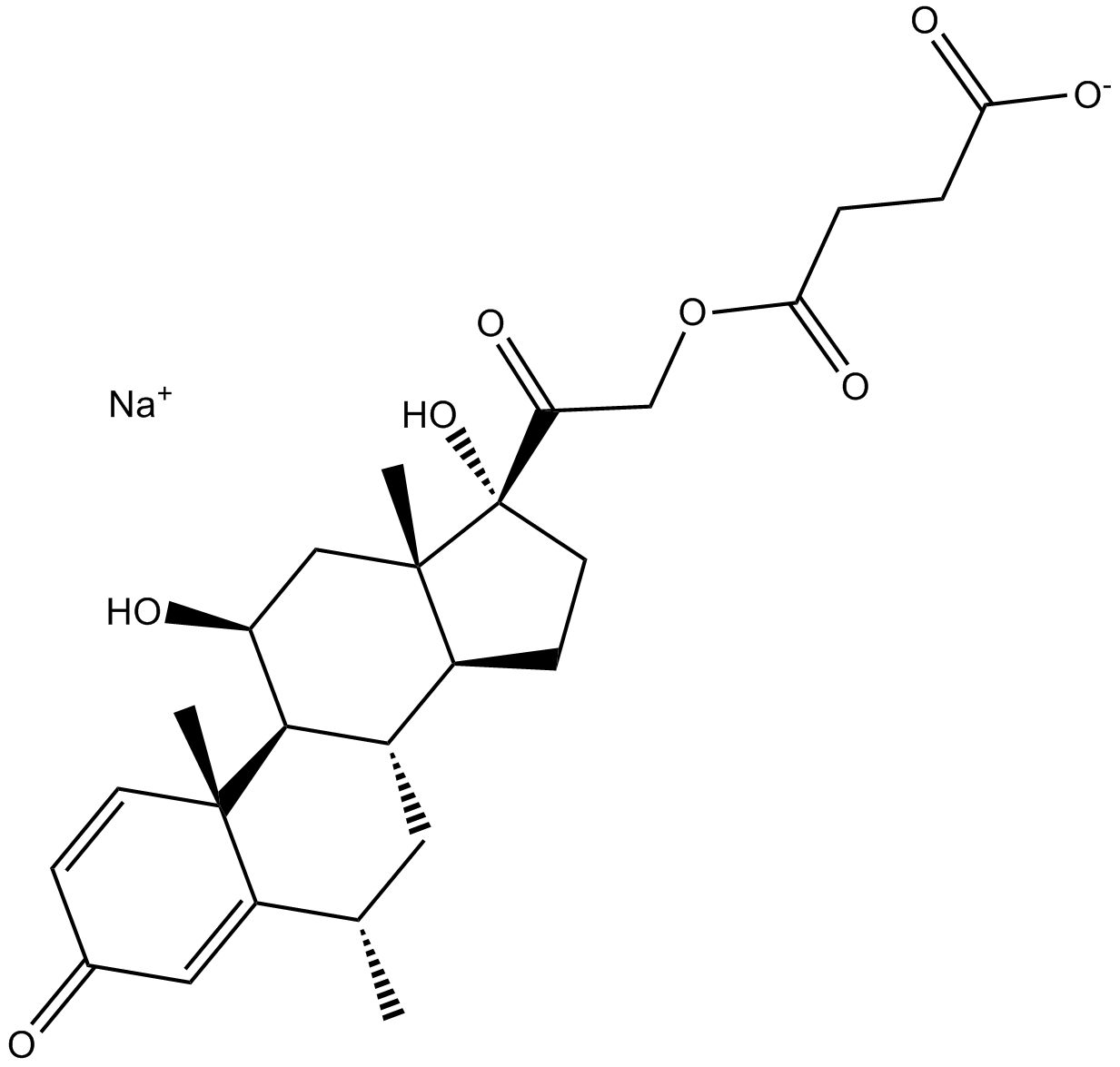
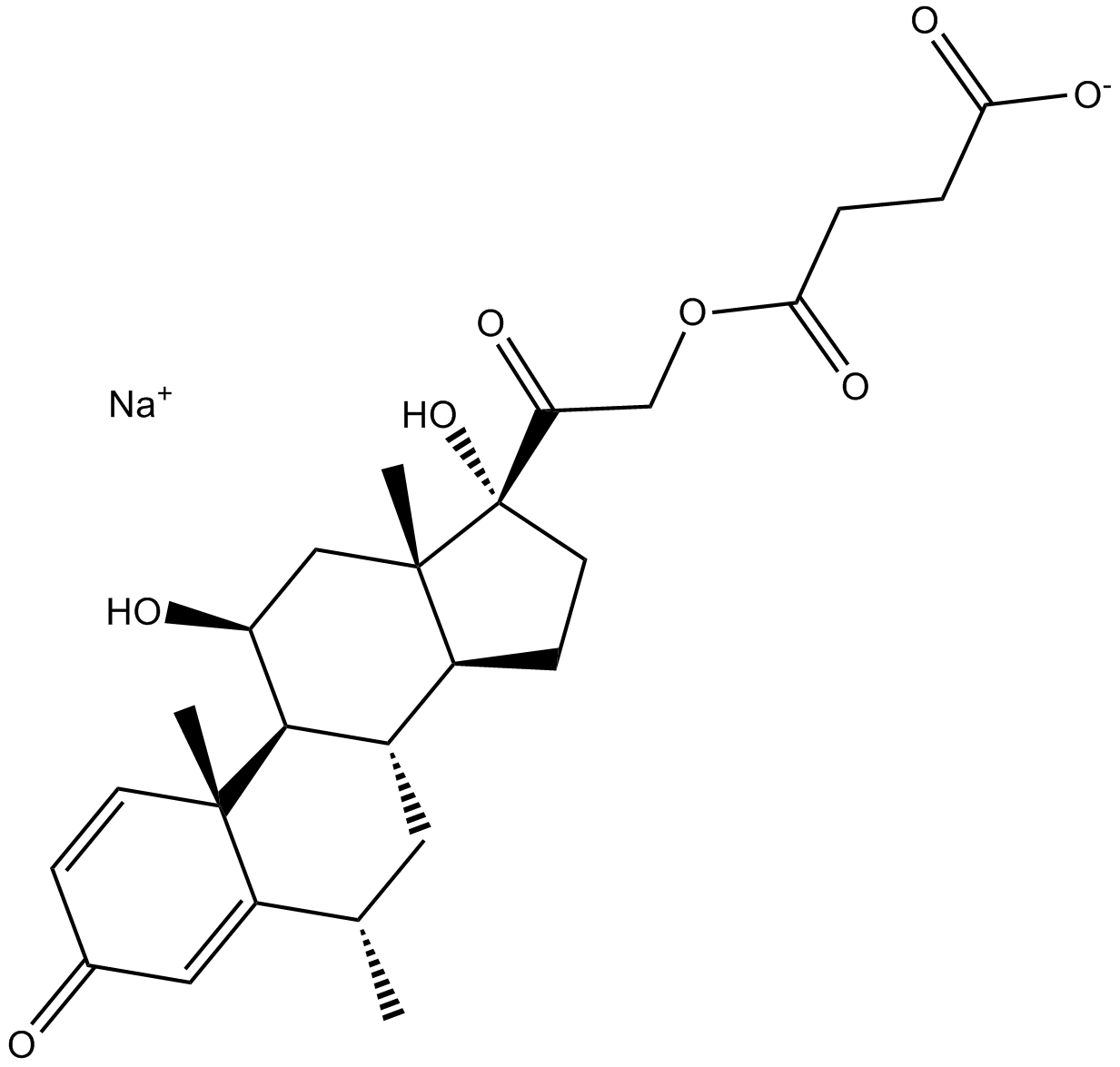
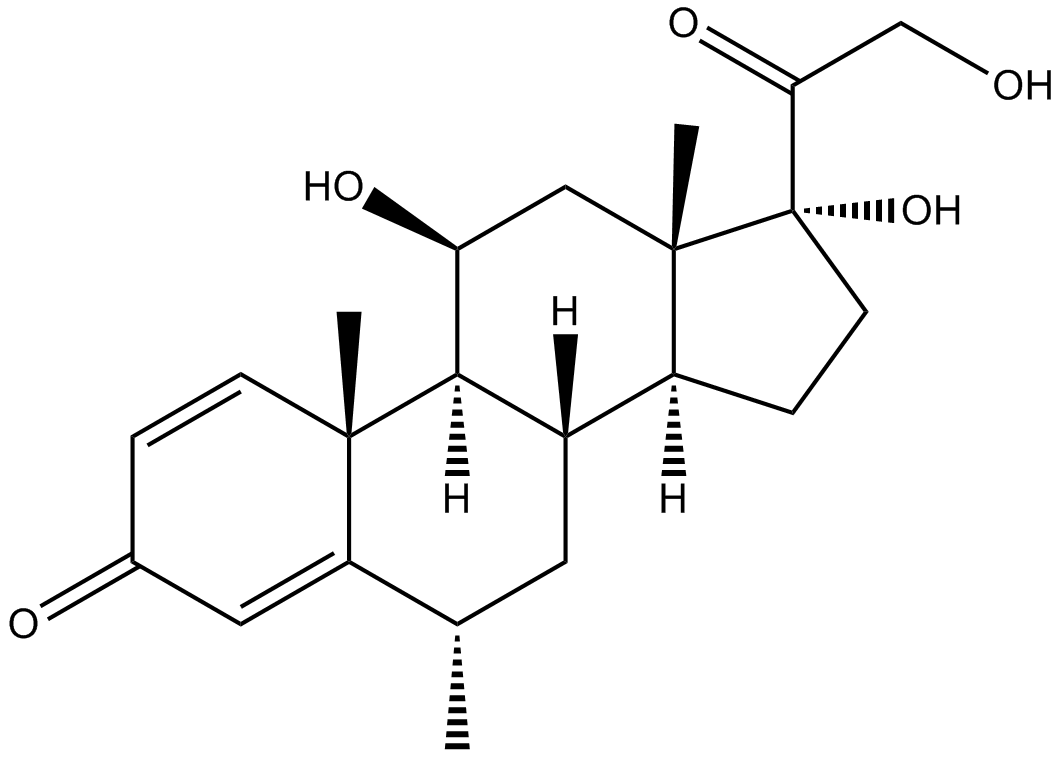
Methylprednisolone Sodium Succinate

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Methylprednisolone sodium succinate is the sodium succinate ester of methylprednisolone which a synthetic corticosteroid with anti-inflammatory and immunomodulating properties [1].
Methylprednisolone binds to specific nuclear receptors, altering gene expression and reducing proinflammatory cytokine production. Methylprednisolone also decreases the number of circulating lymphocytes, induces cell differentiation, and triggers apoptosis in sensitive tumor cell populations [1].
In human polymorphonuclear leukocytes, methylprednisolone sodium succinate exerted no effect on O2-· production, H2O2 production, or chemiluminescence at low concentrations (0.04 ~ 0.22 mM), but triggered significant inhibition at a high concentration of 2.7 mM [2]. In human neutrophils, methylprednisolone sodium succinate at a high concentration of 1 mg/ml inhibited chemotactic response [3].
In the National Acute Spinal Cord Injury Study, methylprednisolone sodium succinate (30 mg/kg bolus followed by 5.4 mg/kg/h infusion for 23 hours) showed no significant differences against the control group overall at 6 weeks. However, the recovery of motor score and sensory touch score at 6 weeks was slightly, but significantly greater in the methylprednisolone sodium succinate-treated group than the placebo group for those treated within 8 hours of injury [4].
References:
[1]. National Center for Biotechnology Information. PubChem Database. Methylprednisolone, CID=6741, https://pubchem.ncbi.nlm.nih.gov/compound/Methylprednisolone (accessed on July 20, 2020)
[2]. Levine P H, Hardin J C, Scoon K L, et al. Effect of corticosteroids on the production of superoxide and hydrogen peroxide and the appearance of chemiluminescence by phagocytosing polymorphonuclear leukocytes. Inflammation, 1981, 5(1): 19-27.
[3]. Majeski J A, Alexander J W. The steroid effect on the in vitro human neutrophil chemotactic response. Journal of Surgical Research, 1976, 21(4): 265-268.
[4]. Steeves J, Blight A. Chapter 24 - Spinal cord injury clinical trials translational process, review of past and proposed acute trials with reference to recommended trial guidelines. Handbook of Clinical Neurology, 2012, 109: 386-397.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 496.53 |
| Cas No. | 2375-03-3 |
| Formula | C26H33NaO8 |
| Solubility | ≥49.7 mg/mL in DMSO; ≥13.1 mg/mL in EtOH; ≥2.94 mg/mL in H2O |
| Chemical Name | sodium 4-(2-((6S,8S,9S,10R,11S,13S,14S,17R)-11,17-dihydroxy-6,10,13-trimethyl-3-oxo-6,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-3H-cyclopenta[a]phenanthren-17-yl)-2-oxoethoxy)-4-oxobutanoate |
| SDF | Download SDF |
| Canonical SMILES | O[C@@]1(C(COC(CCC([O-])=O)=O)=O)[C@](C2)(C)[C@@H](CC1)[C@H](C[C@@H]3C)[C@@H]([C@@](C)(C=C4)C3=CC4=O)[C@H]2O.[Na+] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构