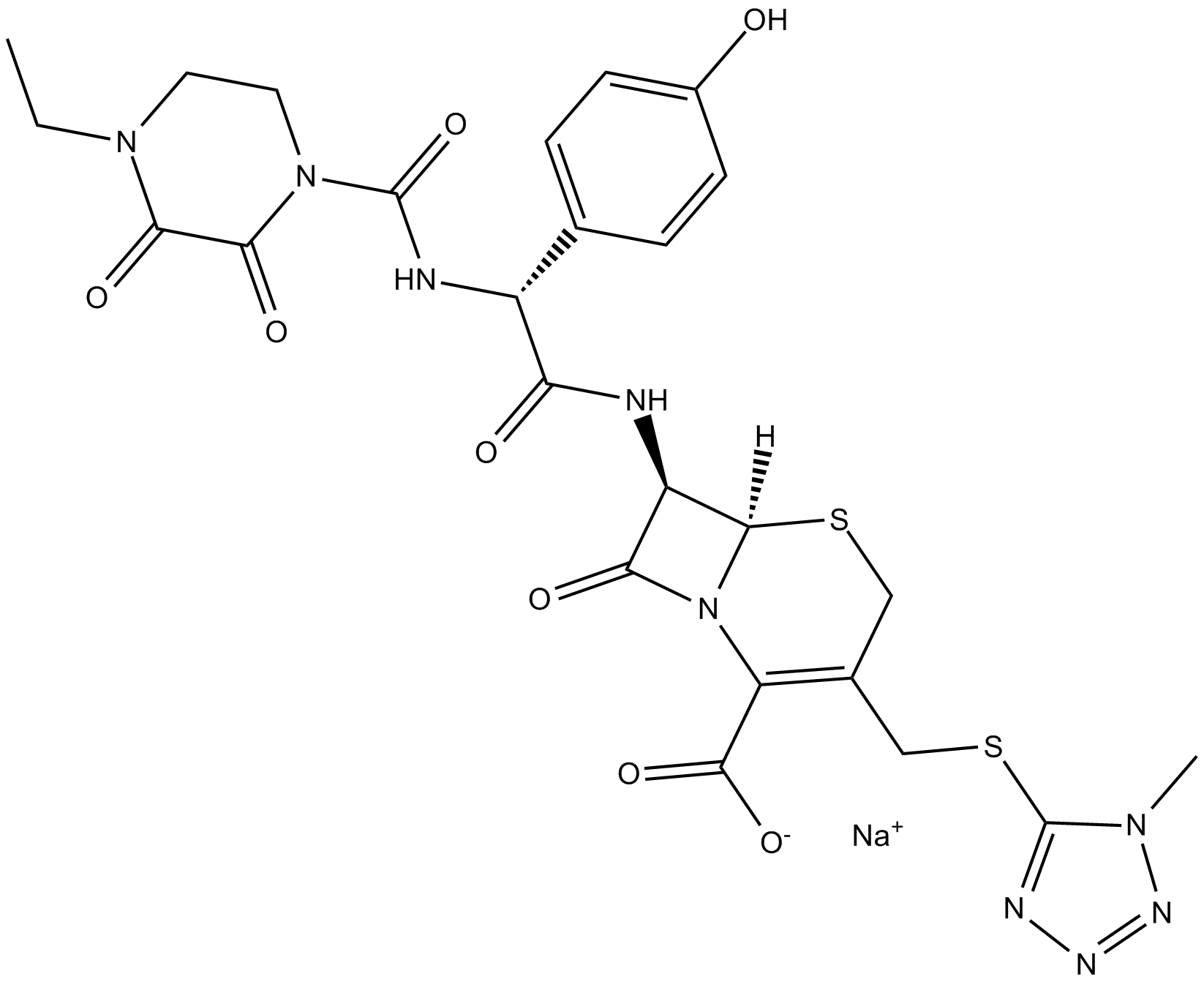
Cefoperazone (sodium salt)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Cefoperazone is a new semisynthetic cephalosporin with a broad spectrum of antibacterial activity. Cefoperazone shows high activity against gram-positive bacteria and gram-negative bacilli, such as Escherichia coli, Klebsiella pneumoniae, and Proteus species [1].
In vitro: There was only a small spread between the minimum inhibitory concentrations and the minimum bactericidal concentrations of cefoperazone and a significant decrease in activity with an increase in inoculum size. Cefoperazone is relatively stable to hydrolysis to β-lactamases produced by gram-negative bacteria. Relative rates of hydrolysis of cefoperazone by cephalosporinases were 7.0 to 0.01[1]. In 50 strains of N. gonorrhoeae, the MIC50 of cefoperazone was ≤ 0.004-0.06 μg/ml [2].
In vivo: In four patients with cholelithiasis and one patient with carcinoma of the head of the pancreas, all of whom had normal renal functions, cefoperazone was intravenously administrated. In common duct bile, the maximum concentrations of cefoperazone ranged from 373.4 to 3,100 μg/ml while the concentrations ranged from 6.8 to 680 μg/ml in gall bladder bile. Cefoperazone concentrations of the gall bladder wall ranged from 16.8 to 48.0 μg/g [3].
References:
[1] Matsubara N, Minami S, Muraoka T, et al. In vitro antibacterial activity of cefoperazone (T-1551), a new semisynthetic cephalosporin[J]. Antimicrobial agents and chemotherapy, 1979, 16(6): 731-735.
[2] Baker C N, Thornsberry C, Jones R N. In vitro antimicrobial activity of cefoperazone, cefotaxime, moxalactam (LY127935), azlocillin, mezlocillin, and other beta-lactam antibiotics against Neisseria gonorrhoeae and Haemophilus influenzae, including beta-lactamase-producing strains[J]. Antimicrobial agents and chemotherapy, 1980, 17(4): 757-761.
[3] Nakamura T, Hashimoto I, Sawada Y, et al. Cefoperazone concentrations in bile and gall bladder wall after intravenous administration[J]. Antimicrobial agents and chemotherapy, 1980, 18(6): 980-982.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 667.7 |
| Cas No. | 62893-20-3 |
| Formula | C25H26N9O8S2·Na |
| Synonyms | Cefoneg,Cefosint,CP 52,640-2,Perocef |
| Solubility | ≥73 mg/mL in DMSO; insoluble in EtOH; ≥34.6 mg/mL in H2O |
| Chemical Name | (6R,7R)-7-[[(2R)-2-[[(4-ethyl-2,3-dioxo-1-piperazinyl)carbonyl]amino]-2-(4-hydroxyphenyl)acetyl]amino]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, monosodium salt |
| SDF | Download SDF |
| Canonical SMILES | CCN(CCN(C(N[C@H](C1=CC=C(O)C=C1)C(N[C@@H]2C(N3[C@]2([H])SCC(CSC4=NN=NN4C)=C3C([O-])=O)=O)=O)=O)C5=O)C5=O.[Na+] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
| 细胞实验 [1]: | |
|
细胞系 |
淋病奈瑟菌 |
|
溶解方法 |
在DMSO中的溶解度≤20mg/ml 。为了获得更高的浓度,可以将离心管在37℃加热10分钟和/或在超声波浴中震荡一段时间。原液可以在-20℃以下储存几个月。 |
|
反应条件 |
0.004-0.06 μg/ml |
|
应用 |
Cefoperazone对由革兰氏阴性细菌产生的β-内酰胺酶水解相对稳定。头孢菌素酶水解cefoperazone的相对速率为7.0至0.01。在50株淋病奈瑟菌中,cefoperazon的MIC50≤0.004-0.06μg/ml |
|
References: [1]. Baker CN, Thornsberry C, Jones RN. In vitro antimicrobial activity of cefoperazone, cefotaxime, moxalactam (LY127935), azlocillin, mezlocillin, and other beta-lactam antibiotics against Neisseria gonorrhoeae and Haemophilus influenzae, including beta-lactamase-producing strains. Antimicrob Agents Chemother. 1980 Apr;17(4):757-61. PubMed PMID: 6249195; PubMed Central PMCID: PMC283867. |
|
质量控制和MSDS
- 批次:
化学结构