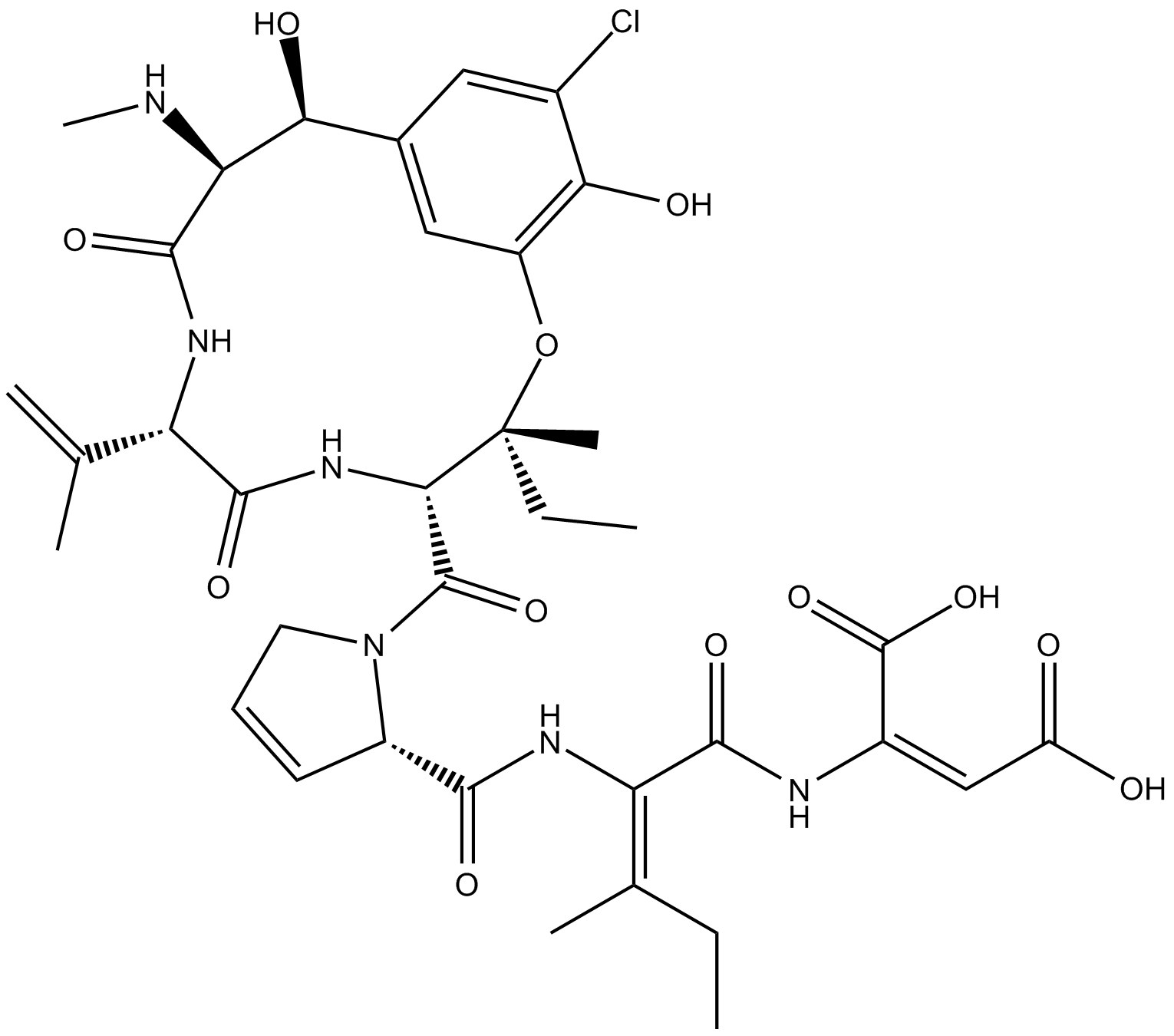
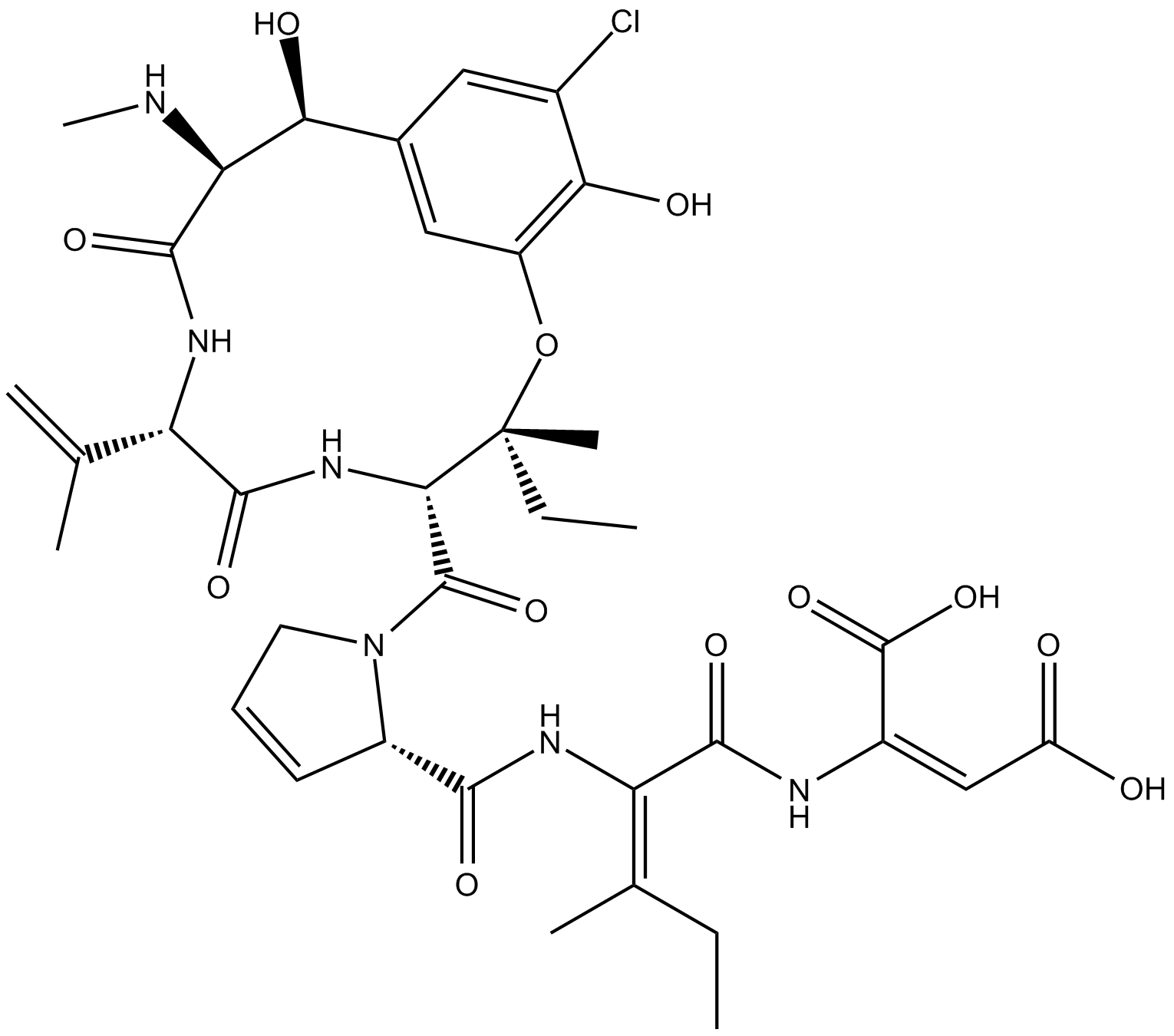
Phomopsin A

mRNA synthesis
In vitro transcription of capped mRNA with modified nucleotides and Poly(A) tail

Tyramide Signal Amplification (TSA)
TSA (Tyramide Signal Amplification), used for signal amplification of ISH, IHC and IC etc.

Phos Binding Reagent Acrylamide
Separation of phosphorylated and non-phosphorylated proteins without phospho-specific antibody

Cell Counting Kit-8 (CCK-8)
A convenient and sensitive way for cell proliferation assay and cytotoxicity assay

SYBR Safe DNA Gel Stain
Safe and sensitive stain for visualization of DNA or RNA in agarose or acrylamide gels.

Inhibitor Cocktails
Protect the integrity of proteins from multiple proteases and phosphatases for different applications.
Phomopsin A is a cyclic hexapeptide mycotoxin that inhibits β-tubulin.
Phomopsins are a family of mycotoxins produced by the fungus Phomopsis leptostomiformis grows on lupins, which cause lupinosis, a severe liver disease of grazing animals [1][2].
Microtubules are one of the major components of the cytoskeleton that are essential in several cellular functions such as cell division and morphogenesis. α- and β-tubulins polymerize into microtubules.
Phomopsin A is a cyclic hexapeptide mycotoxin that binds β-tubulin in a vinca domain, partly overlapping with the site targeted by vinblastine and other tubulin inhibitors [2][3]. Phomopsin A noncompetitively inhibited the binding of radiolabeled vinblastine to tubulin with IC50 and Ki values of 0.8 μM and 2.8 μM, respectively. Phomopsin A potently inhibited tubulin-dependent GTP hydrolysis and nucleotide exchange on tubulin [2]. Phomopsin A, a vinca domain antimitotic peptide, also inhibited microtubule assembly [3][4]. Phomopsin A inhibited microtubule growth, modulated the dynamics of microtubules, and induced the self-association of tubulin dimers into single-walled rings and spirals [4].
References:
[1]. Hamel E. Natural products which interact with tubulin in the vinca domain: maytansine, rhizoxin, phomopsin A, dolastatins 10 and 15 and halichondrin B. Pharmacol Ther. 1992;55(1):31-51.
[2]. Cormier A, Marchand M, Ravelli RB, et al. Structural insight into the inhibition of tubulin by vinca domain peptide ligands. EMBO Rep. 2008 Nov;9(11):1101-6.
[3]. Li Y, Kobayashi H, Hashimoto Y, et al. Binding selectivity of rhizoxin, phomopsin A, vinblastine, and ansamitocin P-3 to fungal tubulins: differential interactions of these antimitotic agents with brain and fungal tubulins. Biochem Biophys Res Commun. 1992 Sep 16;187(2):722-9.
[4]. Mitra A, Sept D. Localization of the antimitotic peptide and depsipeptide binding site on beta-tubulin. Biochemistry. 2004 Nov 9;43(44):13955-62.
| Physical Appearance | A solid |
| Storage | Store at -20°C |
| M.Wt | 789.2 |
| Cas No. | 64925-80-0 |
| Formula | C36H45ClN6O12 |
| Synonyms | NSC 381839 |
| Solubility | Soluble in DMSO |
| Chemical Name | (2E)-(βS)-3-chloro-β,5-dihydroxy-N-methyl-L-tyrosyl-3,4-didehydro-L-valyl-3-hydroxy-L-isoleucyl-3,4-didehydro-L-prolyl-(2E)-2,3-didehydroisoleucyl-2,3-didehydro-aspartic acid, cyclic (15→3)-ether |
| SDF | Download SDF |
| Canonical SMILES | OC(/C(NC(/C(NC([C@H]1N(C([C@@H]2NC([C@H](C(C)=C)NC([C@@H](NC)[C@H](C3=CC(O[C@@]2(CC)C)=C(O)C(Cl)=C3)O)=O)=O)=O)CC=C1)=O)=C(CC)/C)=O)=C\C(O)=O)=O |
| 运输条件 | 蓝冰运输或根据您的需求运输。 |
| 一般建议 | 不同厂家不同批次产品溶解度各有差异,仅做参考。若实验所需浓度过大至产品溶解极限,请添加助溶剂助溶或自行调整浓度。溶液形式一般不宜长期储存,请尽快用完。 |
化学结构