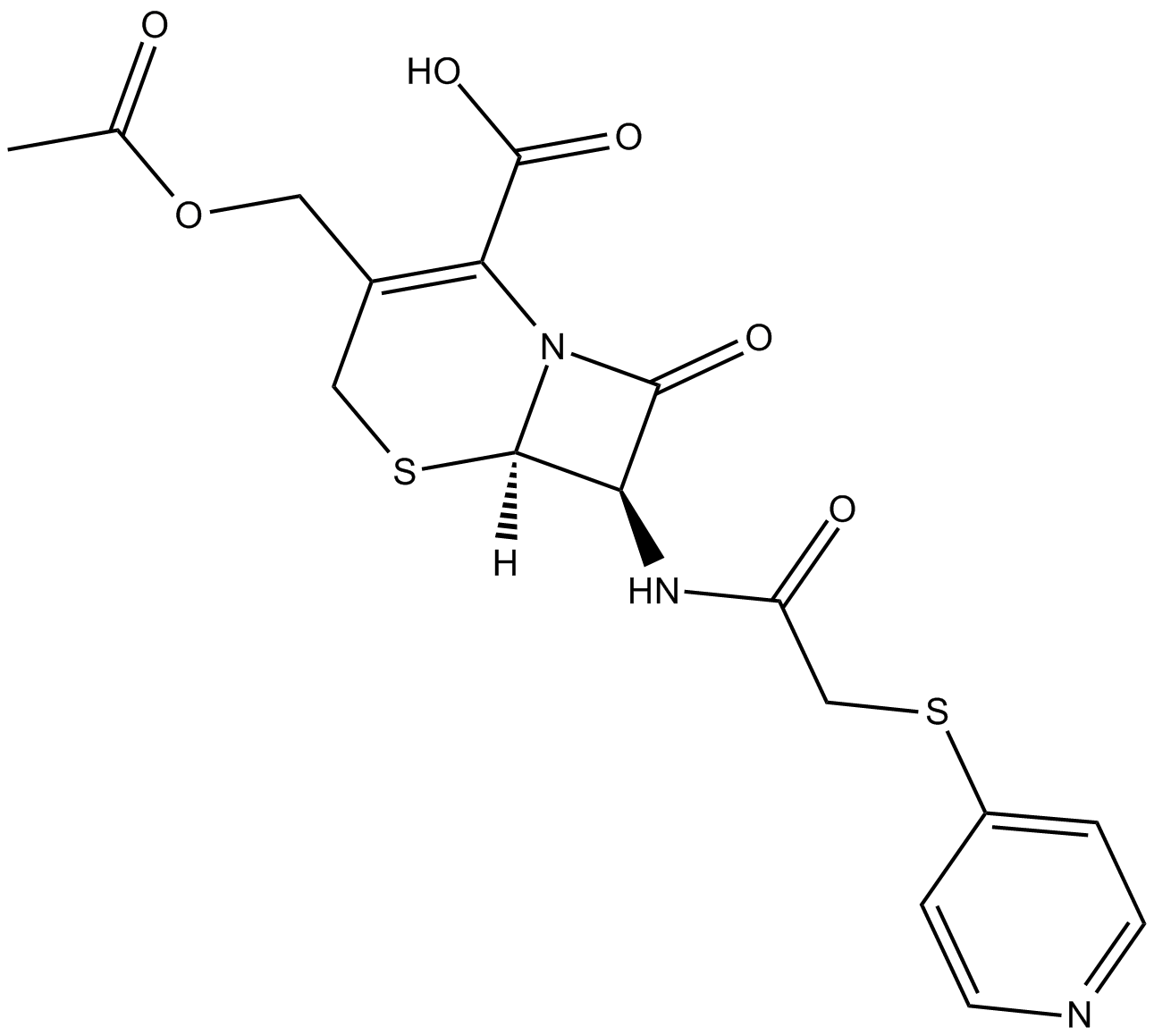
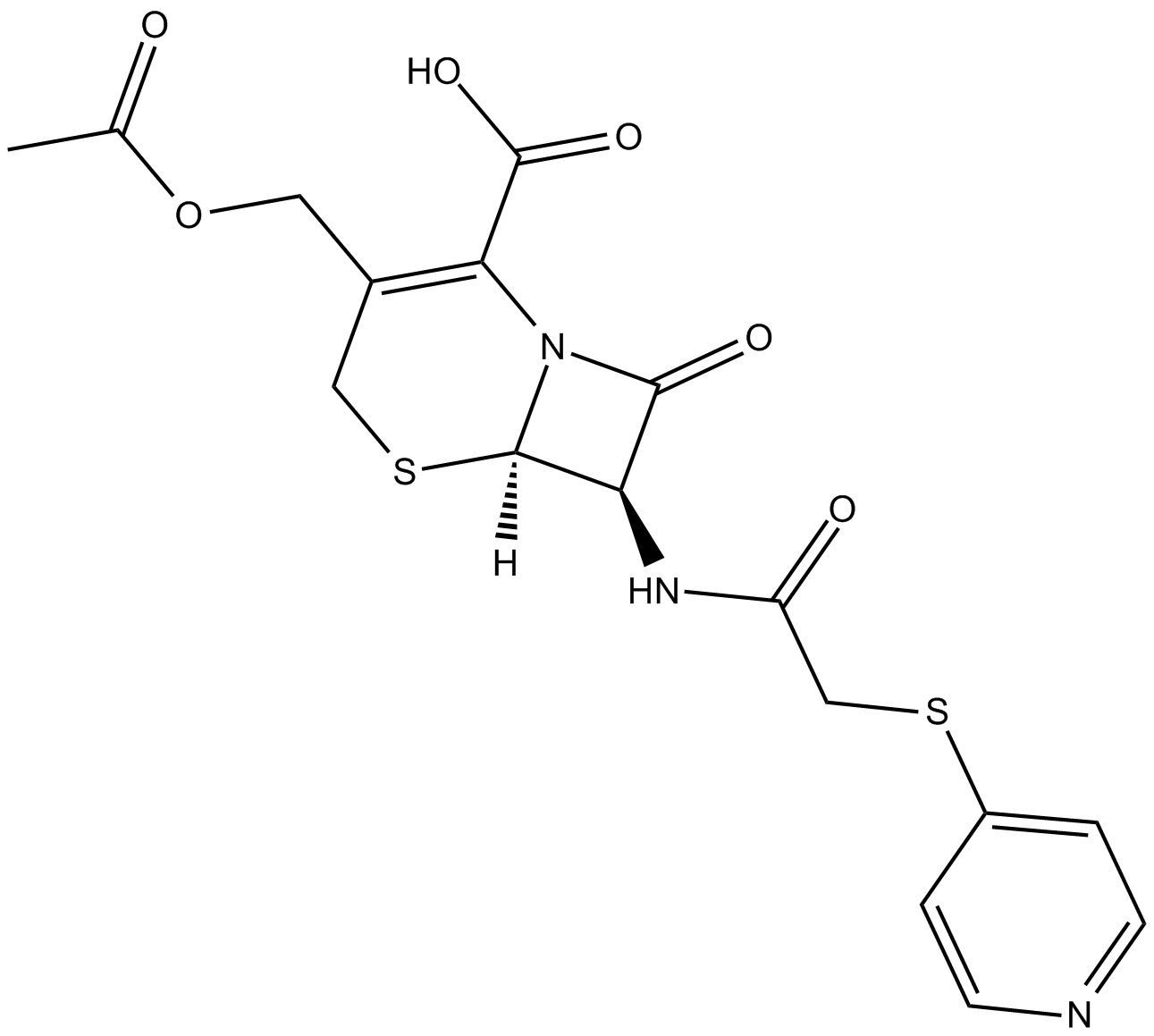
Cefapirin

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Cefapirin is the first generation cephalosporin antibiotic. Cefapirin is broadly effective against gram-negative and gram-positive bacteria [1].
The antibacterial action of cefapirin was due to the inhibition of cell-wall synthesis, mediated by its binding to penicillin binding proteins. It was mainly used in veterinary medicine for intramammary treatment of mastitis and intrauterine treatment of endometritis in cattle [1,2].
Cefapirin was an antibiotic particularly suitable for the treatment of acute otitis media. In 210 strains isolated from the auricular exudate of childrens' acute otitis media, the MIC50 and MIC90 of cefapirin were 2 and 4 mg/l for 112 strains of Haemophilus. The MIC ranged from 0.25 to 4 mg/l for ten strains of Branhamella catarrhalis (9 secreted a beta-lactamase). Cefapirin had an extremely high activity with MIC50 and 90 less than 0.06 mg/l against Streptococcus pneumoniae. For the strains of Staphylococcus aureus sensitive to meticillin, one had a MIC less than 0.06 mg/l, and 11 had a MIC of 0.25 mg/l. For 14 strains of Enterobacteriaceae studied, the MIC 50 was 8 mg/l and the MIC 90 was 32 mg/l [3].
References:
[1] Committee for veterinary medicinal products: Cefapririn. EMEA, 1-6 (2001).
[2] Ray, P. ,Knowlton, K.F.,Shang, C., et al. Development and validation of a UPLC-MS/MS method to monitor cephapirin excretion in dairy cows following intramammary infusion. PLoS One 9(11), 1-12 (2014).
[3] Simonet M, Herrmann J L, Gehanno P, et al. Activity of cefapirin against bacterial strains isolated from acute otitis media in children[J]. Pathologie-biologie, 1990, 38(5): 352-354.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 423.5 |
| Cas No. | 21593-23-7 |
| Formula | C17H17N3O6S2 |
| Synonyms | Cefadyl,Cephapirin |
| Solubility | ≤10mg/ml in DMSO;5mg/ml in dimethyl formamide |
| Chemical Name | (6R,7R)-3-[(acetyloxy)methyl]-8-oxo-7-[[(4-pyridinylthio)acetyl]amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid |
| SDF | Download SDF |
| Canonical SMILES | O=C(N[C@@H]1C(N2[C@]1([H])SCC(COC(C)=O)=C2C(O)=O)=O)CSC3=CC=NC=C3 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |