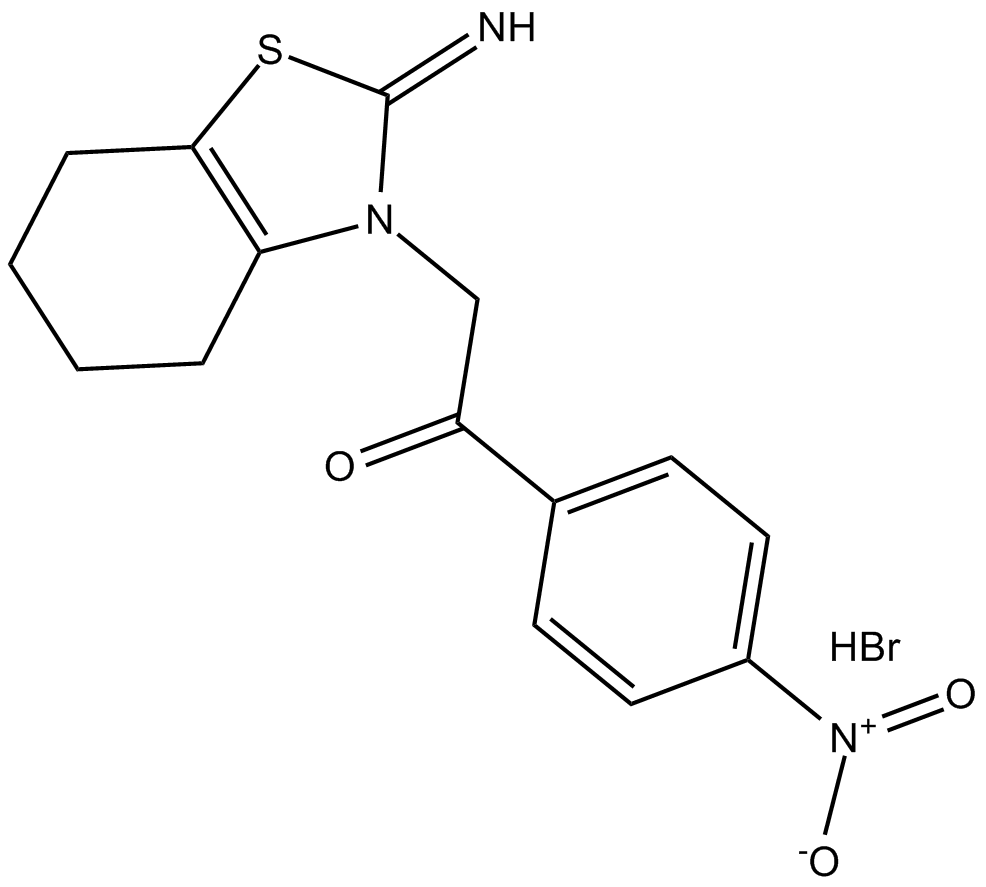
| Chemical Name | 1-(4-nitrophenyl)-2-(4,5,6,7-tetrahydro-2-imino-3(2H)-benzothiazolyl)-ethanone, monohydrobromide |
| SDF | Download SDF |
| Canonical SMILES | N=C(S1)N(CC(C2=CC=C([N+]([O-])=O)C=C2)=O)C3=C1CCCC3.Br |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构
相关生物数据