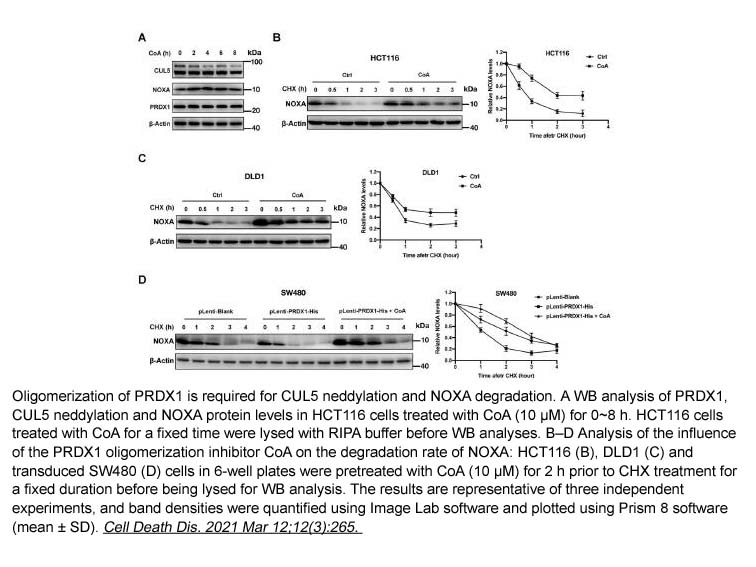
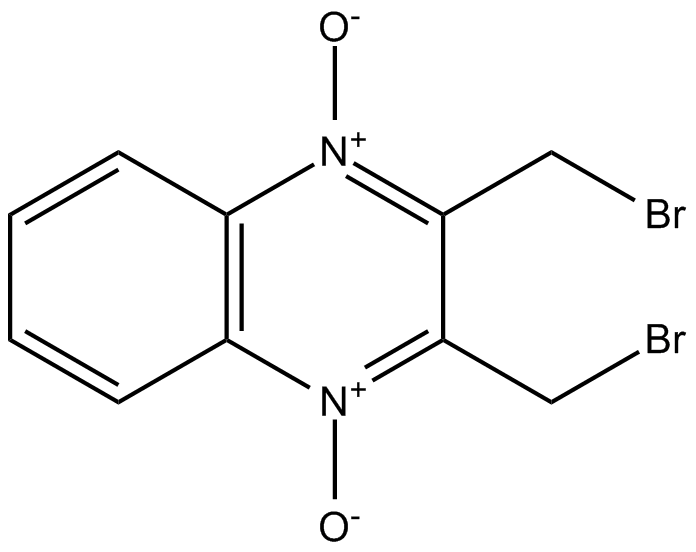
Conoidin A

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Conoidin A is a covalent inhibitor of peroxiredoxin II with IC50 value of 23 μM [2].
Peroxiredoxins are a widely conserved family of enzymes that function in antioxidant defense and signal transduction, and changes in PrxII expression are associated with a variety of human diseases, including cancer [1].
Conoidin A is a novel, cell-permeable and covalent peroxiredoxin II inhibitor with IC50 value of 23 μM [1][2]. Conoidin A bound covalently to the peroxidatic cysteine of the T. gondii enzyme peroxiredoxin II (TgPrxII), inhibiting its enzymatic activity. In human epithelial cells, Conoidin A inhibited hyperoxidation of human PrxII [1]. Conoidin A also inactivated AcePrx-1 (a peroxiredoxin from the hookworm Ancylostoma ceylanicum) by alkylating or crosslinking the catalytic cysteines with IC50 value of 374 μM [2]. Conoidin A inhibited the hyperoxidation of two mammalian peroxiredoxin homologues (PrxI and PrxII) in cells [3].
References:
[1]. Haraldsen JD1, Liu G, Botting CH, et al. IDENTIFICATION OF CONOIDIN A AS A COVALENT INHIBITOR OF PEROXIREDOXIN II. Org Biomol Chem. 2009;7:3040-3048.
[2]. Nguyen JB1, Pool CD, Wong CY, et al. Peroxiredoxin-1 from the human hookworm Ancylostoma ceylanicum forms a stable oxidized decamer and is covalently inhibited by conoidin A. Chem Biol. 2013 Aug 22;20(8):991-1001.
[3]. Liu G, Botting CH, Evans KM, et al. Optimisation of conoidin A, a peroxiredoxin inhibitor. ChemMedChem. 2010 Jan;5(1):41-5.
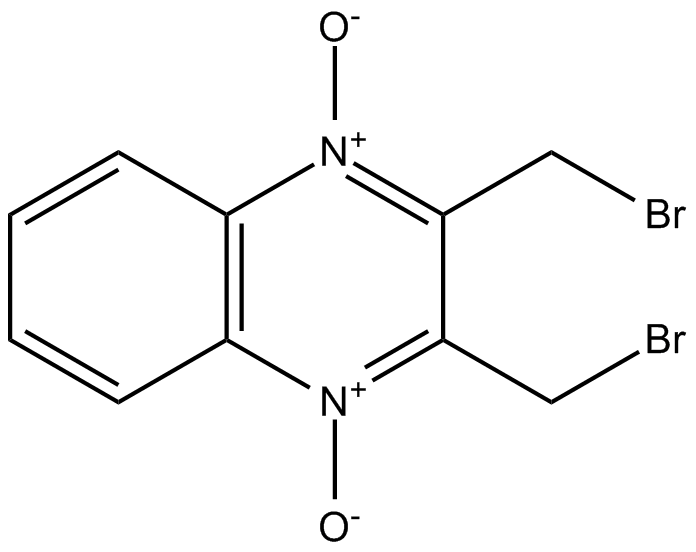
- 1. Shoufang Xu, Yilei Ma, et al. "Cullin-5 neddylation-mediated NOXA degradation is enhanced by PRDX1 oligomers in colorectal cancer." Cell Death Dis. 2021 Mar 12;12(3):265. PMID:33712558
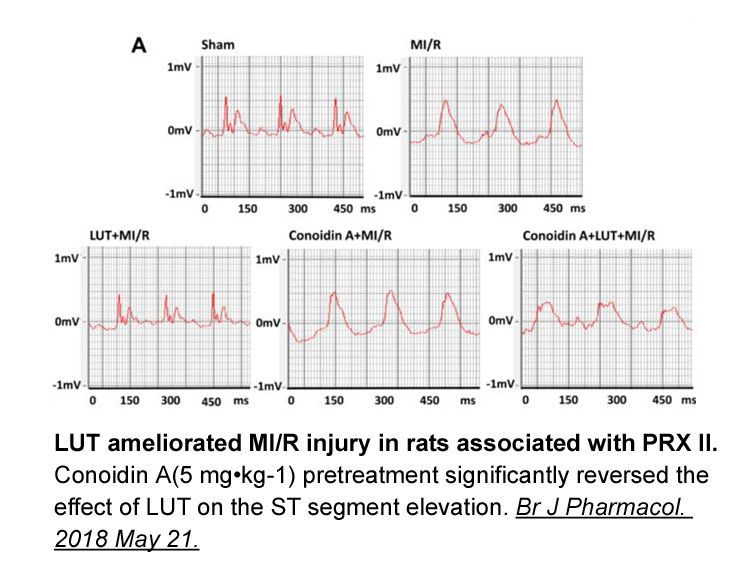
- 2. Wei B, Lin Q, et al. "Luteolin ameliorates rat myocardial ischemia-reperfusion injury through peroxiredoxin II activation." Br J Pharmacol. 2018 May 21. PMID:29782637
| Storage | Store at -20°C |
| M.Wt | 348 |
| Cas No. | 18080-67-6 |
| Formula | C10H8Br2N2O2 |
| Solubility | ≥34.8 mg/mL in DMSO; insoluble in EtOH; insoluble in H2O |
| Chemical Name | 2,3-bis(bromomethyl)-quinoxaline 1,4-dioxide |
| SDF | Download SDF |
| Canonical SMILES | [O-][N+]1=C(CBr)C(CBr)=[N+]([O-])C2=C1C=CC=C2 |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构
相关生物数据
相关生物数据