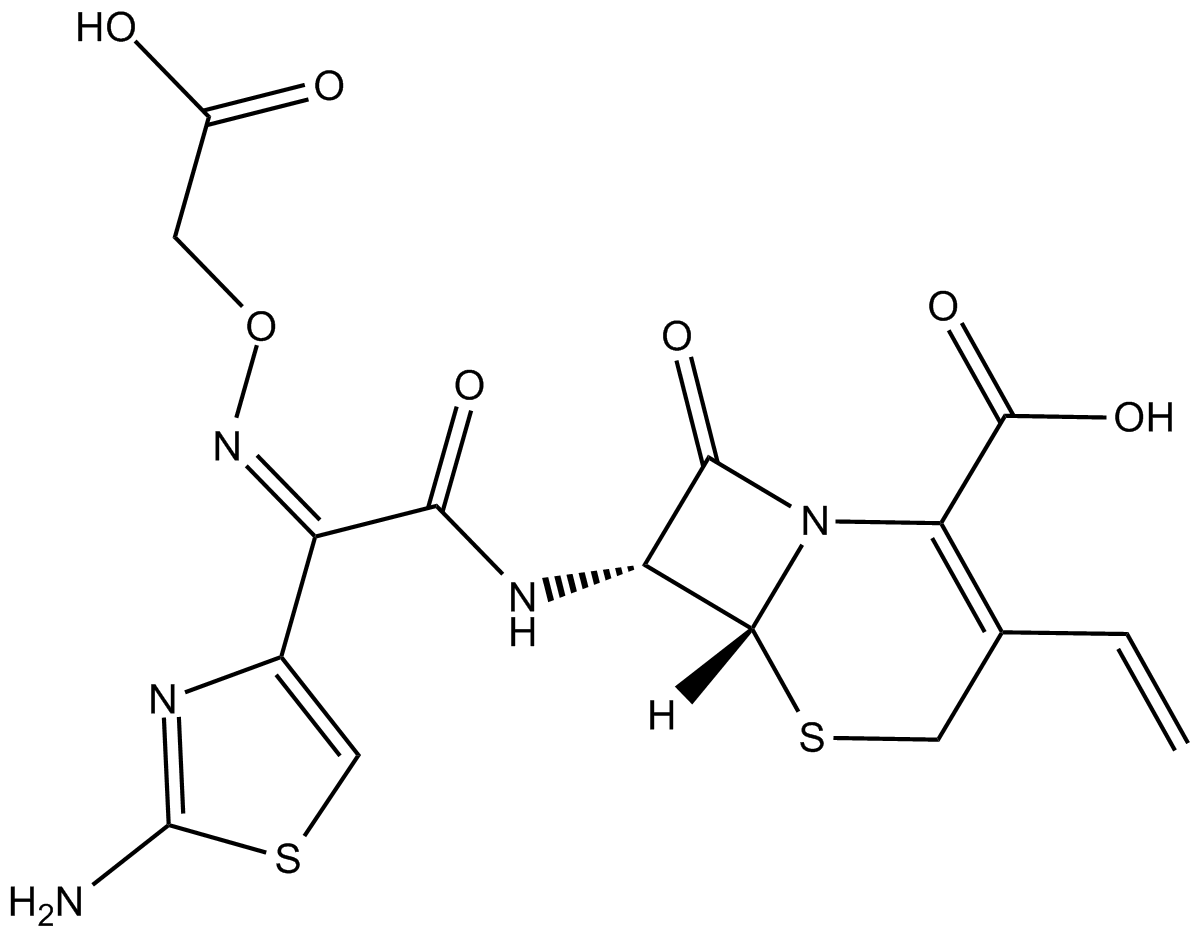
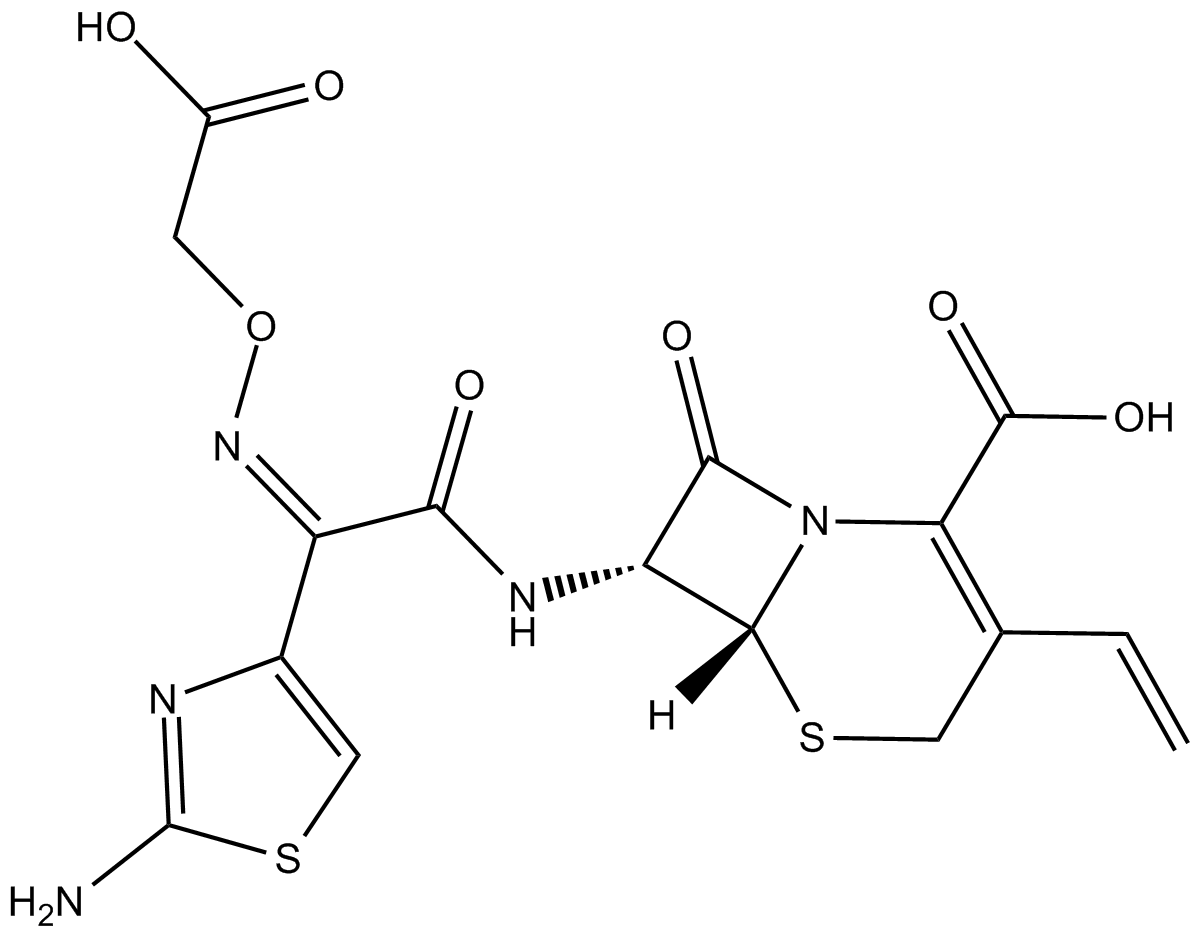
Cefixime

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
MIC: from <0.025 to 25 μg/ml for E. coli, K. pneumoniae, and H. influenzae
Cefixime is a third generation cephalosporin antibiotic.
The cephalosporins, a class of β-lactam antibiotics, are originally derived from the fungus Acremonium.
In vitro: Previous study found that cefixime was more active than cephalexin, cefaclor, and amoxicillin against various gram-negative bacteria. Cefixime was also significantly more active than tested reference drugs against clinical isolates of Klebsiella pneumoniae, Escherichia coli, indole-positive and -negative Proteus species, Providencia species, and Neisseria gonorrhoeae. Moreover, cefixime was active against strains of K. pneumoniae, E. coli, as well as Proteus mirabilis resistant to the reference agents [1].
In vivo: The therapeutic activities of cefixime in mice infected with gram-negative bacilli were found to be far superior to the activities of cephalexin, cefaclor, and amoxicillin, but they were inferior to the activities against infection with Staphylococcus aureus [1].
Clinical trial: Previous study showed that the clinical success was observed in 94% of cefixime-treated patients. At the end of treatment, the overall eradication rate in the cefixime treatment group was 92% and ranged from 76% (cefaclor) to 98% (cefuroxime axetil) in the comparator treatment groups [2].
References:
[1] Kamimura, T. ,Kojo, H.,Matsumoto, Y., et al. In vitro and in vivo antibacterial properties of FK 027, a new orally active cephem antibiotic. Antimicrobial Agents and Chemotherapy 25(1), 98-104 (1984).
[2] Quintiliani R. Cefixime in the treatment of patients with lower respiratory tract infections: results of US clinical trials. Clin Ther. 1996 May-Jun;18(3):373-90; discussion 372.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 453.4 |
| Cas No. | 79350-37-1 |
| Formula | C16H15N5O7S2 |
| Synonyms | CL-284635,FK-027,FR17027 |
| Solubility | insoluble in H2O; ≥46.2 mg/mL in DMSO; ≥6.88 mg/mL in EtOH |
| Chemical Name | (6R,7R)-7-[[(2Z)-2-(2-amino-4-thiazolyl)-2-[carboxymethoxy)imino]acetyl]amino]-3-ethenyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | [H][C@@]1([C@@H]2NC(/C(C3=CSC(N)=N3)=N\OCC(O)=O)=O)N(C(C(O)=O)=C(C=C)CS1)C2=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构