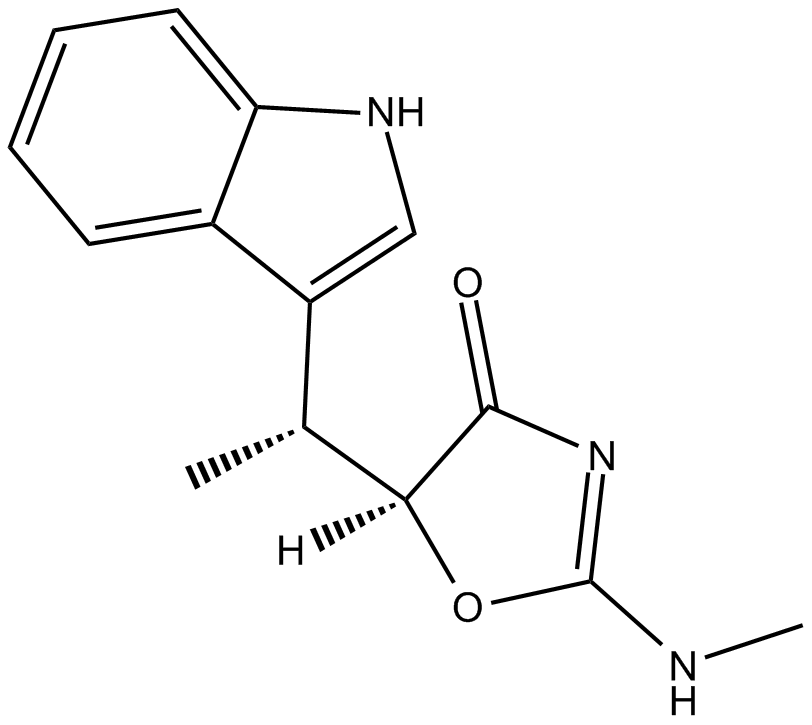
Indolmycin

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
IC50: 30 μM for prokaryotic tryptophanyl-tRNA synthetase
Indolmycin is an antibiotic.
Tryptophanyl-tRNA synthetase (TrpRS) catalyzes activation of tryptophan through ATP and transfer to tRNATrp , leading to translation of the genetic code for tryptophan.
In vitro: Indolmycin was found to ba a bacteriostatic that showed good activity against methicillin-resistant S. aureus, methicillin-susceptible Staphylococcus aureus, and vancomycin-intermediate S. aureus, including strains resistant to mupirocin or fusidic acid. Spontaneous indolmycin-resistant mutants was observed at a lower frequency than those selected by mupirocin or fusidic acid and exhibited no cross-resistance with the comparative drugs. High-level resistance of indolmycin at its MIC of 128 mg/L that was associated with an H43N mutation in tryptophanyl-tRNA synthetase, the target enzyme of indolmycin, led to loss of bacterial fitness. However, the locus responsible for low-level indolmycin resistance (indolmycin MICs 8-32 mg/L) was not identified [1].
In vivo: Animal study found that indolmycin could completely clear H. pylori in experimentally infected Mongolian gerbils at a dose of 10 mg/kg. Therefore, indolmycin could be regarded as a candidate for the treatment of H. pylori infection [2].
Clinical trial: So far, no clinical study has been conducted.
References:
[1] Hurdle JG, O'Neill AJ, Chopra I. Anti-staphylococcal activity of indolmycin, a potential topical agent for control of staphylococcal infections. J Antimicrob Chemother. 2004 Aug;54(2):549-52. Epub 2004 Jul 8.
[2] Vecchione JJ, Sello JK. A novel tryptophanyl-tRNA synthetase gene confers high-level resistance to indolmycin. Antimicrob Agents Chemother 2009; 53: 3972-3980.
| Physical Appearance | A white powder |
| Storage | Store at -20°C |
| M.Wt | 257.3 |
| Cas No. | 21200-24-8 |
| Formula | C14H15N3O2 |
| Synonyms | PA 155A,TAK-083 |
| Solubility | Soluble in ethanol;methanol;DMSO;dimethyl formamide |
| Chemical Name | 5S-[(1R)-1-(1H-indol-3-yl)ethyl]-2-(methylamino)-4(5H)-oxazolone |
| SDF | Download SDF |
| Canonical SMILES | O=C(N=C(NC)O1)[C@]1([H])[C@H](C)C2=CNC3=CC=CC=C32 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |