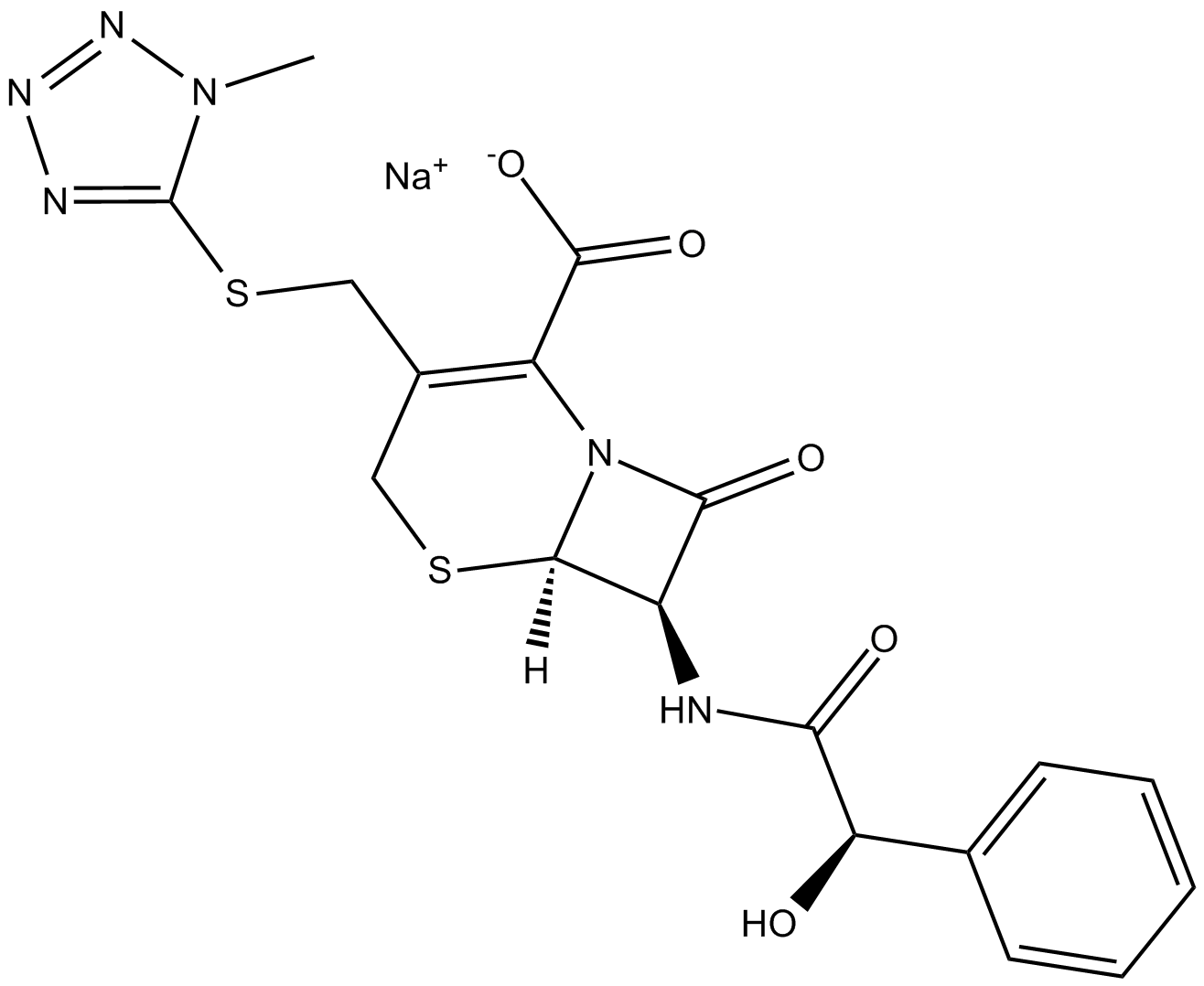
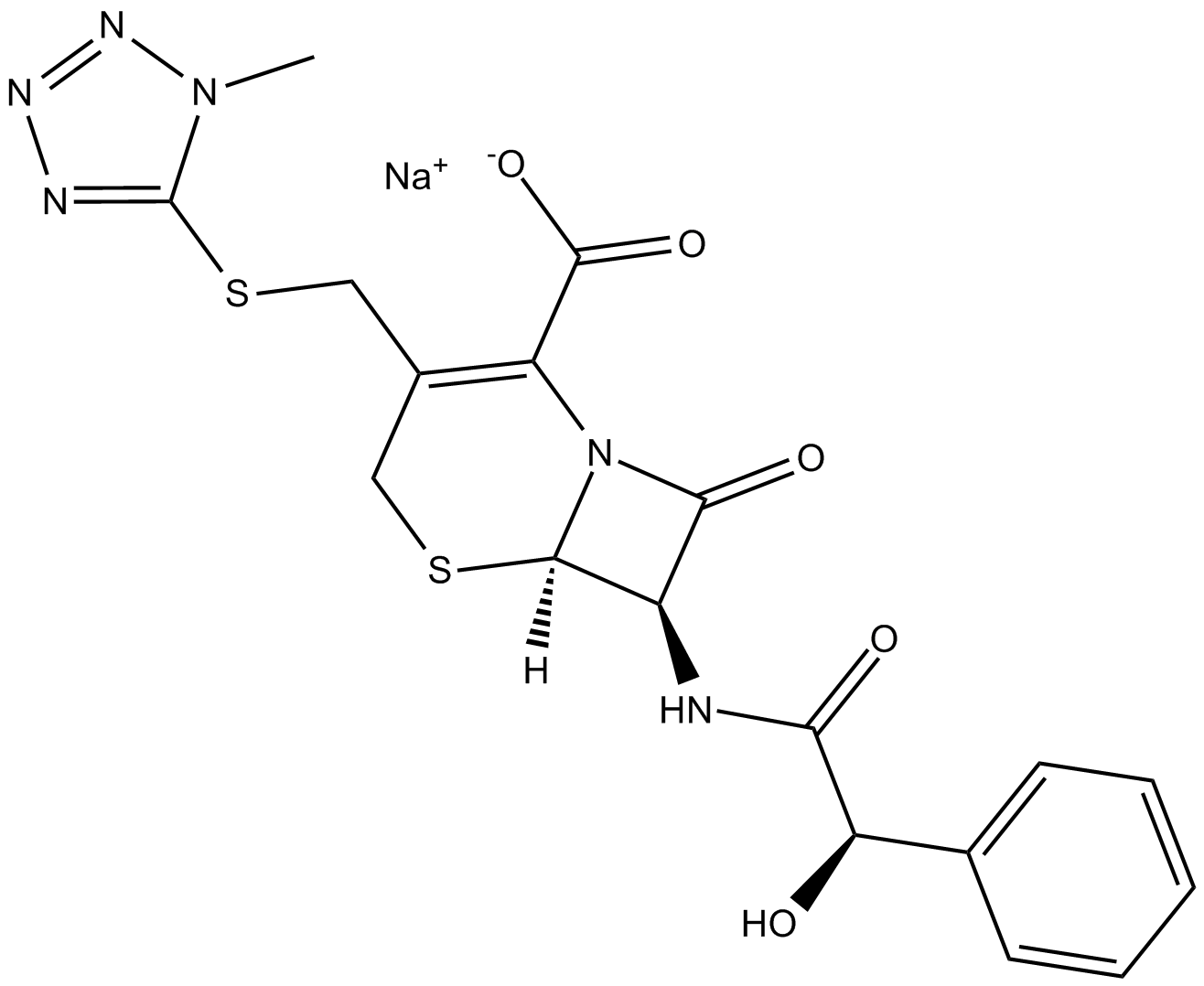
Cefamandole (sodium salt)

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
MIC: 0.25-2 mg/L for different E. coli strains
Cefamandole is a cephalosporin antibiotic.
The cephalosporins are a class of β-lactam antibiotics originally derived from the fungus Acremonium, which was previously known as "Cephalosporium".
In vitro: The in-vitro effect of cefamandole was tested against 645 strains of bacteria isolated from clinical sources. Against gram-positive organisms cefamandole showed great potency, being three- to four-fold more active than cephalexin or cefoxitin. None of the Pseudomonas aeruginosa strains were susceptible to 100 μg of cefamandole per ml [1].
In vivo: The testicular toxicity of cefamandole was evaluated in neonatal rats. Results showed that cefamandole caused delayed maturity of the germinal epithelium of neonatal rats. In rats given daily subcutaneous injections during this period, the most mature germinal cells were acrosome phase spermatids [2].
Clinical trial: A randomized, single-blind comparison of parenteral cefamandole and ampicillin was conducted in adult patients with pneumonia or purulent tracheobronchitis. Results showed that cefamandole was as effective and safe as ampicillin. Of the 14 patients treated with cefamandole, 13 were considered cured, as were 12 of the 13 treated with ampicillin [3].
References:
[1] Eickhoff TC, Ehret JM. In vitro comparison of cefoxitin, cefamandole, cephalexin, and cephalothin. Antimicrob Agents Chemother. 1976 Jun;9(6):994-9.
[2] Hoover DM, Buening MK, Tamura RN, Steinberger E. Effects of cefamandole on spermatogenic development of young CD rats. Fundam Appl Toxicol. 1989 Nov;13(4):737-46.
[3] Delgado DG, Brau CJ, Cobbs CG, Dismukes WE. Clinical and laboratory evaluation of cefamandole in the therapy of Haemophilus spp. Bronchopulmonary infections. Antimicrob Agents Chemother. 1979 Jun;15(6):807-12.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 484.5 |
| Cas No. | 30034-03-8 |
| Formula | C18H17N6O5S2·Na |
| Synonyms | Cephamandole |
| Solubility | ≤30mg/ml in DMSO;30mg/ml in dimethyl formamide |
| Chemical Name | (6R,7R)-7-[[(2R)-2-hydroxy-2-phenylacetyl]amino]-3-[[(1-methyl-1H-tetrazol-5-yl)thio]methyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid, monosodium salt |
| SDF | Download SDF |
| Canonical SMILES | O=C(N[C@@H]1C(N2[C@]1([H])SCC(CSC3=NN=NN3C)=C2C([O-])=O)=O)[C@H](O)C4=CC=CC=C4.[Na+] |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构