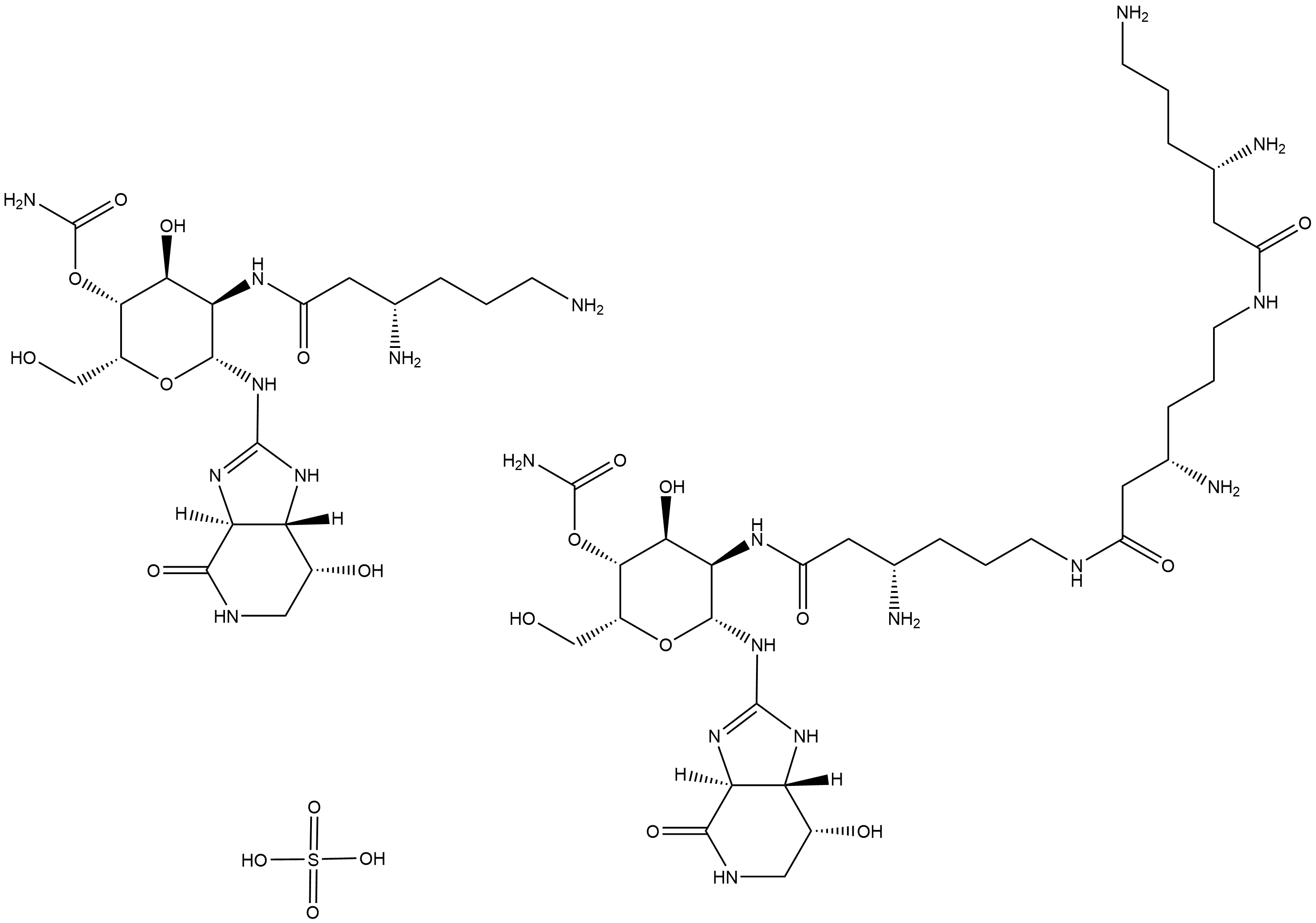
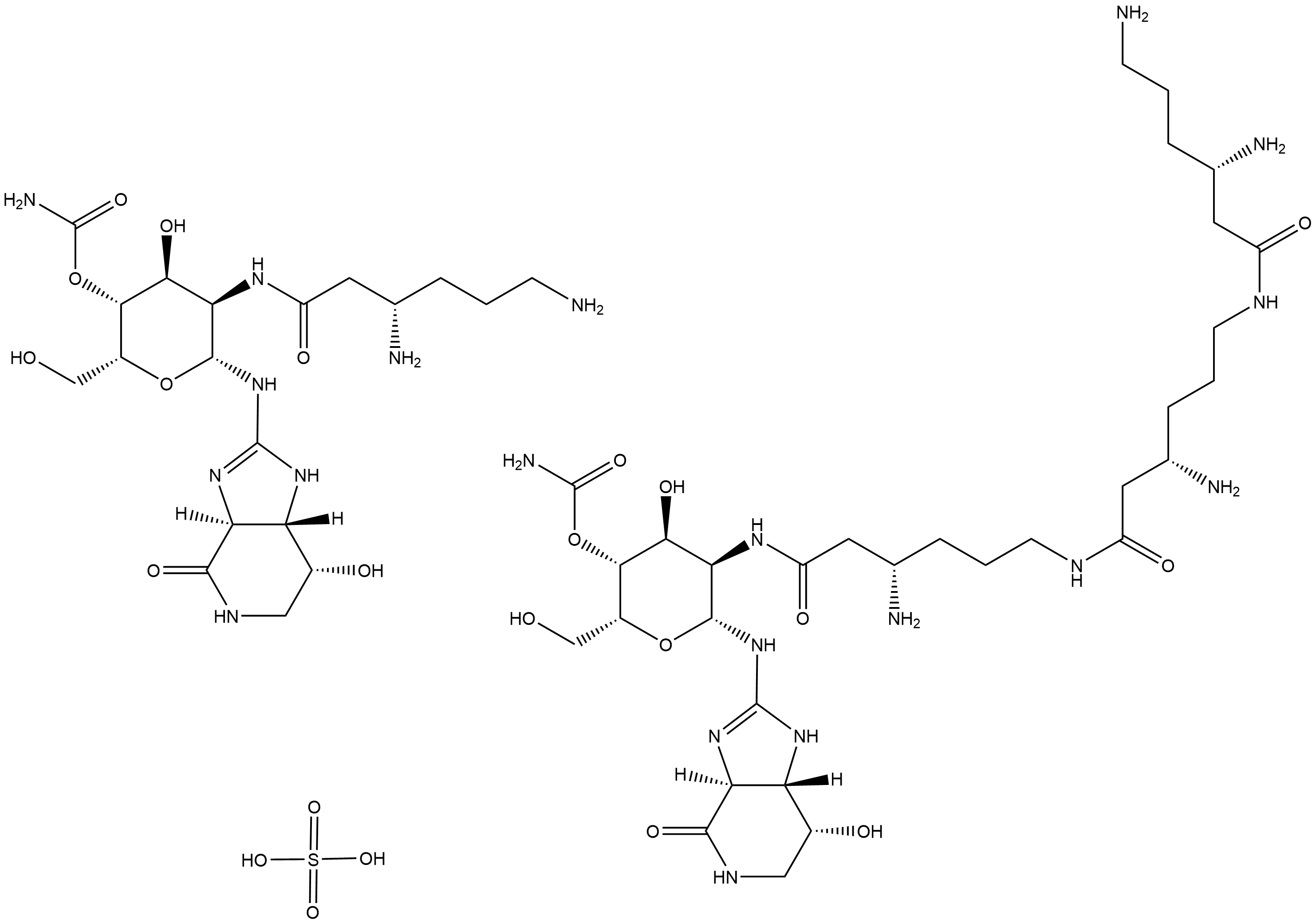
Nourseothricin (sulfate)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Nourseothricin is a broad-spectrum antibiotic.
Nourseothricin is a broad-spectrum antibiotic derived from Streptomyces noursei.
In vitro: Nourseothricin, represents a mixture of streptothricins, mainly D and F. Although very slowly, nourseothricin was found to be able to pass the outer membrane through the porin pores. It has been also shown that nourseothricin was able to generate some channels into the outer membrane. Moreover, resistant strains containing a streptothricin-inactivating acetyl transferase exhibited an additional protecting system, namely a reduced penetrability of the outer membrane. In addition, such strains indeed could be rendered sensitive via damaging the barrier function of the outer membrane [1].
In vivo: Animal study showed that nourseothricin was preferentially excreted via kidney and signs of nephrotoxicity could be seen. Renal handling of nourseothricin was characterized on renal cortical slices under various experimental conditions. Following administration the renal tubular transport system for organic anions was not influenced by nourseothricin. There was a high degree of accumulation of nourseothricin in renal cortical slices. In contrast to PAH accumulation, there was no influence of nitrogen atmosphere, probenecid or trishydroxyaminomethane on nourseothricin accumulation. Age dependent differences in nourseothricin accumulation did not exist [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Seltmann G. Resistance of Escherichia coli to nourseothricin (streptothricin): sensitization of resistant strains by abolition of its outer membrane resistance. Zentralbl Bakteriol. 1992 Jan;276(2):143-51.
[2] Brunlich H, Hoffmann H, Bocker H. Renal handling of nourseothricin. Pharmazie. 1988 Mar;43(3):200-2.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 1359.48 |
| Cas No. | 96736-11-7 |
| Formula | C31H58N12O10·C19H34N8O8·H2SO4 |
| Synonyms | clonNAT; Streptothricin; Streptothricin sulfate |
| Solubility | ≤10mg/ml in PBS, pH 7.2 |
| Chemical Name | sulfuric acid--(2R,3R,4S,5R,6R)-5-((S)-3,6-diaminohexanamido)-4-hydroxy-6-(((3aS,7R,7aS)-7-hydroxy-4-oxo-3a,4,5,6,7,7a-hexahydro-1H-imidazo[4,5-c]pyridin-2-yl)amino)-2-(hydroxymethyl)tetrahydro-2H-pyran-3-yl carbamate--(2R,3R,4S,5R,6R)-5-((S)-3-amino-6-(( |
| SDF | Download SDF |
| Canonical SMILES | O=C(C[C@@H](N)CCCN)N[C@H]1[C@H](NC2=N[C@@](C(NC[C@H]3O)=O)([H])[C@]3([H])N2)O[C@H](CO)[C@H](OC(N)=O)[C@H]1O.OS(=O)(O)=O.O=C(C[C@@H](N)CCCNC(C[C@@H](N)CCCNC(C[C@@H](N)CCCN)=O)=O)N[C@H]4[C@H](NC5=N[C@@](C(NC[C@H]6O)=O)([H])[C@]6([H])N5)O[C@H](CO)[C@H](OC(N) |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构