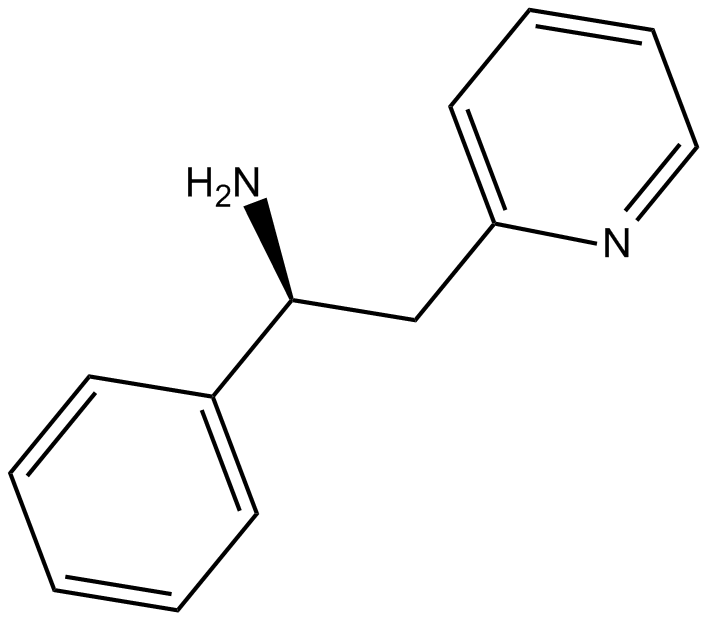
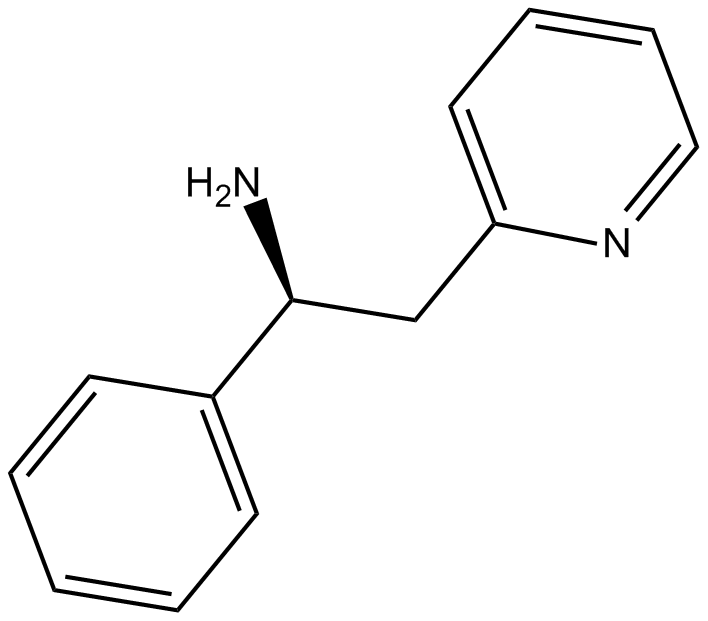
Lanicemine

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 4-7 μM
Lanicemine is a NMDA channel blocker.
NMDA receptors, glutamate-gated cation channels with high calcium permeability, play critical roles in various aspects of the biology. They are important for the development of the CNS, generation of rhythms, and the processes underlying memory, learning, and neuroplasticity.
In vitro: Previous study with lanicemine and ketamine found that both compounds could bind with low-to-moderate affinity to sites within the NMDA channel pore, exhibit strong voltage dependence, and have similar lack of NR2A vs NR2B subunit selectivity [1].
In vivo: In animal study, cortical EEG recordings were obtained from rats trained to perform an auditory detection task for food reward. Results showed that both lanicemine and ketamine produced pronounced dose-dependent elevations in spontaneous gamma-band EEG, but only gamma changes for ketamine were tightly coupled to increases in locomotor activity, indicating that lanicemine not only engaged brain circuits involved in the generation of gamma-EEG, but also influenced these networks independent of the broader systemslevel disruptions typical of ketamine [1].
Clinical trial: A qEEG crossover study was conducted in healthy volunteers but was stopped earlier than planned following two serious adverse events occurring during ketamine infusion. Significant increases in gamma-band EEG were found for both lanicemine and ketamine, and baseline-corrected gamma-EEG following lanicemine treatment was statistically indistinguishable from ketamine. Moreover, both lanicemine and ketamine produced significant reductions in prefrontal theta-cordance. Furthermore, no serious adverse events were observed associated with lanicemine [1].
Reference:
[1] G. Sanacora, M. A. Smith, S. Pathak, et al. Lanicemine: A low-trapping NMDA channel blocker produces sustained antidepressant efficacy with minimal psychotomimetic adverse effects. Molecular Psychiatry 19(9), 978-985 (2014).
| Physical Appearance | A neat oil |
| Storage | Store at -20°C |
| M.Wt | 198.3 |
| Cas No. | 153322-05-5 |
| Formula | C13H14N2 |
| Synonyms | AZD 6765,AR-R 15896AR |
| Solubility | ≤20mg/ml in ethanol;30mg/ml in DMSO;5mg/ml in dimethyl formamide |
| Chemical Name | (αS)-phenyl-2-pyridineethanamine |
| SDF | Download SDF |
| Canonical SMILES | N[C@H](C1=CC=CC=C1)CC2=CC=CC=N2 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构