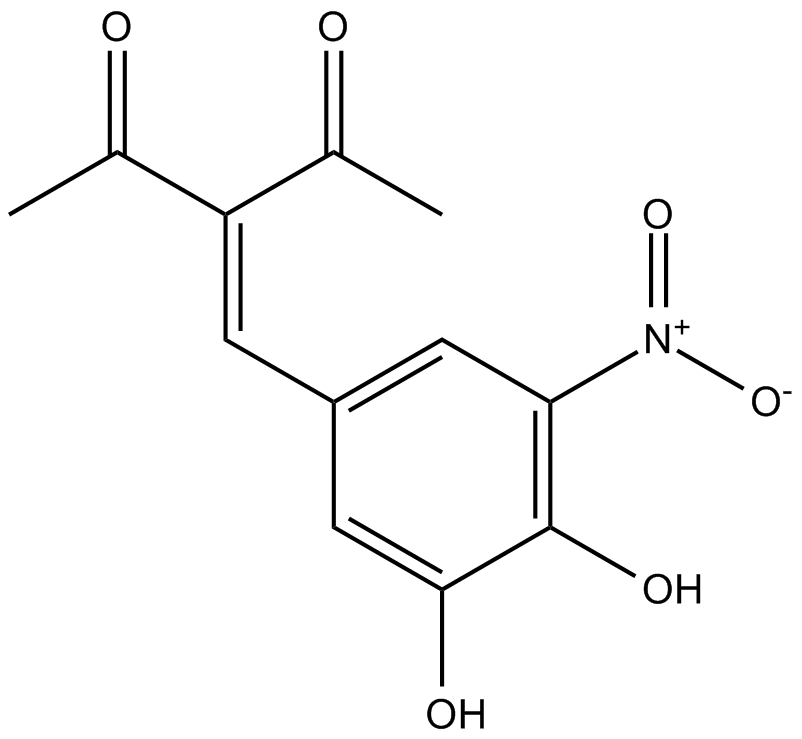
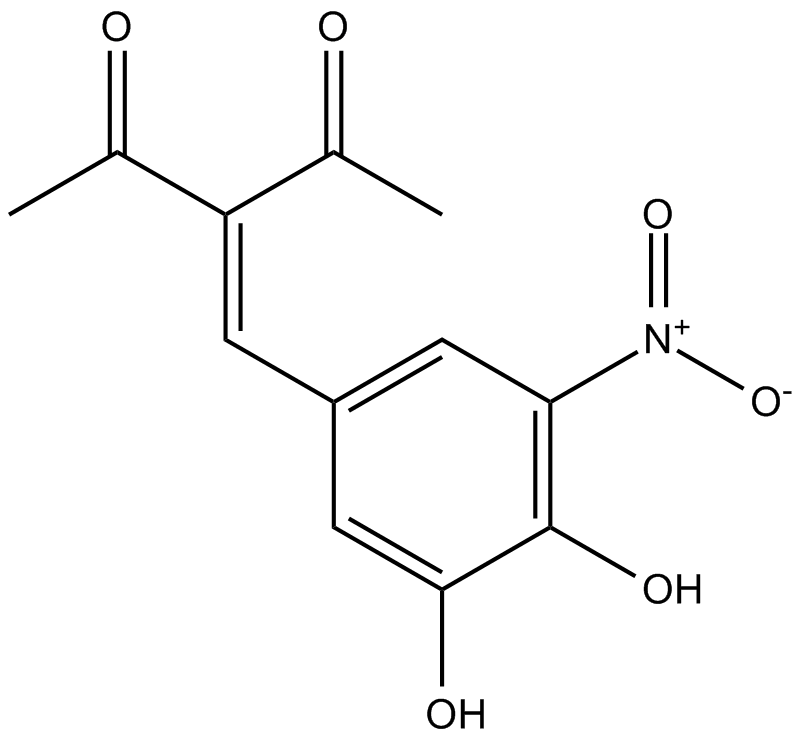
Nitecapone

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Nitecapone is S-COMT inhibitor [1].
Catechol-O-methyltransferase (COMT) plays an essential role in normal brain function and has been implicated in human disorders, such as Parkinson's disease. The COMT has been involved in the degradation of catecholamines including dopamine, epinephrine, and norepinephrine. [1].
In vitro: Nitecapone is a highly effective inhibitor of rat S-COMT with IC50 values of about 300 nM in the liver and 20 nM in the brain tissues. The Ki value in the rat liver was 23 nM. In pure recombinant COMT enzyme forms, the Ki value of nitecapone was around 1 nM [1]. Nitecapone was selective for COMT, inhibiting dopamine-β-hydroxylase, tyrosine hydroxylase, DOPA decarboxylase, and monoamine oxidase-A/B with IC50 values in the micromolar range [1].
In vivo: In rats, oral administration of nitecapone (3-30 mg/kg) in combination with levodopa and carbidopa effectively reduced 3-OMD formation and elevated serum and brain l-dopa, dopamine, DOPAC, and HVA levels. Nitecapone prevented ischemia-reperfusion injury in experimental heart surgery in rats. Nitecapone increased bicarbonate secretion from rat and human duodenum after both i.v. and intraluminal administration. High concentrations of nitecapone increased synthesis and secretion of gastric sulfomucin [1].
Reference:
[1] Mnnist P T, Kaakkola S. Catechol-O-methyltransferase (COMT): biochemistry, molecular biology, pharmacology, and clinical efficacy of the new selective COMT inhibitors[J]. Pharmacological reviews, 1999, 51(4): 593-628.
| Physical Appearance | A crystalline solid |
| Storage | Store at -20°C |
| M.Wt | 265.2 |
| Cas No. | 116313-94-1 |
| Formula | C12H11NO6 |
| Synonyms | OR-462 |
| Solubility | insoluble in H2O; ≥3.64 mg/mL in EtOH with ultrasonic; ≥60.4 mg/mL in DMSO |
| Chemical Name | 3-[(3,4-dihydroxy-5-nitrophenyl)methylene]-2,4-pentanedione |
| SDF | Download SDF |
| Canonical SMILES | OC1=C([N+]([O-])=O)C=C(/C=C(C(C)=O)/C(C)=O)C=C1O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
质量控制和MSDS
- 批次:
化学结构