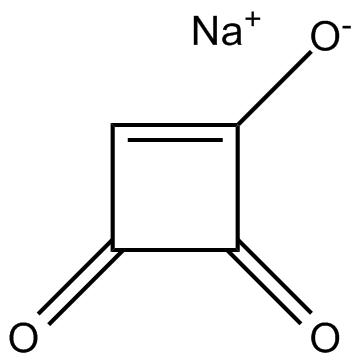
Moniliformin (sodium salt)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Moniliformin induces mitotic arrest at the metaphase stage.
Mitosis is a part of the cell cycle when replicated chromosomes are separated into two new nuclei. The process of mitosis is divided into stages corresponding to the completion of one set of activities and the start of the next.
In vitro: Moniliformin, first isolated as a mycotoxin from Fusarium moniliforme, was found to be phytotoxic and arrests mitosis of maize root meristematic cells at the metaphase stage. The mitotic spindle could be disrupted by the treatment of moniliformin, but no direct effect on tubulin had been observed [1].
In vivo: A previous study was conducted on rat heart to in situ determine the myocardial toxicity of moniliformin, originally isolated from mouldy corn and soil samples in the Keshan disease prevalent area in China. Results showed that perfusion of moniliformin 10-7 mol/liter in isolated heart decreased myocardial contractile force by 52%. Intravenous injection of moniliformin at 1/6 and 1/4 LD50 could markedly inhibit cardiac hemodynamic variables associated with myocardial contractile function. Moreover, moniliformin was able to decrease +/- LV dP/dt max by 52%, and induce ventricular arrhythmia. These findings indicated that moniliformin was toxic to mammalian heart and might be an important factor relative to Keshan disease [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Duke, S. O. and Dayan, F.E. Modes of action of microbially-produced phytotoxins. Toxins (Basel) 3(8), 1038-1064 (2011).
[2] Fan LL, Li J, Sun LH. Effect of moniliformin on myocardial contractility in rats. Biomed Environ Sci. 1991 Sep;4(3):290-4.
| Physical Appearance | A crystalline solid |
| Storage | Store at 4°C |
| M.Wt | 120 |
| Cas No. | 71376-34-6 |
| Formula | C4HO3·Na |
| Solubility | ≤10mg/ml in H2O |
| Chemical Name | 3-hydroxy-3-cyclobutene-1,2-dione, monosodium salt |
| SDF | Download SDF |
| Canonical SMILES | O=C1C(C=C1[O-])=O.[Na+] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
Quality Control & MSDS
- View current batch:
-
Purity ≥ 95.00%
- COA (Certificate Of Analysis)
- MSDS (Material Safety Data Sheet)
Chemical structure