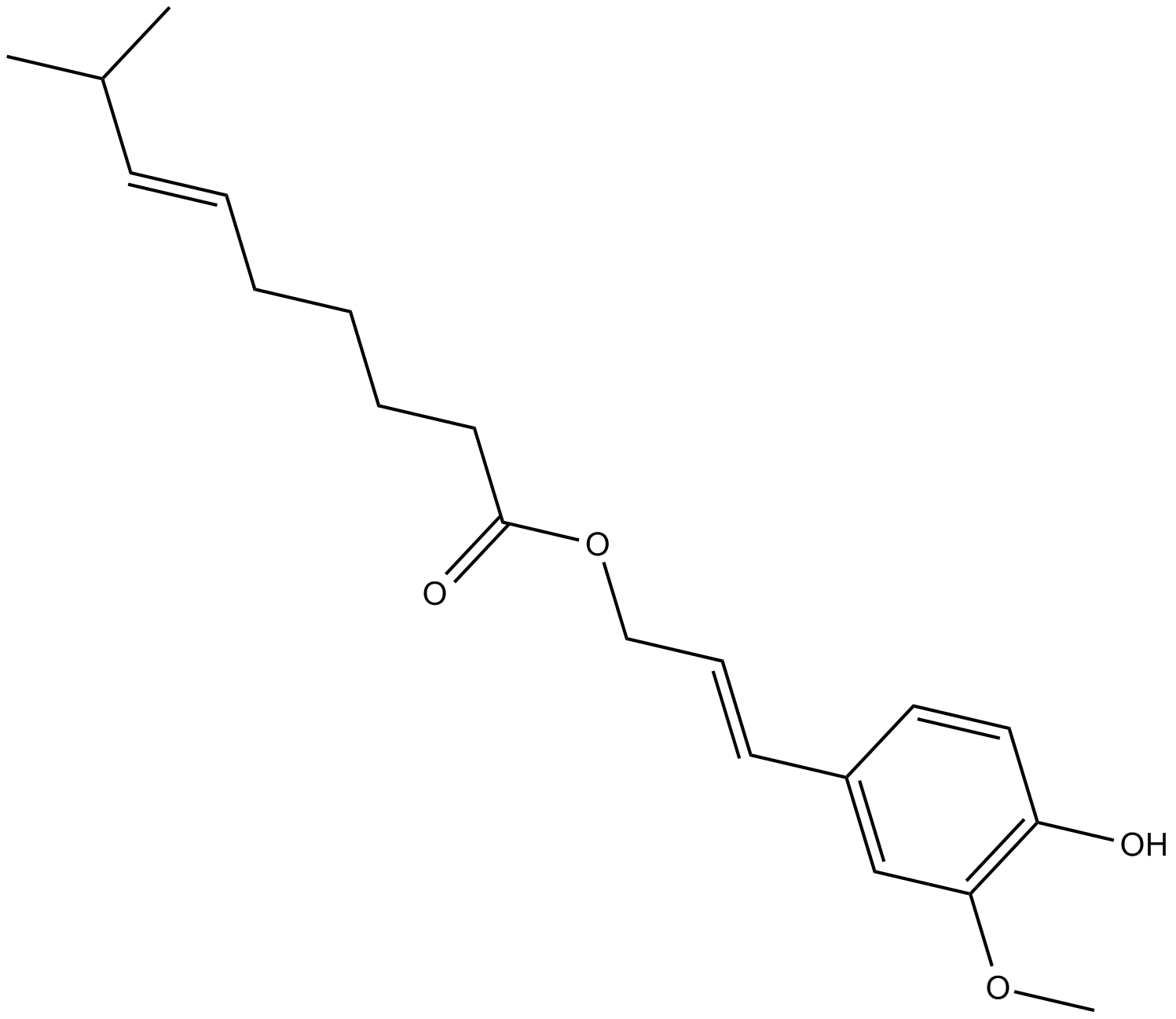
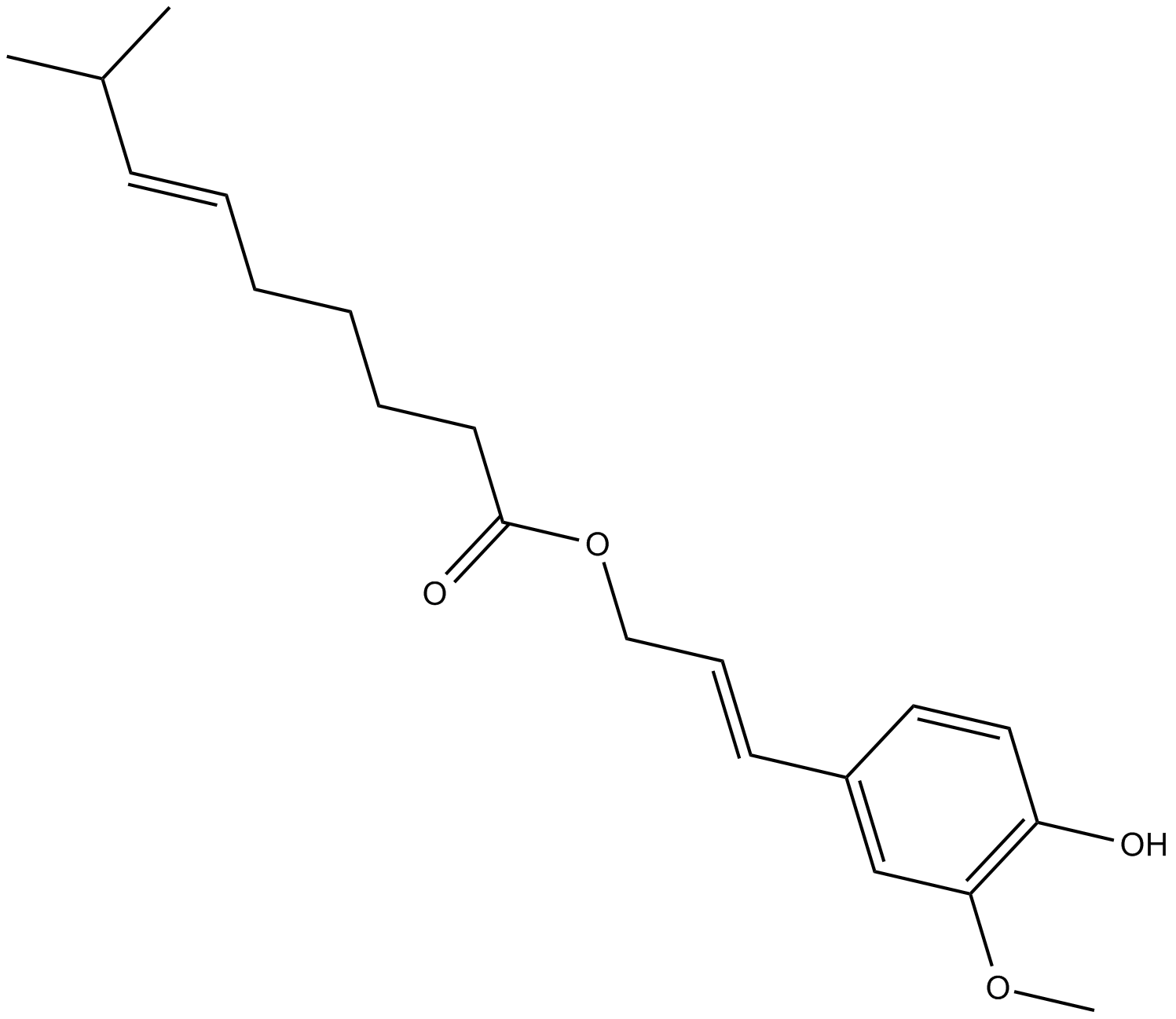
Capsiconiate

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
EC50: 3.2 μM for TRPV1
Capsiconiate is a TRPV1 agonist.
The capsaicin receptor, also termed as transient receptor potential vanilloid type 1 (TRPV1), is an non-selective cation, calcium permeable channel. The activation of TRPV1 has been reported to contribute to the various physiological activities of capsaicin.
In vitro: Capsiconiate was isolated from the fruits of the pepper, Capsicum baccatum L. var. praetermissum. The agonist activity of the capsiconiate for TRPV1 was evaluated by conducting an analysis of the intracellular calcium concentrations in TRPV1-expressing HEK293 cells. The EC50 value of capsiconiate was determined to be 3.2 microM. However, the activity was weaker than that of capsaicinor capsiate. It was found that capsiconiate had no pungency, and therefore, the low activity could be explained on the basis of the structure-activity relationship. The instability capsiconiate in aqueous solvents might also be responsible for its low activity. Even though the agonist activity for TRPV1 was lower than that of capsaicin, its activity was comparable with that of other naturally occurring compounds such as gingerol, piperine, and capsaicinol [1].
In vivo: Up to now, there is no animal in vivo data reported.
Clinical trial: So far, no clinical study has been conducted.
Reference:
[1] Kobata, K. ,Tate, H.,Iwasaki, Y., et al. Isolation of coniferyl esters from Capsicum baccatum L., and their enzymatic preparation and agonist activity for TRPV1. Phytochemistry 69(5), 1179-1184 (2008).
| Physical Appearance | A solution in ethanol. To change the solvent, simply evaporate the ethanol under a gentle stream of nitrogen and immediately add the solvent of choice. |
| Storage | Store at -20°C |
| M.Wt | 332.4 |
| Cas No. | 946572-73-2 |
| Formula | C20H28O4 |
| Synonyms | Coniferyl (E)-8-methyl-6-nonenoate |
| Solubility | ≤20mg/ml in ethanol;20mg/ml in DMSO;20mg/ml in dimethyl formamide |
| Chemical Name | 8-methyl-6E-nonenoic acid 3-(4-hydroxy-3-methoxyphenyl)-2E-propen-1-yl ester |
| SDF | Download SDF |
| Canonical SMILES | COC1=C(O)C=CC(/C=C/COC(CCCC/C=C/C(C)C)=O)=C1 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |